Abstract
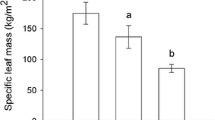
We analyzed leaf functional traits, chemical defenses and their effects on herbivory in three plant species that occur at two contrasting successional stages: mature and secondary forests in Mexico. In each successional stage, 15 individuals of Cordia elaeagnoides, C. alliodora and Achatocarpus gracilis were selected to analyze physical and chemical defenses and herbivory. Chlorophyll content, leaf thickness, leaf water content and leaf fresh mass were higher in mature forests, whereas SLA was higher in secondary forests, partially supporting the prediction that leaf traits vary from conservative to acquisitive along the leaf economics spectrum during succession. Content of secondary compounds did not show a consistent pattern among successional stages, but a relatively consistent trend was observed for C. elaeagnoides and A. gracilis, which exhibited higher levels of phenols and flavonoids in mature forests. Herbivory was higher in secondary than in mature forests for Cordia species, but the opposite pattern was found for A. gracilis. These results contradict the predictions of the resource availability hypothesis for tropical dry plant species, which state that resource-conservative plants in secondary forests invest more in chemical defenses and have less herbivore damage than resource-acquisitive plants in mature forests. Such idiosyncratic responses make it difficult to use a single hypothesis to predict plant defense and herbivory variations along environmental gradients at both intra- and interspecific levels. Considering the current intensity of human disturbance in forests, further studies are necessary to evaluate the impacts of changes in leaf functional traits on plant fitness during succession in the tropics.




Similar content being viewed by others
Data availability
All data obtained in this study are available from the corresponding author upon reasonable request.
References
Aerts R, Chapin FS (2000) The mineral nutrition of wild plants revisited: a re-evaluation. Adv Ecol Res 30:1–55. https://doi.org/10.1016/S0065-2504(08)60016-1
Agrawal AA (2020) A scale-dependent framework for trade-offs, syndromes, and specialization in organismal biology. Ecology 101:e02924. https://doi.org/10.1002/ecy.2924
Alvarez-Añorve MY, Quesada M, Sánchez-Azofeifa G, Avila-Cabadilla LD, Gamon JA (2012) Functional regeneration and spectral reflectance of trees during succession in a highly diverse tropical dry forest ecosystem. Am J Bot 99:816–826. https://doi.org/10.3732/ajb.1100200
Avila-Cabadilla LD, Sanchez-Azofeifa GA, Stoner KE, Alvarez-Añorve MY, Quesada M, Portillo-Quintero CA (2012) Local and landscape factors determining occurrence of phyllostomid bats in tropical secondary forests. PLoS ONE 7:e35228. https://doi.org/10.1371/journal.pone.0035228
Balvanera P, Lott E, Segura G, Siebe C, Islas A (2002) Patterns of β-diversity in a Mexican tropical dry forest. J Veg Sci 13:145–158. https://doi.org/10.1111/j.1654-1103.2002.tb02034.x
Baskett CA, Schemske DW (2018) Latitudinal patterns of herbivore pressure in a temperate herb support the biotic interactions hypothesis. Ecol Lett 21:578–587. https://doi.org/10.1111/ele.12925
Basset Y, Aberlenc HP, Barrios H (2001) Stratification and diel activity of arthropods assemblages. Biol J Linn Soc 72:585–607. https://doi.org/10.1111/j.1095-8312.2001.tb01340.x
Boege K, Dirzo R (2004) Intraspecific variation in growth, defense and herbivory in Dialium guianense (Caesalpiniaceae) mediated by edaphic heterogeneity. Plant Ecol 175:59–69. https://doi.org/10.1023/B:VEGE.0000048092.82296.9a
Boege K, Villa-Galaviz E, López-Carretero A, Pérez-Ishiwara R, Zaldivar-Riverón A, Ibarra A, Del-Val E (2019) Temporal variation in the influence of forest succession on caterpillar communities: a long-term study in a tropical dry forest. Biotropica 51:529–537. https://doi.org/10.1111/btp.12666
