Abstract
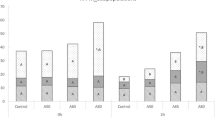
Sperm cryopreservation protocols for brown bear (Ursus arctos) require the centrifugation of semen samples to increase sperm concentration and to clean urine in contaminated samples. We evaluated the effect of centrifugation regimes (time and relative centrifugal force—RCF) on the quantity of sperm recovered and the quality of post-thawed sperm. Thirteen brown bears were electroejaculated. The ejaculates were diluted 1:1 in Tris–citric acid–glucose (TCG) extender and centrifuged with different RCF/time combinations: 600×g, 1,200×g and 2,400×g, for 3, 6 or 12 min. After centrifugation, spermatozoa were diluted in TES–Tris–fructose extender with egg yolk and glycerol (final glycerol concentration of 8%) and frozen in 0.25-mL straws. In the post-thawed semen, motility was assessed by CASA, and acrosomal status (PNA-FITC), viability (SYBR-14 with propidium iodide) and chromatin status (SCSA) were determined by flow cytometry. The longest centrifugation time (12 min) significantly decreased some motility parameters. Sperm recovery significantly decreased in brown bear at 600×g. Our results suggest that brown bear spermatozoa are more sensitive to long centrifugation times than to high RCF. Centrifugation regimes showed no effects on the post-thawing chromatin status. We recommend preparing the brown bear semen for freezing by centrifugation 1,200×g or 2,400×g for 6 min, after electroejaculation and dilution 1:1 in TCG extender, since these procedures increase the spermatozoa recovery without harmful effects on the post-thawed quality of brown bear spermatozoa.



Similar content being viewed by others
References
Aitken RJ, Clarkson JS (1988) Significance of reactive oxygen species and antioxidants in defining the efficacy of sperm preparation techniques. J Androl 9:367–376
Alvarez JG, Lasso JL, Blasco L, Nunez RC, Heyner S, Caballero PP, Storey BT (1993) Centrifugation of human spermatozoa induces sublethal damage; separation of human spermatozoa from seminal plasma by a dextran swim-up procedure without centrifugation extends their motile lifetime. Hum Reprod 8:1087–1092
Anel L, Alvarez M, Martinez-Pastor F, Gomes S, Nicolas M, Mata M, Martinez AF, Borragan S, Anel E, de Paz P (2008) Sperm cryopreservation in brown bear (Ursus arctos): preliminary aspects. Reprod Domest Anim 43(Suppl 4):9–17
Anel L, Gomes-Alves S, Alvarez M, Borragan S, Anel E, Nicolas M, Martinez-Pastor F, de Paz P (2010) Effect of basic factors of extender composition on post-thawing quality of brown bear electroejaculated spermatozoa. Theriogenology 74:643–651
Brinsko SP, Crockett EC, Squires EL (2000) Effect of centrifugation and partial removal of seminal plasma on equine spermatozoal motility after cooling and storage. Theriogenology 54:129–136
Cardullo RA, Cone RA (1986) Mechanical immobilization of rat sperm does not change their oxygen consumption rate. Biol Reprod 34:820–830
Carvajal G, Cuello C, Ruiz M, Vazquez JM, Martinez EA, Roca J (2004) Effects of centrifugation before freezing on boar sperm cryosurvival. J Androl 25:389–396
Carver DA, Ball BA (2002) Lipase activity in stallion seminal plasma and the effect of lipase on stallion spermatozoa during storage at 5 degrees C. Theriogenology 58:1587–1595
Cochran JD, Amann RP, Froman DP, Pickett BW (1984) Effects of centrifugation, glycerol level, cooling to 5 degrees C, freezing rate and thawing rate on the post-thaw motility of equine sperm. Theriogenology 22:25–38
Crockett EC, Graham JK, Bruemmer JE, Squires EL (2001) Effect of cooling of equine spermatozoa before freezing on post-thaw motility: preliminary results. Theriogenology 55:793–803
Garcia-Macias V, Martinez-Pastor F, Alvarez M, Borragan S, Chamorro CA, Soler AJ, Anel L, de Paz P (2006) Seasonal changes in sperm chromatin condensation in ram (Ovis aries), Iberian red deer (Cervus elaphus hispanicus), and brown bear (Ursus arctos). J Androl 27:837–846
Griggers S, Paccamonti DL, Thompson RA, Eilts BE (2001) The effects of pH, osmolarity and urine contamination on equine spermatozoal motility. Theriogenology 56:613–622
Ishikawa A, Matsu M, Sakamoto H, Katagiri S, Takahashi Y (2002) Cryopreservation of the semen collected by electroejaculation from the Hokkaido brown bear (Ursus arctos yesoensis). J Vet Med Sci 64:373–376
Jasko DJ, Lein DH, Foote RH (1991) The repeatability and effect of season on seminal characteristics and computer-aided sperm analysis in the stallion. Theriogenology 35:317–327
Katkov II, Mazur P (1998) Influence of centrifugation regimes on motility, yield, and cell associations of mouse spermatozoa. J Androl 19:232–241
Kawakami E, Morita Y, Hori T, Tsutsui T (2002) Lectin-binding characteristics and capacitation of canine epididymal spermatozoa. J Vet Med Sci 64:543–549
Kim SC, Kim HW (1998) Effects of nitrogenous components of urine on sperm motility: an in vitro study. Int J Androl 21:29–33
