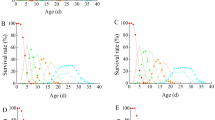
Abstract
Interspecific competition occurs fiercely between invasive and native species which occupy similar niche. Here, we report the competitive mechanism underlying the displacement of Bursaphelenchus mucronatus by Bursaphelenchus xylophilus. In the early stage of the competition, population of both species increase significantly, but their abundance rates were differentiated, B. xylophilus increased, whereas B. mucronatus decreased. Unexpectedly, sex ratio of female to male of both species increased sharply and peaked at 4.87 and 3.88, respectively, which were outclassed that of noncompetitive conditions (2.77 and 2.21). Thereafter, the sex ratio of B. xylophilus declined and stabilized at 2.81, but the sex ratio of B. mucronatus declined till no B. mucronatus was left, indicating the interspecific competition could be dominated by female. The mating behaviors showed that for both species, the mating success rate of one couple dropped slightly when one male or female of other species was added, but dropped significantly when two males or females were added. Surprisingly, almost no successful mating was observed when three males or females of other species were added. We therefore hypothesize that the mating opportunity of one couple could be deprived by the other species once three or more male or female were added. Further, threshold value of competitive displacement experiments and model analysis were completely consistent with the above experiments. Our results elucidate the crucial role of sex ratio in interspecific competition for the first time, as well as their mating disruption, directing a wonderful story of female-dominated war between two xylophilus group nematode species.




Similar content being viewed by others
Availability of data and materials
The materials and datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Akbulut S, Stamps WT (2012) Insect vectors of the pinewood nematode: a review of the biology and ecology of Monochamus species. Forest Pathol 42(2):89–99. https://doi.org/10.1111/j.1439-0329.2011.00733.x
Akbulut S, Yüksel B, Serin M et al (2007) Pathogenicity of Bursaphelenchus mucronatus in pine seedlings under greenhouse conditions. Turk J Agric for 31(3):169–173
Andrews RH, Petney TN, Bull CM (1982) Reproductive interference between three parapatric species of reptile tick. Oecologia 52(2):281–286. https://doi.org/10.1007/BF00363851
Bolla RI, Boschert M (1993) Pinewood nematode species complex: interbreeding potential and chromosome number. J Nematol 25(2):227–238
Braasch H (1996) Pathogenitätstests mit Bursaphelenchus mucronatus an kiefern und fichtensämlingen in Deutschland. Eur J for Pathol 26(4):205–216. https://doi.org/10.1111/j.1439-0329.1996.tb00840.x
Brzeski MW (1997) Survey of Bursaphelenchus (Nematoda: Aphelenchoididae) species in pine wood of Poland. Fragmenta Faunistica 4:103–109. https://doi.org/10.3161/00159301FF1997.40.10.103
Burdfield-Steel ER, Shuker DM (2011) Reproductive interference. Curr Biol 21(12):R450. https://doi.org/10.1016/j.cub.2011.03.063
Caroppo S, Ambrogioni L, Cavalli M et al (1998) Occurrence of the pine wood nematodes, Bursaphelenchus spp., and their possible vectors in Italy. Nematol Mediterr 26:87–92
Case TJ, Gilpin ME (1974) Interference competition and niche theory. Proc Natl Acad Sci USA 71(8):3073–3077. https://doi.org/10.1073/pnas.71.8.3073
Cheng H, Lin M, Qian R (1986) A study on the morphological diagnosis and the pathogenicity of Bursaphelenchus mucronatus. J Nanjing Agric Univ 1:55–61. https://doi.org/10.7685/j.issn.1000-2030.1986.02.008
Cheng X, Xie PZ, Cheng FX et al (2009) Competitive displacement of the native species Bursaphelenchus mucronatus by an alien species Bursaphelenchus xylophilus (Nematoda: Aphelenchida: Aphelenchoididae): a case of successful invasion. Biol Invasions 11(2):205–213. https://doi.org/10.1007/s10530-008-9225-2
Dame EA, Petren K (2006) Behavioural mechanisms of invasion and displacement in Pacific island geckos (Hemidactylus). Anim Behav 71(5):1165–1173. https://doi.org/10.1016/j.anbehav.2005.10.009
de Bruyn PJN, Tosh CA, Bester MN (2008) Sexual harassment of a king penguin by an Antarctic fur seal. J Ethol 26(2):295–297. https://doi.org/10.1007/s10164-007-0073-9
