Abstract
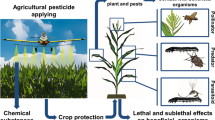
Natural pest control is a significant service supporting agricultural production. However, the relative contribution of several functional groups of natural enemies to natural pest control is unknown for many crop-pest systems. Furthermore, the output of this regulation in terms of damage reduction is rarely assessed. In this study, based on three field experiments, we quantified the effect of ground dwelling predators and parasitoids on two functional groups of broccoli pests. Relying on physical exclusion, we showed that ground dwelling predators significantly lowered the abundance of both the cabbage root fly (Delia radicum) and aphids (Brevicoryne brassicae and Myzus persicae). Ground dwelling predators made a significant contribution to natural pest control, as they lowered pest populations by 37% on average. Parasitoids suppressed 22% of pest populations. Our results suggest that intraguild predation was not an issue since ground dwelling predators did not have a negative impact on the level of natural pest control by parasitoids. Finally, early predation by ground dwelling predators on the cabbage root fly, the most harmful pest in this study, reduced damage in a highly infested context, meaning that natural pest control can effectively support crop production. These results strongly suggest that agricultural practices limiting soil disturbances or even favoring ground dwelling predator overwintering or colonization could lead to fewer damage and losses for farmers.






Similar content being viewed by others
Data availability
Data are freely available at the following link: https://doi.org/10.15454/IOBLWS.
Code availability
The R code used to analyze data and perform figures is freely available at the following link: https://doi.org/10.15454/IOBLWS
References
Agreste (2020) Statistique agricole annuelle 2019 : donnees définitives. Agreste, la statistique agricole, Service de la statistique et de la prospective (SSP), Angélique Toulon
Albrecht M, Kleijn D, Williams NM et al (2020) The effectiveness of flower strips and hedgerows on pest control, pollination services and crop yield: a quantitative synthesis. Ecol Lett. https://doi.org/10.1111/ele.13576
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Begg GS, Cook SM, Dye R et al (2017) A functional overview of conservation biological control. Crop Prot 97:145–158. https://doi.org/10.1016/j.cropro.2016.11.008
Birkhofer K, Gavish-Regev E, Endlweber K et al (2008) Cursorial spiders retard initial aphid population growth at low densities in winter wheat. B Entomol Res 98:249–255. https://doi.org/10.1017/S0007485308006019
Birkhofer K, Bylund H, Dalin P et al (2017) Methods to identify the prey of invertebrate predators in terrestrial field studies. Ecol Evol 7:1942–1953. https://doi.org/10.1002/ece3.2791
Boetzl FA, Schuele M, Krauss J, Steffan-Dewenter I (2020) Pest control potential of adjacent agri-environment schemes varies with crop type and is shaped by landscape context and within-field position. J Appl Ecol 57:1482–1493. https://doi.org/10.1111/1365-2664.13653
Bourhis Y, Poggi S, Mammeri Y et al (2015) Perception-based foraging for competing resources: Assessing pest population dynamics at the landscape scale from heterogeneous resource distribution. Ecol Model 312:211–221. https://doi.org/10.1016/j.ecolmodel.2015.05.029
Boyd J, Banzhaf S (2007) What are ecosystem services? The need for standardized environmental accounting units. Ecol Econ 63:616–626. https://doi.org/10.1016/j.ecolecon.2007.01.002
