Abstract
In contrast to old-growth forests, early-successional stands remain understudied despite potentially harbouring species of conservation interest. With this work, focused on hazel grouse Tetrastes bonasia, a cryptic and indicator species known to select for close-to-natural forests, we evaluated winter densities, home range, microhabitat selection and diet, combining DNA-based mark-recapture and metabarcoding from faecal samples. In total, 216 droppings, collected over 2 years along forest transects in the Italian Alps, were successfully genotyped and 43 individuals were identified. Density estimates were similar to values reported by other studies in the Alps with an average of 4.5 and 2.4 individuals/km2 in the first and second study year, respectively, and mean home ranges estimated at 0.95 km2. According to habitat selection models and eDNA-based diet analysis, hazel grouse selected early-succession secondary-growth forests formed after the abandonment of traditional agropastoral activities. These forests, mostly composed of hazel Corylus avellana, Norway spruce Picea abies and Sorbus spp., provided winter food resources and shelter. The diet analysis also highlighted forest arthropods as a non-negligible source of food. Birds avoided areas subject to intensive browsing by ungulates; small forest roads seasonally closed to traffic had positive influence on hazel grouse (i.e. higher abundance of droppings), while roads open to traffic had no effect. Importantly, despite the high coverage of mature forest habitats of Community Interest (53% of our study area), droppings were more abundant in non-listed early-succession secondary forests with similar plant composition. Our results suggest that forest succession after agropastoral abandonment may be beneficial for some forest birds of conservation interest, while acknowledging its negative effects on the previous grassland biodiversity.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Forests are key to biodiversity conservation and provide essential ecosystem service to mankind (FAO and UNEP 2020). Forest habitats cover 50% of the EU’s Natura 2000 sites (‘N2000’; Sotirov et al. 2017), the largest coordinated network of protected areas in the world, and based on two cornerstones, the Habitats (92/43/EEC) and Birds Directives (2009/147/EU). As forests undergo successional changes following natural processes or anthropogenic disturbance events, faunal assemblages also change the species which inhabit newly disturbed sites are progressively replaced by those favouring older forests (Hilmers et al. 2018; Novoa et al. 2021). Thus, a general approach to biodiversity conservation of forest habitats should not be static and needs to ensure the preservation of all successional stages (Kovac et al. 2018).
Compared to old-growth forests, which are pivotal for conservation but make up only 2.4% of EU forests (Barredo et al. 2021), less attention has been accorded to the ecological value and protection of early-successional stands for biodiversity, notwithstanding recent calls raised by the scientific community to address more research initiatives on this topic (Swanson et al. 2011; Lindenmayer et al. 2019). Despite potentially harbouring species of conservation interest, the value of early-succession stands is usually neglected. Such habitats are frequently regarded as the results of degradation of open habitats (e.g. abandonment of agropastoral activities; Laiolo et al. 2004) when secondary succession leads to shrub and tree encroachment, with the consequent disappearance of open-habitat species (Osińska-Skotak et al. 2019). Understanding the ecological requirements of indicator, umbrella and/or keystone forest species that rely on early-successional stands, generally neglected by many conservation strategies is a key process to improve current forest conservation policies and related management activities (Versluijs et al. 2019).
Habitat protection is a key conservation action for hazel grouse Tetrastes bonasia, a Paleartic forest-specialist bird and indicator species (Kajtoch et al. 2012; Pakkala et al. 2014; Magg et al. 2019) adapted to early-succession forest, but also to multi-layered and old-growth stands with small forest gaps (de Juana and Kirwan 2013). With its cryptic plumage and shy behaviour, it remains one of the least studied tetraonids in Europe despite being a priority species of conservation interest listed in both Annex I and II of the Birds Directive (2009/147/EC). During the twentieth century the distribution of hazel grouse in Western and Central Europe was drastically reduced, and many local populations declined heavily in numbers or even vanished, particularly in the lowlands (Burfield and van Bommel 2004, Klaus and Ludwig 2021). Several factors are believed to have contributed to this decline, including predation, increasingly wet climatic conditions in the breeding season, disturbance caused by human activities and widespread habitat loss owing to forest management changes (de Juana and Kirwan 2013). The latter seems to play a decisive role by converting suitable structurally diverse stands to single-layered forest (Storch 2000). In the European Alps, hazel grouse is a threatened species with several populations being on the brink of extinction, yet this region still represents a stronghold for the species with some populations thriving in richly structured coniferous and mixed forest stands (Knaus et al. 2018). Alpine populations are considered particularly vulnerable due to isolation, exacerbated by the short dispersal distance of the species, which also leads to low recolonization rate (Åberg et al. 1995; Montadert and Léonard 2011). This decline in hazel grouse populations makes it important to increase knowledge about the species’ ecological requirements (IUCN 2020).
Capture-Mark-Recapture (CMR) techniques have been widely used to study demographic parameters of wild populations, such as population size and density. As the traditional approach relies on multiple physical captures of individual animals, the study of elusive, rare or low-density species has often been limited by lack of data (e.g. Lynam et al. 2009; Gervasi et al. 2010). Alternative detection methods have been proposed to improve the sampling outcomes, such as camera trapping (Karanth and Nichols 1998) and non-invasive genetic CMR tecniques (Miller et al. 2005), both with the advantage of reducing the stress caused to animals. In the latter case, physical captures are replaced by genetic tagging of biological samples (Taberlet and Luikart 1999), such as hair, feathers or faeces collected in single or multiple occasions (Lukacs and Burnham 2005). With the advent of environmental DNA (eDNA) metabarcoding (Taberlet et al. 2018) the collection of faecal samples now offered the opportunity of studying the diet of elusive species. Indeed, DNA metabarcoding, i.e. the genetic identification of multiple taxa using a standardized DNA region, is now increasingly used in ecological studies including diet analysis (De Barba et al. 2014; Shehzad et al. 2012; Soininen et al. 2009). Various techniques have been used to assess density, diet, foraging and roosting habitat requirements of tetraonids (e.g. Sachot et al. 2003; Mathys et al. 2006; Kortmann et al. 2018), but, to our knowledge, no study has yet been undertaken on hazel grouse implementing genetic CMR.
In this work we evaluated the role of forest stands of different successional stages on a conservation interest forest specialist species in the Alps during winter. We focused on a N2000 study area, half of which consists of mature forest habitats of Community Interest (HCI), while the other half includes early-succession secondary forests not considered as habitats of Community Interest (non-HCI) due to their recent formation (10–60 years old) following the abandonment of traditional agropastoral activities. This provides an optimal context to evaluate the role of a range of forests structures, including those subject to different levels of consideration within conservation policies. Combining individual identification based on non-invasive genetic sampling with metabarcoding-based diet analysis, we investigated the home range size, densities, correlates of microhabitat selection and diet composition of an Alpine hazel grouse population. We chose to work during the winter as this season is likely to be critical for the survival of the species (i.e. food availability, suitable roosting habitat), while snow cover is also likely to increase the detectability of droppings, and better preserve any DNA present. We hypothesised that in winter birds would select dense early-succession mixed forest stands as ideal sites for sheltering from predators and for roosting. A proportion of the habitat would also include plants known as important food resources during the winter such as Sorbus spp. and hazel Corylus avellana. Finally, being a species of conservation interest and indicator of close-to-natural forests, we expected hazel grouse to favour the more mature HCI forests.