Bonada N, Dolédec S, Statzner, (2011) Spatial autocorrelation patterns of stream invertebrates: exogenous and endogenous factors. J Biogeogr 39:56–68. https://doi.org/10.1111/j.1365-2699.2011.02562.x
Bryant JP, Chapin FS, Klein DR (1983) Carbon/nutrient balance of boreal plants in relation to vertebrate herbivore. Oikos 40:357–368
Bryant JP, Chapin FS, Reichardt PB, Clausen TP (1987) Response of winter chemical defense in Alaska paper birch and green alder to manipulation of plant carbon/nutrient balance. Oecologia 72:510–514. https://doi.org/10.1007/BF00378975
Buzzard V, Hulshof CM, Birt T, Violle C, Enquist BJ (2016) Re-growing a tropical dry forest: functional plant trait composition and community assembly during succession. Funct Ecol 30:1006–1013. https://doi.org/10.1111/1365-2435.12579
Campos RI, Vasconcelos HL, Ribeiro SP, Neves FS, Soares JP (2006) Relationship between tree size and insect assemblages associated with Anadenanthera macrocarpa. Ecography 29:442–450. https://doi.org/10.1111/j.2006.0906-7590.04520.x
Chazdon RL (2014) Second growth: The promise of tropical forests. The University of Chicago Press, Chicago. https://doi.org/10.7208/9780226118109.001.0001
Coley PD (1988) Effects of plant growth rate and leaf lifetime on the amount and type of anti-herbivore defense. Oecologia 74:531–536. https://doi.org/10.1007/BF00380050
Coley PD, Bryant JP, Chapin FS (1985) Resource availability and plant antiherbivore defense. Science 230:895–899. https://doi.org/10.1126/science.230.4728.895
Connell JH, Slatyer RO (1977) Mechanisms of succession in natural communities and their role in community stability and organization. Am Nat 111:1119–1144
Cuevas-Reyes P, Quesada M, Siebe C, Oyama K (2004) Spatial patterns of herbivory by gall-forming insects: a test of the soil fertility hypothesis in a Mexican tropical dry forest. Oikos 107:181–189. https://doi.org/10.1111/j.0030-1299.2004.13263.x
Cuevas-Reyes P, De Oliveira-Ker FT, Fernandes GW, Bustamante M (2011) Abundance of gall-inducing insect species in sclerophyllous savanna: understanding the importance of soil fertility using an experimental approach. J Trop Ecol. https://doi.org/10.1017/S0266467411000368
Cuevas-Reyes P, Gilberti L, González-Rodríguez A, Fernandes GW (2013) Patterns of herbivory and fluctuating asymmetry in Solanum lycocarpum St. Hill (Solanaceae) along an urban gradient in Brazil. Ecol Indic 24:557–561. https://doi.org/10.1016/j.ecolind.2012.08.011
Cuevas-Reyes P, Canché-Delgado A, Maldonado-López Y, Fernandes GW, Oyama K, González-Rodríguez A (2018) Patterns of herbivory and leaf morphology in two Mexican hybrid oak complexes: importance of fluctuating asymmetry as indicator of environmental stress in hybrid plants. Ecol Indic 90:164–170. https://doi.org/10.1016/j.ecolind.2018.03.009
de la Riva EG, Marañón T, Pérez-Ramos IM, Navarro-Fernández CM, Olmo M, Villar R (2018) Root traits across environmental gradients in Mediterranean woody communities: are they aligned along the root economics spectrum? Plant Soil 424:35–48. https://doi.org/10.1007/s11104-017-3433-4
Deléglise C, Loucougaray G, Alard D (2011) Spatial patterns of species and plant traits in response to 20 years of grazing exclusion in subalpine grassland communities. J Veg Sci 22:402–413. https://doi.org/10.1111/j.1654-1103.2011.01277.x
Denno RF, Perfect TJ (1994) Planthoppers: their ecology and management. Chapman and Hall, New York
Díaz S, Kattge J, Cornelissen JH, Wright IJ, Lavorel S, Dray S et al (2016) The global spectrum of plant form and function. Nature 529:167–171
Didham RK, Springate ND (2003) Determinants of temporal variation in community structure. In Basset Y, Novotny V, Miller SE, Kitching RL (eds) Arthropods of tropical forests: spatio-temporal dynamics and resource use in the canopy, pp 28–39.