Brooks ME, Kristensen K, van Benthem KJ et al (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. R J 9:378–400. https://doi.org/10.3929/ethz-b-000240890
Brust G (1991) A method for observing belowground pest-predator interactions in corn agroecosystems. J Entomol Sci 26:1–8
Chau A, Mackauer M (1997) Dropping of pea aphids from feeding site: a consequence of parasitism by the wasp, Monoctonus paulensis. Entomol Exp Appl 83:247–252. https://doi.org/10.1046/j.1570-7458.1997.00179.x
Christensen RHB (2019) Ordinal---regression models for ordinal data. Version R package version 2019.12–10URL https://CRAN.R-project.org/package=ordinal
Coaker TH (1965) Further experiments on the effect of beetle predators on the numbers of the cabbage root fly, Erioischia brassicae (Bouché), attacking Brassica crops. Ann Appl Biol 56:7–20. https://doi.org/10.1111/j.1744-7348.1965.tb01210.x
Colfer RG, Rosenheim JA (2001) Predation on immature parasitoids and its impact on aphid suppression. Oecologia 126:292–304. https://doi.org/10.1007/s004420000510
Collins KL, Boatman ND, Wilcox A et al (2002) Influence of beetle banks on cereal aphid predation in winter wheat. Agric Ecosyst Environ 93:337–350. https://doi.org/10.1016/S0167-8809(01)00340-1
Costello MJ, Altieri MA (1995) Abundance, growth rate and parasitism of Brevicoryne brassicae and Myzus persicae (Homoptera: Aphididae) on broccoli grown in living mulches. Agric Ecosyst Environ 52:187–196. https://doi.org/10.1016/0167-8809(94)00535-M
Dainese M, Schneider G, Krauss J, Steffan-Dewenter I (2017) Complementarity among natural enemies enhances pest suppression. Sci Rep 7:8172. https://doi.org/10.1038/s41598-017-08316-z
Derocles SAP, Navasse Y, Buchard C et al (2020) Generalist aphid parasitoids behave as specialists at the agroecosystem scale. Insects 11:6. https://doi.org/10.3390/insects11010006
Dosdall LM, Herbut MJ, Cowle NT (1994) Susceptibilities of species and cultivars and canola and mustard to infestation by root maggots (Delia spp.) (Diptera, Anthomyiidae). Can Entomol 126:251–260. https://doi.org/10.4039/Ent126251-2
Ekbom BS, Wiktelius S, Chiverton PA (1992) Can polyphagous predators control the bird cherry-oat aphid (Rhopalosiphum padi) in spring cereals?: a simulation study. Entomol Exp Appl 65:215–223. https://doi.org/10.1111/j.1570-7458.1992.tb00674.x
Estorgues V, Cochard L (2004) Maladies et ravageurs des choux : synthèse de 4 années de suivis et d’observations dans le Finistère (2001–2004). Chambre d’agriculture Finistère ; Comité de développement des agriculteurs - zone légumière
Faraway JJ (2006) Extending the linear model with R: generalized linear, mixed effects and non parametric regression models. Chapman & Hall/CRC Taylor & Francis group, London
Finch S, Elliott MS (1994) Predation of cabbage root fly eggs and larvae by carabid ground beetles - fact or fantasy? IOBC-WPRS Bull 17:109–114
Fox J, Weisberg S (2019) An R companion to applied regression, 3rd edn. Sage, Thousand Oaks (CA)
Freuler J, Fischer S (1983) Le piège à oeufs, nouveau moyen de prévision d’attaque pour la mouche du chou, Delia radicum (brassicae) L. Revue Suisse Vitic Arboric Hortic 15:107–110
Geo W, Monteith E (1954) Trybliographa rapae (Westw.) (Hymenoptera: Cynipidae), a parasite of Hylemia spp. (Diptera: Anthomyiidae). Can Entomol 86:145–154
Gontijo LM, Beers EH, Snyder WE (2015) Complementary suppression of aphids by predators and parasitoids. Biol Control 90:83–91. https://doi.org/10.1016/j.biocontrol.2015.06.002
