Abstract
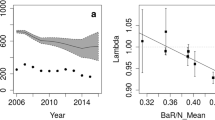
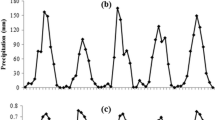
Mast seeding drives large-scale population fluctuations in forest-dwelling animals, including the wild boar Sus scrofa. Many studies of the masting effects on population dynamics of wild boars have been conducted in cool- and warm-temperate forests; however, studies on wild boar populations inhabiting subtropical forests, which have more diverse plant resources than temperate forests, are limited. The mast seeding effects of Castanopsis sieboldii on the reproductive schedule and population fluctuations of wild boars inhabiting the subtropical climate zone of the Ryukyu Islands (Japan) were investigated using 14 years of camera trap data. The reproductive schedule, estimated based on the emergence of piglets, varied in response to masting. The piglet emergence during the January–February period was higher in the years after masting than in the years that followed non-masting events, evidencing that the reproductive season started earlier and lasted longer in the years after masting. The Bayesian state-space model incorporating direct density-dependence and masting effects revealed that the relative population size was negatively affected by the population size of the previous year and positively affected by the previous year’s occurrence of masting. These observations indicate that masting is a critical factor driving population dynamics of wild boars living in subtropical forests and also highlight the importance of mast seeding effects on population fluctuations of wild boars when developing management plans for these animals.





Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
Code availability
The code supporting this study is available from the corresponding author on request.
References
Ballari SA, Barrios-García MN (2014) A review of wild boar Sus scrofa diet and factors affecting food selection in native and introduced ranges. Mammal Rev 44:124–134. https://doi.org/10.1111/mam.12015
Barrios-Garcia MN, Ballari SA (2012) Impact of wild boar (Sus scrofa) in its introduced and native range: a review. Biol Invasions 14:2283–2300. https://doi.org/10.1007/s10530-012-0229-6
Baruzzi C (2017) Friends or foes? Importance of wild ungulates as ecosystem engineers for amphibian communities. North-West J Zool 13:320–325
Bergeron P, Réale D, Humphries MM, Garant D (2011) Anticipation and tracking of pulsed resources drive population dynamics in eastern chipmunks. Ecology 92:2027–2034. https://doi.org/10.1890/11-0766.1
Bieber C, Ruf T (2005) Population dynamics in wild boar Sus scrofa: ecology, elasticity of growth rate and implications for the management of pulsed resource consumers. J Appl Ecol 42:1203–1213. https://doi.org/10.1111/j.1365-2664.2005.01094.x
Bogdziewicz M, Zwolak R, Redosh L et al (2016) Negative effects of density on space use of small mammals differ with the phase of the masting-induced population cycle. Ecol Evol 6:8423–8430. https://doi.org/10.1002/ece3.2513
Bowyer RT, Bleich VC, Stewart KM, Monteith KL (2014) Density dependence in ungulates: a review of causes, and concepts with some clarifications. Calif Fish Game 100:557–572
Burton AC, Neilson E, Moreira D et al (2015) Wildlife camera trapping: a review and recommendations for linking surveys to ecological processes. J Appl Ecol 52:675–685. https://doi.org/10.1111/1365-2664.12432
Cachelou J, Saint-Andrieux C, Baubet E et al (2022) Does mast seeding shape mating time in wild boar? A Comp Study Biol Lett 18:20220213. https://doi.org/10.1098/rsbl.2022.0213
Canu A, Scandura M, Merli E et al (2015) Reproductive phenology and conception synchrony in a natural wild boar population. Hystrix Ital J Mammal 26:77–84. https://doi.org/10.4404/hystrix-26.2-11324
Choquenot D (1998) Testing the relative influence of intrinsic and extrinsic variation in food availability on feral pig populations in Australia’s rangelands. J Anim Ecol 67:887–907