Ding Y, Zang R, Letcher SG, Liu S, He F (2012) Disturbance regime changes the trait distribution, phylogenetic structure and community assembly of tropical rain forests. Oikos 121:1263–1270
Donovan LA, Maherali H, Caruso CM, Huber H, de Kroon H (2011) The evolution of the worldwide leaf economics spectrum. Trends Ecol Evol 26:88–95
Endara MJ, Coley PD (2011) The resource availability hypothesis revisited: a meta-analysis. Funct Ecol 25:389–398. https://doi.org/10.1111/j.1365-2435.2010.01803.x
Ernest KA (1989) Insect herbivory on a tropical understory tree: effects of leaf age and habitat. Biotropica 21:194–199. https://doi.org/10.2307/2388642
Falcão HM, Medeiros CD, Silva BL, Sampai EV, Almeida-Cortez JS, Santos MG (2015) Phenotypic plasticity and ecophysiological strategies in a tropical dry forest chronosequence: a study case with Poincianella pyramidalis. For Ecol Manag 340:62–69. https://doi.org/10.1016/j.foreco.2014.12.029
Fonseca MB, Silva JO, Falcão LA, Dupin MG, Melo GA, Espírito-Santo MM (2018) Leaf damage and functional traits along a successional gradient in Brazilian tropical dry forests. Plant Ecol 219:403–415. https://doi.org/10.1007/s11258-018-0804-8
Frankie GW, Baker HG, Opler PA (1974) Comparative phenological studies of trees in tropical wet and dry forests in the lowlands of Costa Rica. J Ecol 62:881–919. https://doi.org/10.2307/2258961
Fu W, Zhao K, Zhang C, Tunney H (2011) Using Moran’s I and geostatistics to identify spatial patterns of soil nutrients in two different long-term phosphorus-application plots. J Plant Nutr Soil Sci 174:785–798. https://doi.org/10.1002/jpln.201000422
Fu WJ, Jiang PK, Zhou GM, Zhao KL (2014) Using Moran’s I and GIS to study the spatial pattern of forest litter carbon density in a subtropical region of southeastern China. Biogeosciences 11:2401–2409. https://doi.org/10.5194/bg-11-2401-2014
García-Oliva F, Camou A, Maass JM (2002) El clima de la región central de la costa del Pacífico mexicano. In Noguera FA, Vega-Rivera JH, García Aldrete AN, Quesada M (eds) Historia natural de Chamela. Mexico City, pp 3–10.