Harrison XA (2014) Using observation-level random effects to model overdispersion in count data in ecology and evolution. PeerJ 2:e616. https://doi.org/10.7717/peerj.616
Hartig F (2020) Package “DHARMa” Residual Diagnostics for Hierarchical (Multi-Level/Mixed) Regression Models. Version 0.2.7
Holland JM, Thomas SR (1997) Quantifying the impact of polyphagous invertebrate predators in controlling cereal aphids and in preventing wheat yield and quality reductions. Ann Appl Biol 131:375–397. https://doi.org/10.1111/j.1744-7348.1997.tb05167.x
Holland JM, Thomas SR, Hewitt A (1996) Some effects of polyphagous predators on an outbreak of cereal aphid (Sitobion avenae F.) and orange wheat blossom midge (Sitodoplosis mosellana Géhin). Agr Ecosyst Environ 59:181–190. https://doi.org/10.1016/0167-8809(96)01053-5
Hughes RD, Salter DD (1959) Natural mortality of Erioischia brassicae (Bouché) (Diptera, Anthomyiidae) during the immature stages of the first generation. J Anim Ecol 28:231–241. https://doi.org/10.2307/2080
Kamenova S, Tougeron K, Cateine M et al (2015) Behaviour-driven micro-scale niche differentiation in carabid beetles. Entomol Exp Appl 155:39–46. https://doi.org/10.1111/eea.12280
Kendall DA (2003) Soil tillage and epigeal predatory arthropods. Soil tillage in agroecosystems. CRC Press, Florida, pp 297–342
Kleijn D, Baquero RA, Clough Y et al (2006) Mixed biodiversity benefits of agri-environment schemes in five European countries. Ecol Lett 9:243–254. https://doi.org/10.1111/j.1461-0248.2005.00869.x
Kromp B (1999) Carabid beetles in sustainable agriculture: a review on pest control efficacy, cultivation impacts and enhancement. Agr Ecosyst Environ 74:187–228. https://doi.org/10.1016/S0167-8809(99)00037-7
Lamy F, Dugravot S, Cortesero AM et al (2017) One more step toward a push-pull strategy combining both a trap crop and plant volatile organic compounds against the cabbage root fly Delia radicum. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-017-9483-6
Lavorel S, Storkey J, Bardgett RD et al (2013) A novel framework for linking functional diversity of plants with other trophic levels for the quantification of ecosystem services. J Veg Sci 24:942–948. https://doi.org/10.1111/jvs.12083
Leather SR, Walters KFA, Dixon AFG (1989) Factors determining the pest status of the bird cherry-oat aphid, Rhopalosiphum padi (L.) (Hemiptera: Aphididae), in Europe: a study and review. B Entomol Res 79:345–360. https://doi.org/10.1017/S0007485300018344
Lee JC, Edwards DL (2012) Impact of predatory carabids on below- and above-ground pests and yield in strawberry. Biocontrol 57:515–522. https://doi.org/10.1007/s10526-011-9425-z
Lenth RV (2020) emmeans: Estimated Marginal Means, aka Least-Squares Means. Version R package version 1.4.5URL https://CRAN.R-project.org/package=emmeans
Letourneau DK, Jedlicka JA, Bothwell SG, Moreno CR (2009) Effects of natural enemy biodiversity on the suppression of arthropod herbivores in terrestrial ecosystems. Annu Rev Ecol Syst 40:573–592. https://doi.org/10.1146/annurev.ecolsys.110308.120320
Losey JE, Denno RF (1998) Positive predator-predator interactions: enhanced predation rates and synergistic suppression of aphid populations. Ecology 79:2143–2152. https://doi.org/10.1890/0012-9658(1998)079[2143:PPPIEP]2.0.CO;2
Luck RF, Shepard BM, Kenmore PE (1988) Experimental methods for evaluating arthropod natural enemies. Annu Rev Entomol 33:367–389. https://doi.org/10.1146/annurev.en.33.010188.002055
Mesmin X, Vincent M, Tricault Y et al (2019) Assessing the relationship between pest density and plant damage: a case study with the belowground herbivore Delia radicum (Diptera: Anthomyiidae) on broccoli. Appl Entomol Zool 54:155–165. https://doi.org/10.1007/s13355-019-00607-3
