Abstract
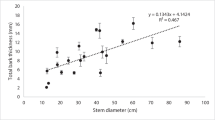
The decomposition rate of CWD is a key missing link for a quantitative understanding of forest ecosystem functioning. We examined factors influencing decomposition rates of bark, roots and branches from aspen (Populus tremula), birch (Betula pendula, B. pubescens), fir (Abies sibirica), spruce (Picea abies, P. obovata), Scots pine (Pinus sylvestris) and Siberian pine (Pinus sibirica) CWD in three primeval European boreal forests. The chronosequence approach with estimates of single exponential decomposition rate (k) based on calculation of mass loss was used. The k of non-stem parts increased in the order: branches (0.006 year−1 for P. sibirica and 0.020 year−1 for other species), roots in poorly drained sites (0.025 year−1), roots in well-drained sites (0.034 year−1) and bark (0.110 and 0.138 year−1 and 0.147 and 0.255 year−1 under poorly and well-drained conditions and from 1 to 3 m and >3 m above the root collar, respectively). Our results predict that the rate of decomposition of whole CWD pieces in European boreal forests is a function of vegetation zone, site conditions, tree species and size.






Similar content being viewed by others
References
Beets PN, Kimberley MO, Paul TSH, Garrett LG (2011) Planted forest carbon monitoring system—forest carbon model validation study for Pinus radiata. New Zeal J For Sci 41:177–189
Boddy L, Heilmann-Clausen J (2008) Basidiomycete community development in temperate angiosperm wood, Chapter 12. In: Boddy L, Frankland JS, West P (eds) Ecology of saprotrophic basidiomycetes. Academic press/Elsevier, London, pp 155–179
Boddy L, Rayner ADM (1983) Ecological roles of basidiomycetes forming decay communities in attached oak branches. New Phytol 93:77–88
Boddy L, Bardsley DW, Gibbon OM (1987) Fungal communities in attached ash branches. New Phytol 107:143–154
Bond-Lamberty B, Gower ST (2008) Decomposition and fragmentation of coarse woody debris: re-visiting a boreal black spruce chronosequence. Ecosystems 11:831–840
Bonh U, Gollub G, Hettwer C [Bearb.] (2000) Karte der natürlichen vegetation Europas/Map of the natural vegetation of Europe. Maßstab/Scale 1:2.500.000. Teil 2/Part 2: Legende/Legend, 153 S.; Teil 3/Part 3: Karten/Maps (9 Blätter/Sheets, Legendenblatt/LegendSheet, Übersichtskarte 1:10 Mio./General Map 1:10 million)—Münster (Landwirtschaftsverlag)
Bonh U, Neuhäusl R, unter Mitarbeit von Gollub G, Hettwer C, Neuhäuslová Z, Shlüter H, Weber H (2000/2003) Karte der natürlichen Vegetation Europas/Map of the Natural Vegetation of Europe. Maßstab/Scale 1:2.500.000. Teil 1/Part 1: Erläuterungsband/Explanatory Text, 655 S.; Teil 2/Part 2: Legende/Legend, 153 S.; Teil 3/Part 3: Karten/Maps (9 Blätter/Sheets, Legendenblatt/Legend Sheet, Übersichtskarte 1:10 Mio./General Map 1:10 million)—Münster (Landwirtschaftsverlag)
Bradford MA, Warren Ii RJ, Baldrian P, Crowther TW, Maynard DS, Oldfield EE, Wieder WR, Wood SA, King JR (2014) Climate fails to predict wood decomposition at regional scales. Nat Clim Change 4:625–630
Chambers JQ, Schimel JP, Nobre AD (2001) Respiration from coarse wood litter in central Amazon forests. Biogeochemistry 52:115–131
Chen H, Harmon M, Griffiths RP (2001) Decomposition and nitrogen release from decomposing woody roots in coniferous forests of the Pacific Northwest: a chronosequence approach. Can J For Res 31:246–260
Cramer W, Bondeau A, Woodward FI, Prentice IC, Betts RA, Brovkin V et al (2001) Global response of terrestrial ecosystem structure and function to CO2 and climate change: results from six dynamic global vegetation models. Glob Change Biol 7:357–373
Dittrich S, Jacob M, Bade C, Leuschner C, Hauck M (2014) The significance of deadwood for total bryophyte, lichen, and vascular plant diversity in an old-growth spruce forest. Plant Ecol 215(10):1123–1137
Erickson HE, Edmonds RL, Peterson CE (1985) Decomposition of logging residues in Douglas-fir, western hemlock, Pacific silver fir, and ponderosa pine ecosystems. Can J For Res 15:914–921