Gianoli E, Salgado-Luarte C (2017) Tolerance to herbivory and the resource availability hypothesis. Biol Lett 13:20170120. https://doi.org/10.1098/rsbl.2017.0120
González-Esquivel JG, Cuevas-Reyes P, González-Rodríguez A, Ávila-Cabadilla LD, Álvarez-Añorve MY, Fagundes M, Maldonado-López Y (2019) Functional attributes of two Croton species in different successional stages of tropical dry forest: effects on herbivory and fluctuating asymmetry patterns. Trop Ecol 60:238–251. https://doi.org/10.1007/s42965-019-00027-y
Grassein F, Till-Bottraud I, Lavorel S (2010) Plant resource-use strategies: the importance of phenotypic plasticity in response to a productivity gradient for two subalpine species. Ann Bot 106:637–645. https://doi.org/10.1093/aob/mcq154
Guariguata MR, Ostertag R (2001) Neotropical secondary forest succession: changes in structural and functional characteristics. For Ecol Manag 148:185–206. https://doi.org/10.1016/S0378-1127(00)00535-1
Hahn PG, Maron JL (2016) A framework for predicting intraspecific variation in plant defense. Trends Ecol Evol 31:646–656. https://doi.org/10.1016/j.tree.2016.05.007
Hamilton JG, Zangerl AR, DeLucia EH, Berenbaum MR (2001) The carbon-nutrient balance hypothesis: its rise and fall. Ecol Lett 4:86–95
Huang Y, Zhao X, Zhang H, Huang G, Luo Y, Japhet W (2009) A comparison of phenotypic plasticity between two species occupying different positions in a successional sequence. Ecol Res 24:1335
Huante P, Rincon E, Acosta I (1995) Nutrient availability and growth rate of 34 woody species from a tropical deciduous forest in Mexico. Funct Ecol 9:849–858. https://doi.org/10.2307/2389982
Jimenez-Rodríguez DL, Alvarez-Añorve MY, Flores-Puerto JI, Oyama K, Avila-Cabadilla LD, Pineda-Cortes M, Benítez-Malvido J (2018) Structural and functional traits predict short term response of tropical dry forests to a high intensity hurricane. Forest Ecol Manage 426:101–114. https://doi.org/10.1016/j.foreco.2018.04.009
Krimmel B, Pearse IS (2016) Tolerance and phenological avoidance of herbivory in tarweed species. Ecology 97:1357–1363. https://doi.org/10.1890/15-1454.1
Lebrija-Trejos E, Meave JA, Poorter L, Pérez-García EA, Bongers F (2010a) Pathways, mechanisms and predictability of vegetation change during tropical dry forest succession. Perspect Plant Ecol Evol Syst 12:267–275. https://doi.org/10.1016/j.ppees.2010.09.002
Lebrija-Trejos E, Pérez-García EA, Meave JA, Bongers F, Poorter L (2010b) Functional traits and environmental filtering drive community assembly in a species-rich tropical system. Ecology 91:386–398. https://doi.org/10.1890/08-1449.1
Lebrija-Trejos E, Pérez-García EA, Meave JA, Poorter L, Bongers F (2011) Environmental changes during secondary succession in a tropical dry forest in Mexico. J Trop Ecol 27:477–489. https://doi.org/10.1017/S0266467411000253
Letcher SG, Chazdon RL (2012) Life history traits of lianas during tropical forest succession. Biotropica 44:720–727
Letcher SG, Lasky JR, Chazdon RL, Norden N, Wright SJ et al (2015) Environmental gradients and the evolution of successional habitat specialization: a test case with 14 Neotropical forest sites. J Ecol 103:1276–1290. https://doi.org/10.1111/1365-2745.12435
Lobregat G, Perilli ML, de Siqueira NF, Campos RI (2018) Fluctuating asymmetry, leaf thickness and herbivory in Tibouchina granulosa: an altitudinal gradient analysis. Arthropod Plant Interact 12:277–282. https://doi.org/10.1007/s11829-017-9568-7
Lohbeck M, Poorter L, Lebrija-Trejos E, Martínez-Ramos M, Meave JA et al (2013) Successional changes in functional composition contrast for dry and wet tropical forest. Ecology 94:1211–1216. https://doi.org/10.1890/12-1850.1