Cutini A, Chianucci F, Chirichella R et al (2013) Mast seeding in deciduous forests of the northern Apennines (Italy) and its influence on wild boar population dynamics. Ann for Sci 70:493–502. https://doi.org/10.1007/s13595-013-0282-z
Ebihara I (2009) Study of the hunting practices and knowledge of a trap-hunter in Iriomote Island, Okinawa: based on trapping field maps over a period of 11 years. Bull Natl Mus Ethnology 34:131–165. (in Japanese with English summary)
Gamelon M, Focardi S, Baubet E et al (2017) Reproductive allocation in pulsed-resource environments: a comparative study in two populations of wild boar. Oecologia 183:1065–1076. https://doi.org/10.1007/s00442-017-3821-8
Gamelon M, Nater CR, Baubet É et al (2021a) Efficient use of harvest data: a size-class-structured integrated population model for exploited populations. Ecography 43:1–15. https://doi.org/10.1111/ecog.05738
Gamelon M, Touzot L, Baubet É et al (2021b) Effects of pulsed resources on the dynamics of seed consumer populations: a comparative demographic study in wild boar. Ecosphere 12:e03395. https://doi.org/10.1002/ecs2.3395
Gelman A, Carlin J, Stern HS, Rubin DB (2004) Bayesian data analysis, 2nd edn. Chapman and Hall/CRC, New York
Gosling LM (1986) Selective abortion of entire litters in the coypu: adaptive control of offspring production in relation to quality and sex. Am Nat 127:772–795
Guo W, Cao G, Quan R-C (2017) Population dynamics and space use of wild boar in a tropical forest, Southwest China. Glob Ecol Conserv 11:115–124. https://doi.org/10.1016/j.gecco.2017.04.005
Handa Y (2000) Questionnaires and hunting statistics for the habitation of Ryukyu wild boars in Amami Islands. J Jpn Vet Med Assoc 53:525–528. (in Japanese)
Hebeisen C, Fattebert J, Baubet E, Fischer C (2008) Estimating wild boar (Sus scrofa) abundance and density using capture–resights in Canton of Geneva, Switzerland. Eur J Wildl Res 54:391–401. https://doi.org/10.1007/s10344-007-0156-5
Heise-Pavlov PM, Heise-Pavlov SR (2003) Feral pigs in tropical lowland rainforest of northeastern Australia: ecology, zoonoses and management. Wildl Biol 9:21–27. https://doi.org/10.2981/wlb.2003.060
Iijima H, Ueno M (2016) Spatial heterogeneity in the carrying capacity of sika deer in Japan. J Mammal 97:734–743. https://doi.org/10.1093/jmammal/gyw001
Imperio S, Focardi S, Santini G, Provenzale A (2012) Population dynamics in a guild of four Mediterranean ungulates: density-dependence, environmental effects and inter-specific interactions. Oikos 121:1613–1626
Inzani E, Marshall HH, Thompson FJ et al (2019) Spontaneous abortion as a response to reproductive conflict in the banded mongoose. Biol Lett 15:20190529. https://doi.org/10.1098/rsbl.2019.0529
Ishigaki C, Shinzato T, Aramoyto M, Wu L (2007) Feed plant, dressing and utilization of carcass of Ryukyuan wild boar in Iriomote Island. Sci Bull Coll Agric Univ Ryukyus 54:23–27. (in Japanese with English summary)
Itô Y (1997) Diversity of forest tree species in Yanbaru, the northern part of Okinawa Island. Plant Ecol 133:125–133
Jędrzejewska B, Jędrzejewski W, Bunevich AN et al (1997) Factors shaping population densities and increase rates of ungulates in Białowieża Primeval Forest (Poland and Belarus) in the 19th and 20th centuries. Acta Theriol 42:399–451
Kawamura A, Chang C-H, Kawamura Y (2016) Middle Pleistocene to Holocene mammal faunas of the Ryukyu Islands and Taiwan: an updated review incorporating results of recent research. Quatern Int 397:117–135
Kelly D, Koenig WD, Liebhold AM (2008) An intercontinental comparison of the dynamic behavior of mast seeding communities. Popul Ecol 50:329–342. https://doi.org/10.1007/s10144-008-0114-4
Kelly D, Sork VL (2002) Mast seeding in perennial plants: why, how, where? Annu Rev Ecol Syst 33:427–447. https://doi.org/10.2307/3069269
Maillard D, Fournier P (2004) Timing and synchrony of births in the wild boar (Sus scrofa Linnaeus, 1758) in a Mediterranean habitat: the effect of food availability. Galemys 16:67–74