Fahey TJ, Hughes JW, Pu M, Arthur MA (1988) Root decomposition and nutrient flux following whole-tree harvest of northern hardwood forest. For Sci 34(3):744–768
Fedorchuk VN, Neshataev VYu, Kuznetsova ML (2005) Forest ecosystems of the north-western regions of Russia: typology, dynamics, management features. Forestry Scientific Research Institute, St. Petersburg, p 382 (in Russian)
Ganjegunte GK, Condron LM, Clinton PW, Davis MR, Mahieu N (2004) Decomposition and nutrient release from radiata pine (Pinus radiata) coarse woody debris. For Ecol Manag 187:197–211
Garrett LG, Kimberley MO, Oliver GR, Pearce SH, Beets PN (2012) Decomposition of coarse woody roots and branches in managed Pinus radiata plantations in New Zealand—a time series approach. For Ecol Manag 269:116–123
Harmon ME, Franklin JF, Swanson FJ, Sollins P, Gregory SV, Lattin JD, Anderson NH, Cline SP, Aumen NG, Sedell JR, Liencamper GW, Cromack K.Jr, Cummins KW (1986) Ecology of coarse woody debris in temperate ecosystems. Adv Ecol Res 15:133–202
Harmon ME, Whigham DF, Sexton J, Olmsted I (1995) Decomposition and mass of woody detritus in the dry tropical forests of the northeastern Yucatan Peninsula, Mexico. Biotropica 27(3):305–316
Harmon ME, Krankina ON, Sexton J (2000) Decomposition vectors: a new approach to estimating woody detritus decomposition dynamics. Can J For Res 30:76–84
Helfenstein J, Kienast F (2014) Ecosystem service state and trends at the regional to national level: a rapid assessment. Ecol Ind 36:11–18
Hyvönen R, Olsson BA, Lundkvist H, Staaf H (2000) Decomposition and nutrient release from Picea abies (L.) Karst. and Pinus sylvestris L. logging residues. For Ecol Manag 126:97–112
Janisch JE, Harmon ME, Chen H, Fasth B, Sexton J (2005) Decomposition of coarse woody debris originating by clearcutting of an old-growth conifer forest. EcoScience 12(2):151–160
Jo I, Fridley JD, Frank DA (2016) More of the same? In situ leaf and root decomposition rates do not vary between 80 native and nonnative deciduous forest species. New Phytol 209(1):115–122
Johnson EA, Miyanishi K (2008) Testing the assumptions of chronosequences in successions. Ecol Lett 11:419–431
Krankina ON, Harmon ME (1995) Dynamics of the dead wood carbon pool in northern-western Russian boreal forests. Water Air Soli Pollut 82:227–238
Kudeyarov VN, Zavarzin GA, Blagodatskaya SA (2007) Carbon pools and fluxes in Russia’s terrestrial ecosystems. Science Publ., Moscow (in Russian)
Laiho R, Prescott CE (2004) Decay and nutrient dynamics of coarse woody debris in northern coniferous forests: a synthesis. Can J For Res 34:763–777
Liu W, Schaefer D, Qiao L, Liu X (2013) What controls the variability of wood-decay rates? For Ecol Manag 310:623–631
Mackensen J, Bauhus J (1999) The decay of coarse woody debris (National Carbon Accounting System technical report no. 6) Commonwealth of Australia
Mäkinen H, Hynynen J, Siitonen J, Sievänen R (2006) Predicting the decomposition of Scots pine, Norway spruce and birch stems in Finland. Ecol Appl 16:1865–1879
Melin Y, Petersson H, Nordfjell T (2009) Decomposition of stump and root systems of Norway spruce in Sweden: a modeling approach. For Ecol Manag 257:1445–1451
Miller WE (1983) Decomposition rates of aspen bole and branch litter. For Sci 29:351–356
Moroni MT, Morris DM, Shaw C, Stokland JN, Harmon ME, Fenton NJ, Merganičová K, Merganič J, Okabe K, Hagemann U (2015) Buried wood: a common yet poorly documented form of deadwood. Ecosystems 18(4):605–628
Mukhin VA (1993) Biota of xylotrophic basidiomycetes of the West-Siberian Plain. Nauka Publ., Moscow (in Russian)
Olajuyigbe SO, Tobin B, Gardiner P, Nieuwenhuis M (2011) Stocks and decay dynamics of above- and belowground coarse woody debris in managed Sitka spruce forests in Ireland. For Ecol Manag 262:1109–1118
Olson JS (1963) Energy storage and the balance of producers and decomposers in ecological systems. Ecology 44:322–330
Palviainen M, Finér L (2015) Decomposition and nutrient release from Norway spruce coarse roots and stumps—a 40-year chronosequence study. For Ecol Manag 358:1–11