Lohbeck M, Poorter L, Martínez-Ramos M, Rodriguez-Velázquez J, van Breugel M, Bongers F (2014) Changing drivers of species dominance during tropical forest succession. Funct Ecol 28:1052–1058. https://doi.org/10.1111/1365-2435.12240
Lohbeck M, Lebrija-Trejos E, Martínez-Ramos M, Meave JA, Poorter L et al (2015) Functional trait strategies of trees in dry and wet tropical forests are similar but differ in their consequences for succession. PLoS ONE 10:e0123741. https://doi.org/10.1371/journal.pone.0123741
Macedo-Reis LE, Novais SM, Monteiro GF, Flechtmann CA, Faria ML, Neves FD (2016) Spatio-temporal distribution of bark and ambrosia beetles in a Brazilian tropical dry forest. J Insect Sci 16:1–9. https://doi.org/10.1093/jisesa/iew027
Maldonado-López Y, Cuevas-Reyes P, Sánchez-Montoya G, Oyama K, Quesada M (2014) Growth, plant quality and leaf damage patterns in a dioecious tree species: Is gender important? Arthropod Plant Interact 8:241–251. https://doi.org/10.1007/s11829-014-9314-3
Marchiosi R, dos Santos WD, Constantin RP, de Lima RB, Soares AR, Finger-Teixeira A et al (2020) Biosynthesis and metabolic actions of simple phenolic acids in plants. Phytochem Rev 19:865–906. https://doi.org/10.1007/s11101-020-09689-2
Matsuura HN, Fett-Neto AG (2015) Plant alkaloids: main features, toxicity, and mechanisms of action. Plant Toxins 2:1–15. https://doi.org/10.1007/978-94-007-6728-7_2-1
Maza-Villalobos S, Balvanera P, Martínez-Ramos M (2011) Early regeneration of tropical dry forest from abandoned pastures: contrasting chronosequence and dynamic approaches. Biotropica 43:666–675. https://doi.org/10.1111/j.1744-7429.2011.00755.x
Maza-Villalobos S, Ackerly DD, Oyama K, Martínez-Ramos M (2020) Phylogenetic trajectories during secondary succession in a Neotropical dry forest: Assembly processes, ENSO effects and the role of legumes. Perspect Plant Ecol Evol Syst 43:125513. https://doi.org/10.1016/j.ppees.2020.125513
Méndez-Alonzo R, Paz H, Zuluaga RC, Rosell JA, Olson ME (2012) Coordinated evolution of leaf and stem economics in tropical dry forest trees. Ecology 93:2397–2406. https://doi.org/10.1890/11-1213.1
Méndez-Alonzo R, Pineda-García F, Paz H, Rosell JA, Olson ME (2013) Leaf phenology is associated with soil water availability and xylem traits in a tropical dry forest. Trees 27:745–754. https://doi.org/10.1007/s00468-012-0829-x
Muscarella R, Uriarte M, Aide TM, Erickson DL, Forero-Montaña J, Kress WJ et al (2016) Functional convergence and phylogenetic divergence during secondary succession of subtropical wet forests in Puerto Rico. J Veg Sci 27:283–294. https://doi.org/10.1111/jvs.12354
Neves FS, Silva JO, Espírito-Santo MM, Fernandes GW (2014) Insect herbivores and leaf damage along successional and vertical gradients in a tropical dry forest. Biotropica 46:14–24. https://doi.org/10.1111/btp.12068
Niinemets Ü (2001) Global-scale climatic controls of leaf dry mass per area, density, and thickness in trees and shrubs. Ecology 82:453–469. https://doi.org/10.1890/0012-9658(2001)082[0453:GSCCOL]2.0.CO;2
Ødegaard F (2003) Taxonomic composition and host specificity of phytophagous beetles in a dry forest in Panama. In: Basset Y, Novotny V, Miller S, Kitching R (eds) Arthropods of tropical forests: spatio-temporal dynamics and resource use in the canopy. Cambridge University Press, Cambridge, pp 220–236
Pascual-Alvarado E, Cuevas-Reyes P, Quesada M, Oyama K (2008) Interactions between galling insects and leaf-feeding insects: the role of plant phenolic compounds and their possible interference with herbivores. J Trop Ecol 24:329–336. https://doi.org/10.1017/S0266467408005038
Pennington TD, Sarukhán J (2005) Árboles tropicales de México. Manual para la identificación de las principales especies. Mexico City, Mexico.