Material and methods
Study area
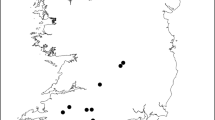
The study site comprises an area of approximately 700 ha in the eastern Italian Alps within the Paneveggio-Pale di San Martino Natural Park (Trentino, Italy; 46°12′17.5″N 11°51′17.1″E; Fig. 1). It is laid out across a moderate elevation gradient (1100–1670 m asl) encompassing a range of vegetation belts typical of the European Alps, from lower montane (comprising the genera Corylus, Fagus and other broadleaves species) to upper subalpine forests (and the genera Fagus, Abies, Picea, Larix). The entire study area (except a very small portion) is included in the N2000 network both as a Special Area of Conservation (SAC; based on the Habitats Directive) and a Special Protection Area (SPA; based on the Birds Directive). Within the site 375 ha (53% of the study area) are classified as HCI of which 228 ha (68% of HCI) are made up of Illyrian beech Fagus sylvatica forest (Aremonio-Fagion; N2000 code: 91K0), 16% by acidophilus Norway spruce Picea abies (N2000: code 9410) forest, and 16% hay meadows (N2000 code: 6520, 6210, 6230). The other half of the study site (ca 325 ha; 47%) is non-HCI and mostly dominated by Norway spruce and European larch Larix decidua in the form of secondary-growth forest (ca 80% or 230 ha; trunks being of 10–60 years old) following agropastoral abandonment. Overall, these forests constitute optimal habitat for hazel grouse and records of the species on the study site stretch back at least to the 1960s (Calovi and Mattedi 1995; Cattadori and Hudson 1999). This hazel grouse population cannot be considered isolated as nearby areas and regions hold populations of this species (Sitzia et al. 2014; Galluzzi et al. 2022).
On top is shown the location of the study area in Italy (Paneveggio-Pale di San Martino Natural Park, Trentino). Map shows the study areas (red line) and the relative N2000 boundary (green line). Black dots represent hazel grouse locations (i.e., faecal samples collected) while white dots represent randomly generated locations used in the habitat selection analysis collected over winter 2017-2018. Red areas represent forests of Community Interest (Illyrian beech forest - N2000 code: 91K0; Norway spruce forests - N2000 code: 9410) while those in blue represent forests of non-Community Interest of recent formation (10-60 years) following the abandonment of traditional agropastoral activities. Areas in yellow represent N2000 hay meadows (code: 6520, 6210, 6230)
Bird survey
We used hazel grouse faeces to assess microhabitat selection, diet, home range and density in the study area during winter across 2 years (2015–2016 and 2017–2018). Samples were collected along pre-identified transects spaced 100 m apart across the entire study area and were repeated five times in each session (see Figure S1, Table S1). To maximize repeatability and reduce observer bias, transects were surveyed by a limited number of people (2 people in year 2015–2016 and 4 people in year 2017–2018). Each surveyor was equipped with a GPS device and a compass, and was asked to follow pre-positioned visual clues (i.e. small flags hanged on tree branches) set along forest transects that helped surveyors walking straight along transects. Potential hazel grouse faeces were collected using sterilized tweezers and stored in two 15 ml test tubes for genetic analyses.
Genetic analyses
All laboratory work was carried out at the Fondazione Edmund Mach—Research and Innovation Centre in a dedicated environmental DNA facility, physically separated from all other molecular biology laboratory facilities and with separate rooms for DNA extraction, PCR preparation and post-amplification steps. Negative controls were added to each DNA extraction and PCR amplification batch to monitor contaminations.
DNA extraction was performed on 100 mg of faecal sample using the Mag-Bind® Stool DNA 96 Kit, and DNA was eluted in 150 µL of elution buffer.
Species confirmation, sexing and individual identification
Species confirmation was carried out by amplifying the mitochondrial control region ("CR"; Cann et al. 1987; Wan et al. 2004) using the primer pair PHDL-PH-H521 (Fumihito et al. 1995; Randi and Lucchini 1998), while for sex identification the nuclear CHD gene (Ellegren 1996) was amplified using the primer pair 1237L-1272H (Kahn et al. 1998). Amplified products were purified using the Exo-SAP kit (USB corporation—USA) and sequenced with the ABI 3130XL DNA sequencer (Applied Biosystem ™). Sequencing products were analyzed with Sequencher v.4.7 (Gene Codes Corporation, Ann Arbor, MI, USA), and haplotypes were identified with Fabox v.1.5 (Villesen 2007) by comparing them with 56 public sequences downloaded from GenBank.
For the individual identification, we selected a set of 11 microsatellite markers previously developed and tested for other tetraonids (Table S11), as no species-specific markers were available for Tetrastes bonasia. Genotyping was carried out on an AB3730XL Genetic Analyzer (Applied Biosystems), and sequencing products were analyzed using GeneMapper v.5.0 (Applied Biosystem™). GenAlEx 6.5 (Peakall and Smouse 2012) was used to calculate the probability of identity (PID and PIDsibs); standard genetic diversity indexes were inferred using r package ‘GenePop’ v.1.1.3 (Rousset 2008). Detailed descriptions of PCR amplification conditions and microsatellite genotyping are provided in Table S10 and Appendix S2.
DNA metabarcoding for diet analysis
Overall, 72 different faecal samples (39 females and 33 males) were analyzed by means of metabarcoding for the characterization of the hazel grouse diet. At least one sample was included in the analysis for the 43 identified individuals; additional samples were processed to ensure adequate representativeness of both sexes and different years (according to sample availability; Table S3).
To identify the plant component of the diet, a short fragment (about 150 bp) of the chloroplast gene trnL was amplified using the universal primers c-A49325 and h-B49466 (Taberlet et al. 2007), while for the characterization of the arthropod component, a short fragment (157 bp) of the cytochrome oxidase 1 (CO1) was amplified using the universal primers ZBJ-ArtF1c (Zeale et al. 2011); each sample was amplified in two independent replicates to account for stochastic PCR variability. The deriving amplicons were sequenced using MiSeq Reagent Kit v3 in an Illumina MiSeq platform.
Taxonomic assignment was performed by comparison with: (a) a custom-made reference database including trnL DNA sequences for most of the Alpine seed plants for trnL (Appendix S2); (b) the Midori-UNIQUE reference database (Leray et al. 2018) for CO1 (arthropods). Details on bioinformatics analysis are reported in Appendix S3.