Elmendorf SC, Harrison SP (2011) Is plant community richness regulated over time? Contrasting results from experiments and long-term observations. Ecology 92(3):602–609. https://doi.org/10.1890/10-0535.1
Fonseca L, Cardoso J, Lopes A et al (2012) The pinewood nematode, Bursaphelenchus xylophilus. Madeira Island Helminthol 49(2):96–103. https://doi.org/10.2478/s11687-012-0020-3
Futai K (1980a) Developmental rate and population growth of Bursaphelenchus lignicolus (Nematoda: Aphelenchoididae) and B. mucronatus. Appl Entomol Zool 15:115–122. https://doi.org/10.1303/aez.15.115
Futai K (1980b) Population dynamics of Bursaphelenchus lignicolus (Nematoda: Aphelenchoididae) and B. mucronatus in pine seedlings. Appl Entomol Zool 15:458–464. https://doi.org/10.1303/aez.15.458
Gröning J, Hochkirch A (2008) Reproductive interference between animal species. Q R Biol 83(3):257–282. https://doi.org/10.1086/590510
Iwahori H, Kanzaki N, Futai K (2000) A simple, polymerase chain reaction-restriction fragment length polymorphism-aided diagnosis method for pine wilt disease. For Pathol 30(3):157–164. https://doi.org/10.1046/j.1439-0329.2000.00201.x
Jikumaru S, Togashi K (2008) Resistance of an indigenous biological system against expansion of the invasive nematode, Bursaphelenchus xylophilus, in cool areas of Japan. Nematology 10(5):679–687
Kang JS, Choi KS, Shin SC et al (2004) Development of an efficient PCR-based diagnosis protocol for the identification of the pinewood nematode, Bursaphelenchus xylophilus (Nematoda: Aphelenchoididae). Nematology 6(2):279–285
Kanzaki N, Futai K (2002) A PCR primer set for determination of phylogenetic relationships of Bursaphelenchus species within the xylophilus group. Nematology 4(1):35–41. https://doi.org/10.1163/156854102760082186
Kikuchi T, Cotton JA, Dalzell JJ et al (2011) Genomic insights into the origin of parasitism in the emerging plant pathogen Bursaphelenchus xylophilus. PLoS Pathog 7(9):e1002219. https://doi.org/10.1371/journal.ppat.1002219
Kulinich OA, Kruglic I, Eroshenko AS et al (1994) Occurrence and distribution of the nematode Bursaphelenchus mucronatus in the Russian Far East. Russ J Nematol 2:113–120
Kyogoku D, Sota T (2015) Exaggerated male genitalia intensify interspecific reproductive interference by damaging heterospecific female genitalia. J Evol Biol 28(6):1283–1289. https://doi.org/10.1111/jeb.12646
Kyogoku D, Kondoh M, Sota T (2019) Does past evolutionary history under different mating regimes influence the demographic dynamics of interspecific competition? Ecol Evol 9:8616–8624. https://doi.org/10.1002/ece3.5397
Liao SM, Kasuga S, Togashi K (2014) Suppressive effects of Bursaphelenchus mucronatus on pine wilt disease development and mortality of B. xylophilus-inoculated pine seedlings. Nematology 16(2):219–227
Lopez LK, Davis AR, Wong MYL (2018) Behavioral interactions under multiple stressors: temperature and salinity mediate aggression between an invasive and a native fish. Biol Invasions 20:487–499. https://doi.org/10.1007/s10530-017-1552-8
Mamiya Y (1988) History of pine wilt disease in Japan. J Nematol 20(20):219–226
Mamiya Y, Enda N (1979) Bursaphelenchus mucronatus n. sp. (Nematoda: Aphelenchoididae) from pine wood and its biology and pathogenicity to pine trees. Nematologica 3(3):353–361. https://doi.org/10.1163/187529279X00091
Mamiya Y, Kiyohara T (1972) Description of Bursaphelenchus lignicolus n. sp. (Nematoda: Aphelenchoididae) from pine wood and histopathology of nematode-infested trees. Nematologica 18(1):120–124. https://doi.org/10.1163/187529272X00296
Mooney HA, Cleland EE (2001) The evolutionary impact of invasive species. Proc Natl Acad Sci USA 98(10):5446–5451. https://doi.org/10.1073/pnas.091093398
Mota MM, Braasch H, Bravo MA et al (1999) First report of Bursaphelenchus xylophilus in Portugal and in Europe. Nematology 1(7):727–734
Niu HT, Zhao LL, Sun JH (2013) Phenotypic plasticity of reproductive traits in response to food availability in invasive and native species of nematode. Biol Invasions 15(7):1407–1415. https://doi.org/10.1007/s10530-012-0379-6