Pérez-Harguindeguy N, Díaz S, Garnier E, Lavorel S, Poorter H, Jaureguiberry P et al (2016) Corrigendum to: new handbook for standardised measurement of plant functional traits worldwide. Aust J Bot 64:715–716. https://doi.org/10.1071/BT12225_CO
Pineda-García F, Paz H, Tinoco-Ojanguren C (2011) Morphological and physiological differentiation of seedlings between dry and wet habitats in a tropical dry forest. Plant Cell Environ 34:1536–1547. https://doi.org/10.1111/j.1365-3040.2011.02351.x
Pineda-García F, Paz H, Meinzer FC (2013) Drought resistance in early and late secondary successional species from a tropical dry forest: the interplay between xylem resistance to embolism, sapwood water storage and leaf shedding. Plant Cell Environ 36:405–418. https://doi.org/10.1111/j.1365-3040.2012.02582.x
Pires CS, Price PW (2000) Patterns of host plant growth and attack and establishment of gall-inducing wasp (Hymenoptera: Cynipidae). Environ Entomol 29:49–54. https://doi.org/10.1603/0046-225X-29.1.49
Poorter L, Van de Plassche M, Willems S, Boot RG (2004) Leaf traits and herbivory rates of tropical tree species differing in successional status. Plant Biol 6:746–754. https://doi.org/10.1055/s-2004-821269
Poorter H, Niinemets Ü, Poorter L, Wright IJ, Villar R (2009) Causes and consequences of variation in leaf mass per area (LMA): a meta-analysis. New Phytol 182:565–588. https://doi.org/10.1111/j.1469-8137.2009.02830.x
Poorter L, Castilho CV, Schietti J, Oliveira RS, Costa FR (2018) Can traits predict individual growth performance? A test in a hyperdiverse tropical forest. New Phytol 219:109–121. https://doi.org/10.1111/nph.15206
POWO (2019) Plants of the World Online. Facilitated by the Royal Botanic Gardens, Kew. Published on the Internet; http://www.plantsoftheworldonline.org/ Retrieved 15 August 2020.
Rasband WS (2006) ImageJ. Maryland, U.S.
Rausher MD (2006) The evolution of flavonoids and their genes. In: Grotewold E (ed) The evolution of flavonoids and their genes. Springer, New York, pp 175–211
Read QD, Moorhead LC, Swenson NG, Bailey JK, Sanders NJ (2014) Convergent effects of elevation on functional leaf traits within and among species. Funct Ecol 28:37–45. https://doi.org/10.1111/1365-2435.12162
Rossetti MR, Tscharntke T, Aguilar R, Batáry P (2017) Responses of insect herbivores and herbivory to habitat fragmentation: a hierarchical meta-analysis. Ecol Lett 20:264–272. https://doi.org/10.1111/ele.12723
Rzedowski J (1978) Vegetación de México. Mexico.