For each PCR replicate, we computed the relative read abundance (RRA), defined as the proportion of unique Illumina sequence reads in a sample divided by the final (i.e. after quality control) number of sequence reads in that sample. Lastly, we averaged the RRA values of the different PCR replicates of the same sample to obtain the mean RRA.
Environmental correlates
To characterize the winter microhabitat preferences of hazel grouse, we collected detailed microhabitat data from late December to early March 2017–2018 at occurrence points (location of hazel grouse faeces) and for an equal number of random locations (pseudo-absences) representative of the local environmental conditions (see Sect. 2.5.1 for more details) which were collected in order to create a perfectly balanced, matched design of ‘cases’ (droppings) and ‘controls’ (random location) for conditional logistic regression modelling, a technique commonly used in other studies (e.g. Brambilla et al. 2018; Alessandrini et al. 2022). At each site (i.e. random and occurrence location) we collected detailed data on vegetation cover and composition within a 10 m radius buffer, equivalent to an area of approximately 314 m2 (see Table S2 for further details). Trunks were characterized at each location by counting the number of trees species according to 5 diameter breast height (DBH) classes (< 10 cm, 10–20 cm, 20–30 cm ∩ , 30–40 cm, 40–50 cm, > 50 cm) which were measured with a tape by fieldworkers. Ground cover characterization in the 10 m radius plots was assessed by visually estimating the percentage cover of the various variables. Topographic variables (i.e. altitude, slope and aspect) were calculated and averaged within the buffer through the r.slope.aspect algorithm from GRASS vers. 7.04 (Neteler et al. 2012) using a DTM derived from a local high-resolution LIDAR (PAT 2009) upscaled at a 10 × 10 m resolution. We also collected information on ungulate browsing intensity (i.e. red deer Cervus elaphus, roe deer Capreolus capreolus, chamois Rupicapra rupicapra) classifying it into four categories: i) none (0% of vegetation browsing/fraying/stripping detectable), little (1–29%), medium (30–49%), high (≥ 50%). The impacts of forest fragmentation and human disturbance on hazel grouse are well-documented in the literature (Kajtoch et al. 2012, Matysek et al. 2020), so for each occurrence and random location we also calculated, via QGIS, the relative distance to three types of roads subject differing degrees of traffic: (i) hiking trails: walk-only paths; (ii) roads closed to traffic: forestry roads with regulated motorized access, usually closed in winter due to adverse weather conditions; (iii) roads open to traffic: describing roads open to traffic all-year round, without regulation.
Statistical analysis
Sample size differed for each analysis according to aims, technical feasibility (e.g. the trade-off between laboratory costs and sample representativeness for the diet analysis) and other analytical issues (e.g. the availability of corresponding microhabitat data); a detailed description of sample sizes is provided in Table S3.
Winter microhabitat and N2000 community interest habitat selection
We tested the null hypothesis that faecal samples of hazel grouse were distributed randomly with respect to environmental and topographical covariates. A binary response variable was created using data for 117 samples and an equal number of random locations. Our dataset was spatially and temporally balanced as for each visit we gathered environmental data for an equal number of occurrences and random locations (Table S1). In detail, one to three random locations was created a priori for each transect using the RandomPoints tool in QGIS (Quantum GIS Development Team 2015). At the random location the surveyors collected environmental data equivalent to that gathered when finding a hazel grouse faecal sample. Once the number of faecal samples available for analysis was known, we selected random points based on location (preferring random points at closer location to faecal samples for stronger comparisons) and on the survey date (to match similar environmental conditions).
To model hazel grouse microhabitat selection, we built models comparing environmental characteristics between occurrence sites and random sites, based on a balanced design. We evaluated occurrence probability (the product between species encounter and occurrence probability) as a function of environmental variables by means of a conditional logistic regression approach (clogit function) in the ‘survival’ R package (Therneau 2021). We compared characteristics of an equal number of sites with real droppings and random locations, belonging to the same ‘stratum’, i.e. the combination of ‘individual x survey date/visit’. Such approach allowed us to control for temporal differences in environmental conditions across each visit, as well as for non-independent data related to the same individual.
We assumed faecal detection probability to be constant across different covariate ranges, and therefore that faecal detection/non-detection represented true microhabitat selection. Model evaluation followed an information-theoretic approach, based on the Akaike’s information criterion for small sample size (AICc) (Burnham and Anderson 2002, 2004). All the covariates were scaled and expressed as both linear and quadratic terms and tested before fitting each model for potential within-group collinearity (VIFs > 3, Kutner et al. 2005) by calculating the variance inflation factor (VIF; package car in R). Due to the large number of explanatory variables and to reduce the chances of model overfitting, we divided the analyses into various steps. Firstly, we modelled the probability of hazel grouse occurrences as a function of tree species’ abundance according to DBH in respect of the null model. To further simplify the model, if trees of the same species, but with different DBHs (e.g. beech with DBH < 10 cm, 10–20 cm, 20–30 cm) showed similar selection patterns (e.g. negatively correlated with the probability of hazel grouse occurrence) we created a new variable (e.g. beech < 30 cm) combining the abundance of all the DBH of the same and refitted the model. The same model structure was then repeated for all ground cover and topographic variables. All supported variables (ΔAICc < 2) were then combined into a global model testing all possible variable combinations using the dredge function in the MuMIn package (Bartoń 2019) and ranked them according to AICc. Averaged parameters of the supporting models with ΔAICc < 2 (n = 4) were calculated following the Johnson and Omland (2004) procedure. Adjusted R2 was assessed using the package ‘rsq’ (Zhang 2021). We tested the potential effects of spatial and temporal autocorrelation in the residuals of the highest ranked models using the package ‘DHARMa’ and ‘gstats’ (Hartig 2020). No significant linear correlation was detected between distance and semivariance for the most supported model. To assess potential sexual differences in microhabitat selection we compared mean values for the most supported variables across sex using unpaired two-samples t test. Finally, we used a two-tailed Chi-squared test to compare frequencies of dropping collected in both surveys (winter 2015–2016 and 2017–2018; n = 192) with respect to an equally random set of newly generated forest points processed in GIS.