Mammalogical Society of Japan (ed) (1997) Red data book—Mammals in Japan. Bun-ichi Co., Ltd, Tokyo. (in Japanese)
Massei G, Genov PV (2004) The environmental impact of wild boar. Galemys 16:135–145
Massei G, Kindberg J, Licoppe A et al (2015) Wild boar populations up, numbers of hunters down? A review of trends and implications for Europe: wild boar and hunter trends in Europe. Pest Manag Sci 71:492–500. https://doi.org/10.1002/ps.3965
Mauget R (1982) Seasonality of reproduction in the wild boar. In: Cole DJA, Foxcroft GR (eds) Control of pig reproduction. Butterworth Press, London, pp 509–526
Mo M, Meade J, Price J et al (2022) Synchronous abortion events in the grey-headed flying-fox (Pteropus poliocephalus). Pac Conserv Biol. https://doi.org/10.1071/PC21060
Mori E, Ferretti F, Lagrotteria A et al (2020) Impact of wild boar rooting on small forest-dwelling rodents. Ecol Res 35:675–681. https://doi.org/10.1111/1440-1703.12113
Ohashi H, Saito M, Horie R et al (2013) Differences in the activity pattern of the wild boar Sus scrofa related to human disturbance. Eur J Wildl Res 59:167–177. https://doi.org/10.1007/s10344-012-0661-z
Ohdachi SD, Ishibashi Y, Fukui D, Saitoh T (eds) (2015) The wild mammals of Japan, 2nd edn. Shokado, Kyoto
Omori A, Hosoi E (2022) Contribution of acorn masting to food composition and body condition of and crop damage by wild boars (Sus scrofa) inhabiting evergreen forests in Japan. Mammal Study 47:65–75. https://doi.org/10.3106/ms2021-0024
Onodera R, Akimoto Y, Shimada T, Saitoh T (2017) Different population responses of three sympatric rodent species to acorn masting—the role of tannin tolerance. Popul Ecol 59:29–43. https://doi.org/10.1007/s10144-017-0570-9
Ostfeld R, Keesing F (2000) Pulsed resources and community dynamics of consumers in terrestrial ecosystems. Trends Ecol Evol 15:232–237
Palencia P, Vada R, Zanet S et al (2023) Not just pictures: utility of camera trapping in the context of African swine fever and wild boar management. Transbound Emerg Dis. https://doi.org/10.1155/2023/7820538
Plummer M (2003) JAGS: A program for analysis of Bayesian graphical models using Gibbs sampling. In: Hornik K, Leisch F, Zeileis A (eds) Proceedings of the 3rd international workshop on distributed statistical computing. Vienna, Austria. https://www.r-project.org/conferences/DSC-2003/Proceedings/Plummer.pdf
R Core Team (2022). R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rossi S, Fromont E, Pontier D et al (2005) Incidence and persistence of classical swine fever in free-ranging wild boar (Sus scrofa). Epidemiol Infect 133:559–568. https://doi.org/10.1017/S0950268804003553
Saitoh T, Bjørnstad ON, Stenseth NC (1999) Density dependence in voles and mice: a comparative study. Ecology 80:638–650
Saitoh T, Osawa J, Takanishi T et al (2007) Effects of acorn masting on population dynamics of three forest-dwelling rodent species in Hokkaido, Japan. Popul Ecol 49:249–256. https://doi.org/10.1007/s10144-007-0041-9
Saitoh T, Vik JO, Stenseth NC et al (2008) Effects of acorn abundance on density dependence in a Japanese wood mouse (Apodemus speciosus) population. Popul Ecol 50:159–167. https://doi.org/10.1007/s10144-008-0076-6
Santos P, Fernández-Llario P, Fonseca C et al (2006) Habitat and reproductive phenology of wild boar (Sus scrofa) in the western Iberian Peninsula. Eur J Wildl Res 52:207–212. https://doi.org/10.1007/s10344-005-0025-z
Servanty S, Gaillard J-M, Toïgo C et al (2009) Pulsed resources and climate-induced variation in the reproductive traits of wild boar under high hunting pressure. J Anim Ecol 78:1278–1290
Shimada T, Saitoh T (2006) Re-evaluation of the relationship between rodent populations and acorn masting: a review from the aspect of nutrients and defensive chemicals in acorns. Popul Ecol 48:341–352
Stenseth NC (1999) Population cycles in voles and lemmings: density dependence and phase dependence in a stochastic world. Oikos 87:427–461. https://doi.org/10.2307/3546809