Sánchez-Azofeifa GA, Quesada M, Cuevas-Reyes P, Castillo A, Sánchez-Montoya G (2009) Land cover and conservation in the area of influence of the Chamela-Cuixmala Biosphere Reserve. Mexico for Ecol Manag 258:907–912. https://doi.org/10.1016/j.foreco.2008.10.030
Schönbeck L, Lohbeck M, Bongers F, Ramos M, Sterck F (2015) How do light and water acquisition strategies affect species selection during secondary succession in moist tropical forests? Forests 6:2047–2065. https://doi.org/10.3390/f6062047
Silva JO, Espírito-Santo MM, Melo GA (2012) Herbivory on Handroanthus ochraceus (Bignoniaceae) along a successional gradient in a tropical dry forest. Arthropod Plant Interact 6:45–57. https://doi.org/10.1007/s11829-011-9160
Silva JO, Espírito-Santo MM, Morais HC (2015) Leaf traits and herbivory on deciduous and evergreen trees in a tropical dry forest. Basic Appl Ecol 16:210–219. https://doi.org/10.1016/J.BAAE.2015.02.005
Somit D, Priyankar D, Kumar CT (2013) Quantification and correlation of the bioactive phytochemicals of Croton bonplandianum leaves of Sub-Himalayan region of West Bengal. Asian J Pharm Clin Res 6:142–147
Stiling P, Moon DC (2005) Quality or quantity: the direct and indirect effects of host plants on herbivores and their natural enemies. Oecologia 142:413–420
Strong DR, Lawton JH, Southwood SR (1984) Insects on plants. Community patterns and mechanisms. Blackwell Scientific Publications, New York
Tan SP, Parks SE, Stathopoulos CE, Roach PD (2014) Extraction of flavonoids from bitter melon. Food Nutr Sci 5:458–465. https://doi.org/10.4236/fns.2014.55054
Tiffin P, Ross-Ibarra J (2014) Advances and limits of using population genetics to understand local adaptation. Trends Ecol Evol 29:673–680. https://doi.org/10.1016/j.tree.2014.10.004
Uriarte M, Lasky JR, Boukili VK, Chazdon RL (2016) A trait-mediated, neighbourhood approach to quantify climate impacts on successional dynamics of tropical rainforests. Funct Ecol 30:157–167
Varanda EM, Pais MP (2006) Insect folivory in Didymopanax vinosum (Apiaceae) in a vegetation mosaic of Brazilian Cerrado. Braz J Biol 66:671–680. https://doi.org/10.1590/S1519-69842006000400011
War AR, Paulraj MG, Ahmad T, Buhroo AA, Hussain B, Ignacimuthu S, Sharma HC (2012) Mechanisms of plant defense against insect herbivores. Plant Signal Behav 7(10):1306–1320. https://doi.org/10.4161/psb.21663
Wright IJ, Westoby M (2002) Leaves at low versus high rainfall: coordination of structure, lifespan and physiology. New Phytol 155:403–416. https://doi.org/10.1046/j.1469-8137.2002.00479.x
Wright IJ et al (2004) The worldwide leaf economics spectrum. Nature 428:821–827. https://doi.org/10.1007/sll466-009-0028-z
Wu SJ, Ng LT (2008) Antioxidant and free radical scavenging activities of wild bitter melon (Momordica charantia Linn. var. abbreviata Ser.) in Taiwan. LWT-Food Sci Technol 41:323–330. https://doi.org/10.1016/j.lwt.2007.03.003
Acknowledgements
We thank the authorities of Chamela and Cuixmala Biosphere Reserve for their permission to conduct this study.
Funding
Aguilar-Peralta is a doctoral student from the Programa de Doctorado en Ciencias Biomédicas, Universidad Nacional Autónoma de México (UNAM), and has received CONACyT fellowship no. 620147. The study was funded by Coordination of Scientific Research (UMSNH), as part of research project 001. This project was supported by CONACYT Project No. CB222202. MMES acknowledges scholarships granted by CNPq (308471/2017–2) and CAPES (88881.337120/2019–01).
Author information
Authors and Affiliations
Contributions
PCR, LDAC, JSAP and MYAA contributed to the ideas and design research of this study. JSAP and MSVS collected the field data. RRC, KO and YML contributed to the design and direction of chemical methods to analyze foliar chemical defense. JSAP, MSVS and MF carried out statistical analysis. JSAP, PCR, KO, MMES and YML wrote the manuscript. All authors discussed the results and reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Additional information
Communicated by Claus Bässler.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aguilar-Peralta, J.S., Maldonado-López, Y., Espírito-Santo, M.M. et al. Contrasting successional stages lead to intra- and interspecific differences in leaf functional traits and herbivory levels in a Mexican tropical dry forest. Eur J Forest Res 141, 225–239 (2022). https://doi.org/10.1007/s10342-021-01434-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-021-01434-4