Hazel grouse density estimation
We used spatial capture-recapture (SCR) models (Efford and Fewster 2013; Royle et al. 2014) to account for animal movement and imperfect detectability in density estimation. Specifically, we used a model formulation that accounted for area-search sampling, where a sample area (i.e. the area sampled around all transects, Figure S1) is searched thoroughly and all detected animals are uniquely identified (Royle et. al. 2008; Efford 2011). In our case, study area was sampled on T = 5 occasions in two consecutive seasons (2015–2016 and 2017–2018) and yielded capture histories on unique individuals with a spatial location for each detection. We used a half-normal encounter model where detectability p is a function of the baseline encounter probability (p0) and the spatial scale parameter σ, which determines how encounter probability decreases with an increase in the distance between detection location and the individual activity centre. We fitted a stratified population model (Royle et al. 2014) to data grouped by year and derived year-specific population densities using the R package ‘secr’ v. 3.2.0 (Efford 2014). No spatially explicit covariates were available for the entire state space, and therefore, we did not model spatial variation of density. We investigated the effect of sex, year and time (i.e. seasonality) on species detectability and the scale parameter σ by contrasting different models based on the plausible combination (both additive and interaction) of different covariates (Table S7). We expected encounter rate to change between the two sampling years, between sexes and during the sampling season. We anticipated that males and females hazel grouse would move differently, thus having different home range sizes. For further details on model specification and calculation of density and home range size, see Appendix S1.
Results
Genetic analyses: individual identification and sexing
In total, 216 of the collected droppings were confirmed as Tetrastes bonasia faecal samples by mtDNA analysis and were successfully genotyped at the selected 11 microsatellite markers, resulting in the overall identification of 43 individuals (i.e. different genotypes) in the study area. Thirty-four individuals were sampled in 2015–2016 and 24 in 2017–2018; 15 individuals were sampled in both sampling years. Among the 43 individuals, we were able to identify 22 females and 21 males (17 females and 17 males in 2015–2016 and 13 females and 11 males in 2017–2018).
Despite generally low variability levels (mean number of alleles: Na = 5.5; expected heterozygosity: He = 0.59), our marker panel allowed for a clear distinction between different individuals. Probability of identity (PID and PIDsibs) varied from 6.0 × 10–1 and 7.8 × 10–1 at locus ADL184 to 4.2 × 10–2 and 3.4 × 10–1 at locus BG16; the cumulative PID (2.3 × 10–8) and PIDsibs (6.0 × 10–4) values were sufficiently small to exclude different individuals in the population having the same multilocus genotype (probability of correct assignment is between 99.94 and 100%).
Hazel grouse density and home range
Overall, in the two sampling years 76 and 117 detections were achieved, referring to 34 and 24 individuals, respectively (see Table S9 for individual capture histories). Based on AICc, the most parsimonious model on which we based our inference included an effect of year on the baseline encounter probability (p0) and an equal scale parameter σ across sexes (model M11 in Table S7). Baseline encounter probability varied from 0.003 (0.002–0.006, 85%CI) in the first year to 0.008 (0.006–0.011) in the second year (Table S8). An average estimate of 95% home range size, derived from the spatial scale parameter of the half-normal encounter model, was 0.955 km2. Predicted density was 4.5 (3.1–6.7) individuals per km2 in the first year decreasing to 2.4 (1.6–3.6) individuals per km2 in the second year (Table S8).
DNA metabarcoding for diet analysis
The DNA metabarcoding (trnL gene) for the characterization of the plant component of the diet on 72 individual samples, resulted in the identification of 54 plant taxa. Only 10/54 plant taxa were detected with an overall RRA (relative read abundance) value higher than 0.01 (see Fig. 2a). Betulaceae—subfamily Coryloideae was the most common and abundant recorded plant taxon, being found in 61/72 samples for an overall RRA of 0.64. The other nine most abundant plant taxa, all found with an overall RRA < 0.1, were (each one reported at the lowest reliable taxonomic level): Rosaceae—tribe Maleae (found in 42/72 samples); Pinaceae—Picea spp. (22/72); Ericaceae—Vaccinium spp. (16/72); Pinaceae—Larix decidua (14/72); Rosaceae—tribes Potentilleae/Rosoideae (13/72); Oxalidaceae—Oxalis spp. (9/72); Fagaceae—Fagus sylvatica (9/72); Rosaceae—tribe Sanguisorbeae (6/72); and Salicaceae—Salix spp. (6/72). The complete list of plant taxa recorded per sample is reported in Tables S11-S12.
Hazel grouse diet composition in winter (2015–2016 and 2017–2018) based on DNA metabarcoding: a plant component (trnL barcode region); b arthropod component (CO1 barcode region). The most frequent taxa are reported, based on their RRA (relative read abundance; expressed here as percentage) in the whole dataset (plant component) and in the faecal samples containing at least one arthropod taxon (arthropod component; 35/72, 48.61%)
The DNA metabarcoding (CO1 gene) for the identification of the arthropod component of the diet resulted in the identification of 44 taxa, but only 19 of them were recorded with a total RRA > 0.01. Interestingly, at least one taxon was found in as many as 35/72 (48.61%) of the analyzed faecal samples. The most abundant insect orders resulted to be Lepidoptera (total RRA = 0.39) and Diptera (total RRA = 0.31). Among the Lepidoptera, the Tortricidae was the most frequent family, being found in 11/72 (15.28%; overall RRA = 0.20) samples with four different genera: Epinotia (in 6/72 samples), Celypha (4/72), Zeiraphera, and Crocidosema (1/72). Among the Diptera, Cecidomyiidae were found in 6/72 samples (8.33%; overall RRA = 0.09); Arachnida (family Araneae) were found in 5/72 samples (6.94%; overall RRA = 0.11). A synthetic representation of the overall arthropod component is reported in Fig. 2b; the complete list of recorded arthropod taxa per sample is reported in Tables S13-S14.
Winter microhabitat selection
We found a most supported model describing the probability of occurrence of hazel grouse in our study area in relation to various environmental variables (see Table 1 and Table S4 for top candidate models) collected in winter. The most supported model explained a good proportion of the variation (marginal R2 = 0.56) and included both topographic and vegetation variables. Highest probability of occurrence peaked around 1400 m asl and on gentle slopes. In terms of vegetation, in winter hazel grouse favoured high densities of hazel (1–50 ca stems with DBH < 10 cm), Norway spruce (up to 50 stems with DBH < 30 cm) and the presence of Sorbus spp. (Sorbus aria and S. acuparia). In contrast, the probability of hazel grouse occurrence in winter was negatively correlated with beech, grass cover, snow depth and areas associated with high evidences of browsing/fraying/stripping by ungulates (Fig. 4). There was a higher probability of encountering an evidence for hazel grouse along roads seasonally closed to traffic (up to 160 m). At a univariate level (Table S4) roads open to traffic had a neutral effect on hazel grouse occurrence, while there was a negative linear relationship between the probability of hazel grouse occurrence and distance from hiking trails. No major sex differences in winter microhabitat selection were detected apart from 2017 to 2018 when females were recorded at slightly higher elevations (μ females = 1,398 m a.s.l, SE = 13.38; μ males = 1,328 m a.s.l. SE = 12.97, p < 0.001) and at further distances from roads closed to traffic than males (females μ = 199.7 m SE = 17.14; males μ = 138.44 SE = 14.76, p = 0.01; Table S5).