Kojima E, Tsuruga H, Komatsu T, Murase T, Tsubota T, Kita I (2001) Characterization of semen collected from beagles and captive Japanese black bears (Ursus thibetanus japonicus). Theriogenology 55:717–731
Makler A, David R, Blumenfeld Z, Better OS (1981) Factors affecting sperm motility. VII. Sperm viability as affected by change of pH and osmolarity of semen and urine specimens. Fertil Steril 36:507–511
Matas C, Decuadro G, Martinez-Miro S, Gadea J (2007) Evaluation of a cushioned method for centrifugation and processing for freezing boar semen. Theriogenology 67:1087–1091
Ng FL, Liu DY, Baker HW (1992) Comparison of Percoll, mini-Percoll and swim-up methods for sperm preparation from abnormal semen samples. Hum Reprod 7:261–266
Nicolas N, Alvarez M, Gomes-Alves S, Mata-Campuzano M, Borragán S, Martinez-Pastor F, de Paz P, Anel L (2011) Effects on brown bear (Ursus arctos) spermatozoa freezability of different extender and dilution ratios used for pre-freezing centrifugation. Eur J Wildl Res 57:259–266
Okano T, Murase T, Tsubota T (2004a) Electroejaculation and semen cryopreservation of free-ranging Japanese black bears (Ursus thibetanus japonicus). J Vet Med Sci 66:1371–1376
Okano T, Murase T, Asano M, Tsubota T (2004b) Effects of final dilution rate, sperm concentration and times for cooling and glycerol equilibration on post-thaw characteristics of canine spermatozoa. J Vet Med Sci 66:1359–1364
Okano T, Murase T, Yayota C, Komatsu T, Miyazawa K, Asano M, Tsubota T (2006) Characteristics of captive Japanese black bears (Ursus thibetanus japonicus) semen collected by electroejaculation with different voltages for stimulation and frozen-thawed under different conditions. Anim Reprod Sci 95:134–143
Parinaud J, Le LD, Vieitez G, Griveau JF, Milhet P, Richoilley G (1997) Enhancement of motility by treating spermatozoa with an antioxidant solution (Sperm-Fit) following ejaculation. Hum Reprod 12:2434–2436
Pellicer-Rubio MT, Magallon T, Combarnous Y (1997) Deterioration of goat sperm viability in milk extenders is due to a bulbourethral 60-kilodalton glycoprotein with triglyceride lipase activity. Biol Reprod 57:1023–1031
Pickett BW, Sullivan JJ, Byers WW, Pace MM, Remmenga EE (1975) Effect of centrifugation and seminal plasma on motility and fertility of stallion and bull spermatozoa. Fertil Steril 26:167–174
Rijsselaere T, Van SA, Maes D, de Kruif A (2002) Effect of centrifugation on in vitro survival of fresh diluted canine spermatozoa. Theriogenology 57:1669–1681
Ritar AJ, Salamon S (1982) Effects of seminal plasma and of its removal and of egg yolk in the diluent on the survival of fresh and frozen-thawed spermatozoa of the Angora goat. Aust J Biol Sci 35:305–312
Rota A, Strom B, Linde-Forsberg C (1995) Effects of seminal plasma and three extenders on canine semen stored at 4 degrees C. Theriogenology 44:885–900
Schafer-Somi S, Kluger S, Knapp E, Klein D, Aurich C (2006) Effects of semen extender and semen processing on motility and viability of frozen-thawed dog spermatozoa. Theriogenology 66:173–182
Sieme H, Katila T, Klug E (2004) Effect of semen collection practices on sperm characteristics before and after storage and on fertility of stallions. Theriogenology 61:769–784
Spindler RE, Huang Y, Howard JG, Wang P, Zhang H, Zhang G, Wildt DE (2004) Acrosomal integrity and capacitation are not influenced by sperm cryopreservation in the giant panda. Reproduction 127:547–556
Varisli O, Uguz C, Agca C, Agca Y (2009) Various physical stress factors on rat sperm motility, integrity of acrosome, and plasma membrane. J Androl 30:75–86
Waite JA, Love CC, Brinsko SP, Teague SR, Salazar JL Jr, Mancill SS, Varner DD (2008) Factors impacting equine sperm recovery rate and quality following cushioned centrifugation. Theriogenology 70:704–714
Way AL, Griel LC Jr, Killian GJ (2000) Effects of accessory sex gland fluid on viability, capacitation, and the acrosome reaction of cauda epididymal bull spermatozoa. J Androl 21:213–219
Weiss S, Janett F, Burger D, Hassig M, Thun R (2004) The influence of centrifugation on quality and freezability of stallion semen. Schweiz Arch Tierheilkd 146:285–293
Woelders H (1997) Fundamentals and recent development in cryopreservation of bull and boar semen. Vet Q 19:135–138
Acknowledgements
This work was supported in part by CICYT (CGL 2007-63748/BOS) and CANTUR S.A. Felipe Martínez-Pastor was supported by the Ramón y Cajal program (Spanish Ministry of Science and Innovation). The authors thank Miguel Angel Marañon and the game keepers of the Cabarceno Nature Park, Susana Gomes Alves, María Mata Campuzano, Manuel Alvarez Rodriguez, Julio Tamayo Canul and Elena Lopez Urueña.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by C. Gortázar
Rights and permissions
About this article
Cite this article
Nicolas, M., Alvarez, M., Anel, E. et al. Spermatozoa recovery and post-thawing quality of brown bear ejaculates is affected for centrifugation regimes. Eur J Wildl Res 58, 77–84 (2012). https://doi.org/10.1007/s10344-011-0544-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10344-011-0544-8