Prospero S, Polomski J, Rigling D (2015) Occurrence and ITS diversity of wood-associated Bursaphelenchus nematodes in Scots pine forests in Switzerland. Plant Pathol 64(5):1190–1197. https://doi.org/10.1111/ppa.12356
Riga E, Beckenbach K, Webster JM (1992) Taxonomic relationships of Bursaphelenchus xylophilus and B mucronatus based on interspecific and intraspecific cross-hybridization and DNA analysis. Fundam Appl Nematol 15(5):391–395
Robertson L, Arcos SC, Escuer M et al (2011) Incidence of the pinewood nematode Bursaphelenchus xylophlius Steiner & Buhrer, 1934 (Nickle, 1970) in Spain. Nematology 13(6):755–757
Taga Y, Goto S, Matsunaga K et al (2011) Temporal changes in characteristics of populations originating from interbreeding between Bursaphelenchus xylophilus and B. mucronatus. Nematology 13(6):701–712
Tares S, Lemontey JM, de Guiran G et al (1994) Use of species-specific satellite DNA from Bursaphelenchus xylophilus as a diagnostic probe. Phytopathology 84(3):294–298. https://doi.org/10.1094/Phyto-84-294
Vicente CSL, Soares M, Faria JMS et al (2021) Insights into the fole of fungi in pine wilt disease. J Fungi 7:e780. https://doi.org/10.3390/jof7090780
Viglierchio DR, Schmitt RV (1983) On the methodology of nematode extraction from field samples: Baermann funnel modifications. J Nematol 15(3):438–444
Vilà M, Espinar JL, Martin H et al (2011) Ecological impacts of invasive alien plants: a meta-analysis of their effects on species, communities and ecosystems. Ecol Lett 14(7):702–708. https://doi.org/10.1111/j.1461-0248.2011.01628.x
Vincent B, Altemayer V, Rouxmorabito G et al (2008) Competitive interaction between Bursaphelenchus xylophilus and the closely related species Bursaphelenchus mucronatus. Nematology 10(2):219–230
Wang Y, Yamada T, Sakaue D et al (2005) Variations in life history parameters and their influence on rate of population increase of different pathogenic isolates of the pine wood nematode, Bursaphelenchus Xylophilus. Nematology 7(3):459–467
Wauters LA, Mazzamuto MV, Santicchia F et al (2019) Interspecific competition affects the expression of personality-traits in natural populations. Sci Rep 9(1):e11189. https://doi.org/10.1038/s41598-019-47694-4
Yi CK, Byun BH, Park JD et al (1989) First finding of the pine wood nematode, Bursaphelenchus xylophilus (Steiner & Buhrer) Nickle and its insect vector in Korea. Res Rep for Res Inst 38:141–149
Zhao J, Han X, Shi J (2017) Potential distribution of Bursaphelenchus xylophilus in China due to adaptation cold conditions. J Biosaf 26(3):191–198. https://doi.org/10.3969/j.issn.2095-1787.2017.03.003
Zhao L, Zhang X, Wei Y et al (2016) Ascarosides coordinate the dispersal of a plant-parasitic nematode with the metamorphosis of its vector beetle. Nat Commun 7:e12341. https://doi.org/10.1038/ncomms12341
Zhou LF, Chen FM, Xie LY et al (2017) Genetic diversity of pine-parasitic nematodes Bursaphelenchus xylophilus and Bursaphelenchus mucronatus in China. For Pathol 47(4):e12334. https://doi.org/10.1111/efp.1233
Zhu N, Bai L, Schütz S et al (2016) (2016) Observation and quantification of mating behavior in the pinewood nematode, Bursaphelenchus xylophilus. J vis Exp 118:e54842. https://doi.org/10.3791/54842
Funding
This work was supported by the National Natural Science Foundation of China (31670652 and 31870633) and the Launching Funds for Talents of Zhejiang A & F University (2022LFR004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethical Approval
Not applicable.
Additional information
Communicated by Aurelio Ciancio.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file2 (MP4 5423 kb)
Supplementary file3 (MP4 5255 kb)
Supplementary file4 (MP4 5261 kb)
Supplementary file5 (MP4 6734 kb)
Supplementary file6 (MP4 5813 kb)
Supplementary file7 (MP4 8788 kb)
Supplementary file8 (MP4 5483 kb)
Supplementary file9 (MP4 5582 kb)
Supplementary file10 (MP4 5747 kb)
Supplementary file11 (MP4 7232 kb)
Supplementary file12 (MP4 5965 kb)
Supplementary file13 (MP4 6391 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, L., Liu, W., Bai, L. et al. Female's war: a story of the invasion and competitive displacement between two xylophilus group nematode species. J Pest Sci 96, 1301–1311 (2023). https://doi.org/10.1007/s10340-023-01603-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-023-01603-x