Stephens RB, Hobbie EA, Lee TD, Rowe RJ (2019) Pulsed resource availability changes dietary niche breadth and partitioning between generalist rodent consumers. Ecol Evol 9:10681–10693. https://doi.org/10.1002/ece3.5587
Takahashi R, Gushiken R, Nagata K et al (2019) Genetic structure of the Ryukyu wild boar population on Tokunoshima Island (Japan) based on modern and ancient DNA analyses: evidence of recent gene flow from pigs to wild boars. Mammal Study 44:157. https://doi.org/10.3106/ms2018-0046
Takashima A, Kinjo T (2018) Annual variability in seedfall of the dominant tree species of a natural forest in the Yambaru area of Okinawa Island. Kyushu J for Res 71:109–112
Takashima A, Kudaka N, Abe S et al (2021) Mast seeding monitoring on Castanopsis sieboldii in the Central Ryukyus of Nansei Islands using a binocular. Kyushu J for Res 74:69–72
Takashima A, Matsumoto K (2017) From field research sites (34) Subtropical Field Science Center, Faculty of Agriculture, University of the Ryukyus. Jpn J Ecol 67:61–66. (in Japanese)
Takashima A, Oshima J, Kudaka M, Saito K (2014) Stand structure of the subtropical natural forest in the Yambaru area of Okinawa Island: a comparison of a 60-year-old secondary forest and a mature non-clear-cut forest. Jpn J Forest Plan 48:27–34
Teruya K (2015) Research on acorn production phenology, granivorous pest insects of Fagaceous tree species and its soil environment in subtropical Okinawa Island. Dissertation, Kagoshima University
Touzot L, Schermer É, Venner S et al (2020) How does increasing mast seeding frequency affect population dynamics of seed consumers? Wild boar as a case study. Ecol Appl 30:e02134. https://doi.org/10.1002/eap.2134
Uni S, Fukuda M, Uga S et al (2021) Prevalence of Onchocerca japonica and O. takaokai infections in the Japanese wild boar, Sus scrofa leucomystax, and the Ryukyu wild boar, S. s. riukiuanus, in Japan. Parasitol Int 83:102313. https://doi.org/10.1016/j.parint.2021.102313
Wright JS (2002) Plant diversity in tropical forests: a review of mechanisms of species coexistence. Oecologia 130:1–14. https://doi.org/10.1007/s004420100809
Yamada E, Anezaki T, Hongo H (2018) Tooth outline shape analysis of the Ryukyu wild boar (Sus scrofa riukiuanus) and the Japanese wild boar (S. s. leucomystax) by geometric morphometrics. Mammal Study 43:99–107
Yamada H, Miyaura T (2005) Geographic variation in nut size of Castanopsis species in Japan. Ecol Res 20:3–9. https://doi.org/10.1007/s11284-004-0006-9
Yang LH, Bastow JL, Spence KO, Wright AN (2008) What can we learn from resource pulses. Ecology 89:621–634
Acknowledgements
We are grateful to Natsuko Kudaka and Masahiro Kudaka for their assistance with the field research and Katsushi Nakata for supporting this research project and providing valuable field information. We also thank Takashi Saitoh for his help with the initial data analysis and two anonymous reviewers for their constructive comments on the manuscript.
Funding
This study was funded by the Environment Research and Technology Development Fund (JPMEERF20154003 and JPMEERF20184004) of the Environmental Restoration and Conservation Agency of Japan.
Author information
Authors and Affiliations
Contributions
NK conceived, designed, and conducted the field survey. TS and HI analyzed the data. TS led the writing of the manuscript. All authors contributed to improving the draft manuscript.
Corresponding author
Ethics declarations
Ethics approval
The methods and study design were approved by the Ministry of the Environment, Japan.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shimada, T., Iijima, H. & Kotaka, N. Wild boar population fluctuations in a subtropical forest: the crucial role of mast seeding in Ryukyu Islands, Japan. Eur J Wildl Res 70, 41 (2024). https://doi.org/10.1007/s10344-024-01797-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10344-024-01797-0