Across both winter surveys (2015–2016 and 2017–2018), hazel grouse faeces (n = 192) were more frequent in non-HCI than HCI, when compared to an equal number of random locations (χ2 = 64.28, df = 1, p < 0.001; Fig. 3). By analysing data of random points alone, we found important differences in abundance of trees and scrub between HCI and non-HCI sites based on DBH size (Table S6). Non-HCI had a generally higher abundance of trees with smaller size DBH, particularly Norway spruce and hazel, while deciduous trees (i.e. beech) were less common compared to HCI.
Frequency comparison between hazel grouse droppings collected in winter 2015–2016 and 2017–2018 (n = 192) and an equal number of randomly created locations in respect of Natura 2000 Community Interest forest habitats and non-Community Interest forest habitat. Hazel grouse droppings were more abundant in non-Community Interest forest (χ2 = 64.28 df = 1, p < 0.001) of recent formation (10–60 years old) following the abandonment of traditional agropastoral activities
Discussion
To our knowledge, this is one of the few works that demonstrates how early-successional secondary forests, generally neglected by conservation policies, could be critical for the conservation of a priority species during winter months. In our case study, despite the high availability of mature forest, largely included in HCI within the N2000 site, hazel grouse selected younger secondary forest stands (non-HCI) with a similar arboreal composition but in early-succession phase, which have developed following the abandonment of traditional agropastoral activities. Winter microhabitat selection and diet analysis suggested that these forests provided fundamental food resources and shelter during winter. Future works should evaluate the potential role of other early-successional forest stages for the conservation of threatened species.
Population density, home range and sex ratio
Winter densities of hazel grouse have been little investigated in the literature as most estimates have been produced during spring and autumn when birds are more responsive to play-back/whistles of the territorial song (e.g. Swenson 1991). In our study area winter densities were similar to other studies in the European Alps but obtained during spring or autumn (von Blotzheim and Solari 1985, Montadert and Leonard 2003, De Franceschi 1995). Such densities remain much lower if compared to populations present in Central and Northern Europe where 10–11 birds/km2 and 4.6–37 birds/km2 have been, respectively, estimated (de Juana and Kirwan 2020). We found a balanced sex ratio across our study which differs with findings in France (Montadert and Leonard 2006b) and in Sweden (Swenson 1993) where a male-biased population was detected. We believe that such results may be non-representative of the true ratio as these are likely influenced by male-biased bird survey techniques (i.e. playback calls of territorial males) or based on shooting bag data.
Winter microhabitat selection and diet
Results from models evaluating environmental correlates of winter microhabitat selection in hazel grouse suggested a preference for the following species: Norway spruce, hazel and Sorbus spp., all with relatively small DBH (ca < 30 cm; see Fig. 4). The selections for dense early-succession coniferous stands are likely representative of roosting/shelter microhabitats where hazel grouse can hide from predators or from adverse weather conditions such as low temperatures and snow storms which are common events in the study area. Such findings are in line with other studies located in forests where both coniferous and deciduous trees are found such as in the European Alps or in the Bohemian Forest (Sachot et al. 2003; Schäublin and Bollmann 2011, Ludwig et al. 2017). Swenson and Olsson (1991) found that in Norway, despite the presence of deciduous trees, Norway spruce was the most selected tree for night roosts being the tree species providing maximum height cover in dense stands. These findings were strongly corroborated by the results of the DNA metabarcoding-based diet analysis which highlighted hazel, Sorbus spp., as well as, Norway spruce and European larch, as some of the most frequently recorded taxa. A further confirmation came from the arthropod component detected in the diet. Commonly recorded Lepidoptera species such as Epinotia tedella and E. tenerana feed primarily on Norway spruce and hazel, respectively, while the larvae of the frequently recorded family Diptera: Cecidomyiidae are renown galls midgets. Further studies should evaluate whether hazel grouse selects galls containing these insects on purpose or accidentally ingests shoots. However, these are reported to be an important winter food resource for certain birds and mammals (Turcek 1951). Our findings, with 48.61% of the analysed faecal samples containing arthropod DNA, seem to confirm this pattern for the hazel grouse. Diet analyses highlighted that the species also foraged on ground flora such as on Vaccinium spp. and other herbaceous plants within the wood sorrel family (most likely Oxalis acetosella). Although we cannot rule out potential issues with detectability which could not be accounted for in the microhabitat selection analysis due to low number of recaptures, the negative relationship found between snow depth and hazel grouse occurrence probability might be representative of sites rendered inaccessible (i.e. deep snow) for foraging on such plants.
The presence of ungulates, particularly where browsing activity was high, was negatively associated with the occurrence of hazel grouse. Red deer, which occurs here at high density (Apollonio et al. 2019), is known to reduce tree species richness, with cascading effects on many parts of the ecosystem. This is particularly true for the observed declines in rowan (D’Aprile et al. 2020) and the growth of the bilberry (Motta et al. 2014), which are important components in hazel grouse diet. Similar negative effects of herbivore browsing impacts were also demonstrated for capercaillie Tetrao urogallus (Partel 2018) within the park.
Although hazel grouse is considered a species highly susceptive to habitat fragmentation and human activity, we found a negative relationship between the probability of occurrence of hazel grouse and the distance from the nearest hiking trails (univariate effect) and roads closed to traffic during the winter. These results contrast with other studies on hazel grouse and other tetraonids (e.g. Matysek et. al 2020) and are probably linked to the fact that, in our study area, non-intensive management of trails and forest roads harbours suitable ecotones with edible shrubs and trees, while we argue that other studies considered roads and trails generally more frequented by people.
The value of young and traditionally neglected forest habitats
Despite mature forests being available in our study area, where they are mostly classified as HCI, hazel grouse were more frequently recorded in younger (non-HCI) forests. These two were similar in tree species’ composition, but differed in DBH (Table S6). Indeed, in our study area, younger forests are of secondary formation, being former hay meadows and larch wood pasture which, following agropastoral abandonment after post-World War 2, have now evolved into early-succession secondary growth Norway spruce and European larch stands. The fact that non-HCI forests were more important than mature HCI ones for hazel grouse, confirms that N2000 sites, if correctly delimited and managed, may truly act as parts of a broader ecological network, which goes beyond the preservation of a relatively few classified and listed habitats. This also calls for more investigation and proper management efforts of habitats of apparently lower value. Within our study area, which is both a SAC and a SPA, according to N2000 management targets, early stands should be managed with the perspective of achieving stand maturation. This process is likely to be detrimental for hazel grouse as suitable early-succession stands will be replaced by unsuitable mature ones. Instead, an ideal management perspective should aim for the preservation of both suitable early-succession and older stands for hazel grouse. Early-succession forests are not the only habitat subject to this controversy. A similar argument may be raised for other habitats, such as e.g. reed beds (e.g. Phragmites australis), which are non-HCI, but are a key feeding and breeding habitats for numerous wetland species, including several birds of conservation importance (e.g. Morganti et al. 2019). We therefore recommend careful evaluation and planning also for non-HCI habitat as well, in the light of their potential value for animal species of conservation interest.