Palviainen M, Finér L, Laiho R, Shorohova E, Kapitsa E, Vanha-Majamaa I (2011) Phosphorus and base cation accumulation and release patterns in decomposing Scots pine, Norway spruce and silver birch stumps. For Ecol Manag 260:1478–1489
Parameswaran N, Wilhelm GE, Liese W (1976) Ultrastructural aspects of beech bark degradation by fungi. Eur J For Pathol 6:274–286
Polubojarinov OI (1976) Density of wood, vol 159. Nauka Publ., Moskow (in Russian)
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org/
Rayner ADM, Boddy L (1988) Fungal decomposition of wood: its biology and ecology. Wiley, Chichester
Russell MB, Fraver S, Aakala T, Gove JH, Woodall CW, D’Amato AW, Ducey MJ (2015) Quantifying carbon stores and decomposition in dead wood: a review. For Ecol Manag 350:107–128
Rypaček V (1957) Biologie dřeva dřevokaznŷch hub. Praha (in Czech)
Shaw RG, Mitchell-Olds T (1993) Anova for unbalanced data: an overview. Ecology 74:1638–1645
Shorohova E, Kapitsa E (2014a) Mineralization and fragmentation rates of bark attached to logs in a northern boreal forest. For Ecol Manag 315:185–190
Shorohova E, Kapitsa E (2014b) Influence of the substrate and ecosystem attributes on the decomposition rates of coarse woody debris in European boreal forests. For Ecol Manag 356:273–284
Shorohova E, Kapitsa E (2015) Stand and landscape scale variability in the amount and diversity of coarse woody debris in primeval European boreal forests. For Ecol Manag 315:185–190
Shorohova E, Ignatyeva O, Kapitsa E, Kauhanen H, Kuznetsov A, Vanha-Majamaa I (2012) Stump decomposition rates after clear-felling with and without prescribed burning in southern and northern boreal forests in Finland. For Ecol Manag 263:74–84
Stokland JN, Siitonen J, Jonsson BG (2012) Biodiversity in dead wood. Cambrigde Univ. Press, Cambrigde
Swift MJ, Healey IN, Hibberd JK, Sykes JM, Bampoe V, Nesbitt ME (1976) The decomposition of branch-wood in the canopy and floor of a mixed deciduous woodland. Oecologia 26(2):139–149
Tarasov ME, Birdsey RA (2001) Decay rate and potential storage of course woody debris in the Leningrad region. Ecol Bull 49:137–149
Tetioukhin SV, Minayev VN, Bogomolova LP (2004) Forest Mensuration and inventory: reference book for the North-Western Russia. StP. FTA Publ, St. Petersburg (in Russian)
Tobin B, Black K, Mcgurdy M, Nieuwenhuis M (2007) Estimates of decay rates of components of coarse woody debris in thinned Sitka spruce forests. Forestry 80(4):455–469
Weedon JT, Cornwell WK, Cornelissen JHC, Zanne AE, Wirth C, Coomes DA (2009) Global meta-analysis of wood decomposition rates: a role for trait variation among tree species? Ecol Lett 12:45–56
Yatskov M, Harmon M, Krankina O (2003) A chronosequence of wood decomposition in the boreal forests of Russia. Can J For Res 33:1211–1226
Yin X (1999) The decay of forest woody debris: numerical modeling and implications based on some 300 data cases from North America. Oecologia 121:81–98
Zamolodchicov DG, Utlin AI, Korovin GN (1998) Determination of carbon stores by conversion-volumetric coefficients related to age of stands. Lesovedenie 3:84–93 (in Russian)
Zamolodchicov DG, Grabovsky VI, Kraev VN (2011) Dynamics of carbon budget of Russia’s forests during the last two decades. Lesovedenie 6:16–28 (in Russian)
Acknowledgments
The research was supported by the Russian Science Foundation (15-14-10023). The data were collected with financial support from the Russian Foundation of Basic Research (09-04-00209-a). The staff of Nature Park “Vepssky Forest,” Central Forest Biosphere State Natural Reserve and Yugyd-va National Park helped with practicalities during expeditions. We sincerely thank all people who helped us in the fieldwork. Olga Lisitsyna created the map of the study sites. Carla Burton revised the language.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shorohova, E., Kapitsa, E. The decomposition rate of non-stem components of coarse woody debris (CWD) in European boreal forests mainly depends on site moisture and tree species. Eur J Forest Res 135, 593–606 (2016). https://doi.org/10.1007/s10342-016-0957-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10342-016-0957-8