Hazel grouse conservation and management
Although limited to the winter period, this study provides some novel information on the biology and ecology of a poorly studied species, highlighting important potential conservation management trends. We suggest the creation and maintenance of a mosaic of microhabitats characterized by uneven aged forest composed of both coniferous and deciduous trees. Small areas with early-succession stands should be achieved by enhancing old-growth areas characterized by dead horizonal logs and vertical snags which favour the formation of rejuvenation areas of deciduous trees, whilst forming a natural shelter protecting young trees from browsing ungulates. It is important to maintain the number of ungulates at a density compatible with the regeneration of the forest, with particular reference to plant that act as fundamental food resources such as Sorbus spp., Vaccinium spp., wood sorrel and members of the Rosaceae family. Forest–grassland ecotones may favour the formation of edible deciduous trees, but careful planning should be made as hazel grouse is a poor disperser and avoids open areas (Montadert and Leonard 2003, 2006a; Kajtoch et al. 2012). The presence of hiking trails and roads should also be carefully planned in light of the potential impacts associated with high human frequentation (e.g. Formenti et al. 2015) and forest fragmentation effects (Matysek et. al 2020). Hazel grouse is considered a forest indicator species; hence, the application of such management directions is likely to be favoured by other forest species of conservation interest. Other tetraonids and songbirds might also be attracted by hazel grouse foraging sites particularly where high abundance of autumn and winter berries occurs (Fox et al. 2009; Bocca et al. 2014; Thiel et al. 2007). Finally, these habitats are unlikely to be suitable only during the winter period in that early-succession stands have also been shown to be important during spring and summer providing direct food resources, hunting areas and coverage for various songbirds, grouse species (including hazel grouse) and raptors (Cody et al. 1985, Kortmann et al. 2018, Ratajac et al. 2022).
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Code availability
The code generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Åberg J, Jansson G, Swenson JE, Angelstam P (1995) The effect of matrix on the occurrence of hazel grouse (Bonasa bonasia) in isolated habitat fragments. Oecologia 103:265–269
Alessandrini C, Scridel D, Boitani L, Pedrini P, Brambilla M (2022) Remotely sensed variables explain microhabitat selection and reveal buffering behaviours against warming in a climate-sensitive bird species. Remote Sens Ecol Conaserv. https://doi.org/10.1002/rse2.265
Apollonio M, Chirichella R, De Marinis AM et al (2019) Camoscio, cervo e capriolo in Trentino Rapporto su status e gestione. Quad Assoc Cacciatori Trent 1:248
Barredo JI, Brailescu C, Teller A et al (2021) Mapping and assessment of primary and old-growth forests in Europe. Publications Office of the European Union, Luxembourg
Bartoń K (2019) MuMIn: multi-model inference. R Package Version 1.43.6. https:// CRAN.R- proje ct. org/ ack age= MuMIn.
Bocca M, Caprio E, Chamberlain D, Rolando A (2014) The winter roosting and diet of Black Grouse Tetrao tetrix in the north-western Italian Alps. J Ornithol 155:183–194
Brambilla M, Resano-Mayor J, Scridel D, Anderle M, Bogliani G, Braunisch V et al (2018) Past and future impact of climate change on foraging habitat suitability in a high-alpine bird species: management options to buffer against global warming effects. Biol Cons 221:209–218
Burfield I, van Bommel F (eds) (2004) Birds in Europe: population estimates trends and conservation status
Burnham KP, Anderson DR (2002) Model Selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, Newyork
Burnham KP, Anderson DR (2004) Multimodel Inference. Sociol Methods Res 33:261–304
Calovi F, Mattei S (1995) Piano faunistico del Parco Naturale di Paneveggio - Pale di San Martino. pp 117–128
Cann RL, Stoneking M, Wilson AC (1987) Mitochondrial DNA and human evolution. Nature 325:31–36
Cattadori IM, Hudson PJ (1999) Temporal dynamics of grouse populations at the southern edge of their distribution. Ecography 22:374–383
Cody ML (1985) Habitat selection in the sylviine warblers of western Europe and North Africa. In: Cody ML (ed) Habitat selection of birds. Academic Press, Newyork, pp 85–129
D’Aprile D, Vacchiano G, Meloni F et al (2020) Effects of twenty years of ungulate browsing on forest regeneration at Paneveggio Reserve, Italy. Forests 11–612:1–12
De Barba M, Miquel C, Boyer F et al (2014) DNA metabarcoding multiplexing and validation of data accuracy for diet assessment: application to omnivorous diet. Mol Ecol Resour 14:306–323
De Franceschi P, Bottazzo M (1995). Habitat characteristics of brood-rearing sites of Hazel Grouse Bonasa bonasia in the Eastern Alps (Friuli-Venezia Giulia, Italy). In: JENKINS (Ed), Proc. Intern. GrouseSymp, 101–105
de Juana E, Kirwan GM (2013) Hazel grouse (Bonasa bonasia). In: del Hoyo J, Elliott A, Sargatal J, Christie DA, de Juana E (eds) Handbook of the birds of the world alive. Lynx Edicions, Barcelona
de Juana E, Kirwan GM (2020) Hazel grouse (Tetrastes bonasia), version 1.0. In: del Hoyo J, Elliott A, Sargatal J, Christie DA, de Juana E (eds) Birds of the world. Cornell Lab of Ornithology, Ithaca, NY. https://doi.org/10.2173/bow.hazgro1.01
Efford MG (2011) Estimation of population density by spatially explicit capture-recapture analysis of data from area searches. Ecology 92:2202–2207
Efford MG, Fewster RM (2013) Estimating population size by spatially explicit capture-recapture. Oikos 122:918–928
Efford, M.G. (2014) Secr: Spatially Explicit Capture-Recapture Models. http://cran.r-project.org/package=secr.
Ellegren H (1996) First gene on the avian W Chromosome (CHD) provides a tag for universal sexing of non-ratite birds. Proc R Soc B 263:1635–1641
FAO and UNEP (2020) The State of the World’s Forests 2020. Forests biodiversity and people, Rome
Formenti N, Viganò R, Bionda R (2015) Increased hormonal stress reactions induced in an Alpine Black Grouse (Tetrao tetrix) population by winter sports. J Ornithol 156:317–321
Fox AD, Kobro S, Lehikoinen A, Lyngs P, Vaisanen R (2009) Northern bullfinch Pyrrhula p pyrrhula irruptive behavior linked to rowanberry Sorbus aucuparia abundance. Ornis Fenn 86:51–60
Fumihito A, Miyake T, Takada M et al (1995) The genetic link between the Chinese Bamboo Partridge (Bambusicola Thoracica) and the Chicken and Junglefowls of the Genus Gallus. Proc Natl Acad Sci 92:11053–11056
Galluzzi, M., Puletti, N., Armanini, M., Chirichella, R., Mustoni, A. (2022) Mobile Laser Scanner understory characterization: an exploratory study on hazel grouse in Italian Alps. https://doi.org/10.1101/2022.04.26.489487
Gervasi V, Ciucci P, Davoli F et al (2010) Addressing challenges in non invasive capture-recapture based estimates of small populations: a pilot study on the Apennine brown bear. Conserv Genet 11:2299–2310
Hartig, F. (2020) DHARMa: Residual diagnostics for hierarchical (multi-level/mixed) regression models. R package version .320
Hilmers T, Friess N, Bässler C, Heurich M, Brandl R, Pretzsch H, Seidl R, Müller J (2018) Biodiversity along temperate forest succession. J Appl Ecol 55:2756–2766
IUCN (2020) The IUCN Red List of Threatened Species. Version 2020–2. https://www.iucnredlist.org. Downloaded on 09 July 2020.
Johnson JB, Omland KS (2004) Model selection in ecology and evolution. Trends Ecol Evol 19:101–108
Kahn NW, St. John J, Quinn TW (1998) Chromosome-specific intron size differences in the avian CHD gene provide an efficient method for sex identification in birds. Auk 115:1074–1078
Kajtoch Ł, Żmihorski M, Bonczar Z (2012) Hazel Grouse occurrence in fragmented forests: habitat quantity and configuration is more important than quality. Eur J Forest Res 131:1783–1795
Karanth KU, Nichols JD (1998) Estimation of tiger densities in India using photographic captures and recaptures. Ecology 79:2852–2862
Klaus S, Ludwig T (2021) Long-term trends of hazel grouse (Tetrastes bonasia) in the bohemian forest (Šumava), Czech Republic, 1972–2019. Birds 2:127–137
Knaus P, Antoniazza S, Wechsler S et al (2018) Distribution and population trends of birds in Switzerland and Liechtenstein. Swiss breeding bird atlas. Swiss Ornithological Institute, Switzerland
Kortmann M, Heurich M, Latifi H (2018) Forest structure following natural disturbances and early succession provides habitat for two avian flagship species, capercaillie (Tetrao urogallus) and hazel grouse (Tetrastes bonasia). Biol Cons 226:81–91
Kovac M, Hladnik D, Kutnar L (2018) Biodiversity in (the Natura 2000) forest habitats is not static: its conservation calls for an active management approach. J Nat Conserv 43:250–260
Kutner MH, Nachtsheim CJ, Neter J, Li W (2005) Applied linear statistical models, 5th edn. McGraw-Hill Irwin, Newyork
Laiolo P, Dondero F, Ciliento E, Rolando A (2004) Consequences of pastoral abandonment for the structure and diversity of the alpine avifauna. J Appl Ecol 41:294–304
Leray M, Shian-Lei H, I-Jeng, L., Ryuji, J.M. (2018) MIDORI Server: a webserver for taxonomic assignment of unknown metazoan mitochondrial-encoded sequences using a curated database. Bioinformatics 34:3753–3754
Lindenmayer DB, Westgate MJ, Scheele BC et al (2019) Key perspectives on early successional forests subject to stand-replacing disturbances. For Ecol Manage 454:117656
Ludwig T, Klaus S (2017) Habitat selection in the post-breeding period by Hazel Grouse Tetrastes bonasia in the Bohemian Forest. J Ornithol 158:101–112
Lukacs PM, Burnham KP (2005) Review of capture-recapture methods applicable to non-invasive genetic sampling. Mol Ecol 14:3909–3919
Lynam AJ, Rabinowitz A, Myint T et al (2009) Estimating abundance with sparse data: tigers in northern Myanmar. Popul Ecol 51:115–121
Magg N, Ballenthien E, Braunisch V (2019) Faunal surrogates for forest species conservation: a systematic niche-based approach. Ecol Ind 102:65–75
Mathys L, Zimmermann NE, Zbinden N, Suter W (2006) Identifying habitat suitability for hazel grouse Bonasia bonasia at the landscape scale. Wildl Biol 12:357–366
Matysek M, Gwiazda R, Bonczar Z (2020) The importance of habitat diversity and plant species richness for hazel grouse occurrence in the mixed mountain forests of the Western Carpathians. Eur J Forest Res 139:1057–1065. https://doi.org/10.1007/s10342-020-01307-2
Miller CR, Joyce P, Waits LP (2005) A new method for estimating the size of small populations from genetic mark-recapture data. Mol Ecol 14:1991–2005
Montadert M, Leonard P (2003) Survival in an expanding hazel grouse Bonasa bonasia population in the southeastern French Alps. Wildl Biol 9:357–364
Montadert M, Léonard P (2006a) Post-juvenile dispersal of Hazel Grouse Bonasa bonasia in an expanding population of the southeastern French Alps. Ibis 148:1–13
Montadert M, Léonard P (2006b) Skewed sex ratio and differential adult survival in the Hazel Grouse Bonasia bonasa. Acta Zool Sinica 52:655–662
Montadert M, Léonard P (2011) Natal dispersal affects population dynamics of hazel grouse in heterogeneous landscapes. In: Sandercock B, Martin K, Segelbacher G (eds) Ecology, conservation, and management of grouse, berkeley and los angeles. University of California Press, CA
Morganti M, Manica M, Bogliani G et al (2019) Multi-species habitat models highlight the key importance of flooded reedbeds for inland wetland birds: implications for management and conservation. Avian Res 10:15
Motta R., Berretti R., Vacchiano G., Meloni F. (2014) Impatto della Fauna ungulata selvatica sulle foreste. Ungulati e gallo cedrone una convivenza possibile? Relazione dell'attività di ricerca condotta da DISAFA per conto dell'Ente Parco Paneveggio - Pale di San Martino, pagg. 111.
Neteler M, Bowman MH, Landa M, Metz M (2012) GRASS GIS: a multi-purpose open source GIS. Environ Model Softw 31:124–130
Novoa FJ, Altamirano TA, Bonacic C, Martin K, Ibarra JT (2021) Fire regimes shape biodiversity: responses of avian guilds to burned forests in Andean temperate ecosystems of southern Chile. Avian Conser Ecol 16(2):22
Osińska-Skotak K, Jełowicki Ł, Bakuła K et al (2019) Analysis of using dense image matching techniques to study the process of secondary succession in Non-Forest Natura 2000 Habitats. Remote Sens 11:893
Pakkala T, Lindén L, Tiainen J, Tomppo E, Kouki J (2014) Indicators of forest biodiversity: which bird species predict high breeding bird assemblage diversity in boreal forests at multiple spatial scales? Ann Zool Fenn 51:457–476
Partel, P. (2018) Ricerca, conservazione e gestione del gallo cedrone nel Parco. Quaderni del Parco n. 14. Ente Parco Paneveggio Pale di San Martino. Publistampa Arti grafiche, Pergine Valsugana (TN): 208
PAT - Provincia Autonoma di Trento - Ufficio Sistemi Informativi - Servizio autorizzazioni e valutazioni ambientali (2009) LIDAR rilievo 2006/2007/2008.
Peakall R, Smouse PE (2012) GenALEx 6.5: Genetic analysis in excel. population genetic software for teaching and research-an update. Bioinformatics 28:2537–2539
Quantum GIS Development Team (2015) Quantum GIS Geographic Information System. Open Source Geospatial Foundation Project.
Randi E, Lucchini V (1998) Organization and evolution of the mitochondrial DNA Control region in the avian genus Alectoris. J Mol Evol 47:449–462
Ratajc U, Breskvar M, Džeroski S, Vrezec A (2022) Differential responses of coexisting owls to annual small mammal population fluctuations in temperate mixed forest. Ibis 164:535–551
Rousset F (2008) GENEPOP’ 007: a Complete Re-implementation of the GENEPOP software for windows and Linux. Mol Ecol Resour 8:103–106
Royle JA, Young KV (2008) A hierarchical model for spatial capture-recapture data. Ecology 89:2281–2289
Royle JA, Richard BC, Sollmann R, Gardner B (2014) Spatial capture-recapture. Academic Press, Waltham
Sachot S, Perrin N, Neet C (2003) Winter habitat selection by two sympatric forest grouses in western Switzerland: implications for conservation. Biol Cons 112:373–382
Schäublin S, Bollmann K (2011) Winter habitat selection and conservation of Hazel Grouse (Bonasa bonasia) in mountain forests. J Ornithol 152:179–192
Shehzad W, Riaz T, Nawaz MA et al (2012) Carnivore diet analysis based on next-generation sequencing: application to the leopard cat (Prionailurus bengalensis) in Pakistan. Mol Ecol 21:1951–1965
Sitzia T, Dainese M, Clementi T et al (2014) Capturing cross-scalar variation of habitat selection with grid sampling: an example with hazel grouse (Tetrastes bonasia L). Eur J Wildl Res 60:177–186
Soininen EM, Valentini A, Coissac E et al (2009) Analysing diet of small herbivores: the efficiency of DNA barcoding coupled with high-throughput pyrosequencing for deciphering the composition of complex plant mixtures. Fronteers in Zoology 6:16
Sotirov M, Bastrup-Birk A, Blum M et al. (2017) Natura 2000 and forests: assessing the state of implementation and effectiveness. Retrieved from https://www.efi.int/publications-bank/natura2000-and-forests-assessing-state-implementation-and-effectiveness
Storch I (2000) Conservation status and threats to grouse worldwide: an overview. Wildl Biol 6:195–204
Swanson ME, Franklin JF, Beschta RL et al (2011) The forgotten stage of forest succession: early-successional ecosystems on forest sites. Front Ecol Environ 9:117–125
Swenson JE (1991) Evaluation of a density index for territorial male Hazel Grouse Bonasa bonasia in spring and autumn. Ornis Fennica 68:57–65
Swenson JE (1993) Hazel grouse (Bonasa bonasia) pairs during the nonbreeding season: mutual benefits of a cooperative alliance. Behav Ecol 4:14–21
Swenson JE, Olsson B (1991) Hazel grouse night roost site preferences when snow-roosting is not possible in winter. Ornis Scand 22:284–286
Taberlet P, Coissac E, Pompanon F et al (2007) Power and limitations of the chloroplast trn L (UAA) intron for plant DNA barcoding. Nucleic Acids Res 35:e14–e14
Taberlet P, Bonin A, Zinger L, Coissac E (2018) Environmental DNA: For biodiversity research and monitoring. Oxford University Press, UK
Taberlet P, Luikart G (1999) Non-invasive genetic sampling and indiv identification. Biological Journal of the Linnean Society 68:41–55
Therneau TM (2021) Survival: Survival analysis. R package version 3.2-11. https://CRAN.R-roject.org/package=survival
Thiel D, Unger C, Kéry M, Jenni L (2007) Selection of night roosts in winter by capercaillie Tetrao urogallus in central Europe. Wildlife Biol 13:73–86
Turcek FJ (1951) Galls and gall-insects as food of certain birds and mammals. Annal Entomol Fenn 17:17–22
Versluijs M, Hjältén J, Roberge JM (2019) Ecological restoration modifies the value of biodiversity indicators in resident boreal forest birds. Ecol Ind 98:104–111
Villesen P (2007) FaBox: an online toolbox for FASTA sequences. Mol Ecol Notes 7:965–968
Von Blotzheim, G., Solari, C. (1985) Tetraonidi. Rapporto della stazione ornitologica svizzera per l’Associazione degli amici della stazione ornitologica.
Wan QH, Hua W, Tsutomu F, Fang SG (2004) Which genetic marker for which conservation genetics issue? Electrophoresis 25:2165–2176
Zeale MR, Butlin RK, Barker GL, Lees DC, Jones G (2011) Taxon-specific PCR for DNA barcoding arthropod prey in bat faeces. Mol Ecol Resour 11:236–244
Zhang, D. (2021) rsq: R-Squared and related measures. R package version 2.2. https://CRAN.R-project.org/package=rsq
Acknowledgements
We thank A. Iemma for GIS support and two anonymous reviewers for helpful comments. We would also like to thank Paul Tout for proofreading and correcting the English in the manuscript.
Funding
This work was funded by Parco Naturale Paneveggio Pale di San Martino, Italy.
Author information
Authors and Affiliations
Contributions
All laboratory procedures dedicated to non-invasive samples were carried out in the ‘Department of Biology and Molecular Ecology’ laboratory at Fondazione Edmund Mach—Research and Innovation Center. PP, CV, PP, ST, and DS conceived the presented idea. RC, AF, MA, GV, ED, PP, and DS carried out fieldwork activities. DS and MB performed microhabitat selection analyses, whilst ST and DS worked with SCR models for home range estimates. RC, IF, AM, BC, CV performed all genetic-related analyses including diet. DS wrote the first version of the manuscript, and all authors equally contributed to the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could influence the work reported in this paper.
Ethical approval
Not applicable.
Consent to partecipate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Claus Bässler.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Scridel, D., Tenan, S., Brambilla, M. et al. Early-succession secondary forests following agropastoral abandonment are key winter habitats for the conservation of a priority bird in the European Alps. Eur J Forest Res 141, 1029–1043 (2022). https://doi.org/10.1007/s10342-022-01485-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-022-01485-1