Navasse Y, Derocles SAP, Plantegenest M, Ralec AL (2017) Ecological specialization in Diaeretiella rapae (Hymenoptera: Braconidae: Aphidiinae) on aphid species from wild and cultivated plants. B Entomol Res. https://doi.org/10.1017/S0007485317000657
Navasse Y (2016) Spécialisation parasitaire chez les Aphidiinae : existe-t-il des parasitoïdes de pucerons généralistes? PhD thesis, Université de Rennes 1
Neuville S, Le Ralec A, Outreman Y, Jaloux B (2016) The delay in arrival of the parasitoid Diaeretiella rapae influences the efficiency of cabbage aphid biological control. Biocontrol 61:115–126. https://doi.org/10.1007/s10526-015-9702-3
Ortiz-Martínez S, Staudacher K, Baumgartner V et al (2020) Intraguild predation is independent of landscape context and does not affect the temporal dynamics of aphids in cereal fields. J Pest Sci 93:235–249. https://doi.org/10.1007/s10340-019-01142-4
Östman Ö, Ekbom B, Bengtsson J (2003) Yield increase attributable to aphid predation by ground-living polyphagous natural enemies in spring barley in Sweden. Ecol Econ 45:149–158. https://doi.org/10.1016/S0921-8009(03)00007-7
Polis GA, Myers CA, Holt RD (1989) The ecology and evolution of intraguild predation: potential competitors that eat each other. Annu Rev Ecol Syst 20:297–330. https://doi.org/10.1146/annurev.es.20.110189.001501
Pollard E (1969) The Effect of Removal of Arthropod Predators on an Infestation of Brevicoryne brassicae (Hemiptera, Aphididae) on Brussels Sprouts. Entomol Exp Appl 12:118–124. https://doi.org/10.1111/j.1570-7458.1969.tb02504.x
Prenter J, Pérez-Staples D, Taylor PW (2010) The effects of morphology and substrate diameter on climbing and locomotor performance in male spiders. Funct Ecol 24:400–408. https://doi.org/10.1111/j.1365-2435.2009.01633.x
R core team (2019) R: a language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria
Sabelis MW (1992) Predatory arthropods. In: Crawley MJ (ed) Natural enemies. John Wiley & Sons Ltd, Amsterdam, pp 225–264
Schmidt MH, Lauer A, Purtauf T et al (2003) Relative importance of predators and parasitoids for cereal aphid control. Proc R Soc B 270:1905–1909. https://doi.org/10.1098/rspb.2003.2469
Sigsgaard L (2002) A survey of aphids and aphid parasitoids in cereal fields in Denmark, and the parasitoids’ role in biological control. J Appl Entomol 126:101–107. https://doi.org/10.1046/j.1439-0418.2002.00611.x
Snyder WE, Ives AR (2001) Generalist predators disrupt biological control by a specialist parasitoid. Ecology 82:705–716. https://doi.org/10.1890/0012-9658(2001)082[0705:GPDBCB]2.0.CO;2
Statistiska Centralbyrån Spring barley and oats, yield per hectare. http://www.scb.se/en/finding-statistics/statistics-by-subject-area/agriculture-forestry-and-fishery/agricultural-production/production-of-cereals-dried-pulses-and-oil-seeds/pong/tables-and-graphs/spring-barley-and-oats-yield-per-hectare/. Accessed from 25 Apr 2021
Staudacher K, Jonsson M, Traugott M (2016) Diagnostic PCR assays to unravel food web interactions in cereal crops with focus on biological control of aphids. J Pest Sci 89:281–293. https://doi.org/10.1007/s10340-015-0685-8
Stiling P, Cornelissen T (2005) What makes a successful biocontrol agent? A meta-analysis of biological control agent performance. Biol Control 34:236–246. https://doi.org/10.1016/j.biocontrol.2005.02.017
Symondson WOC, Glen DM, Ives AR et al (2002a) Dynamics of the relationship between a generalist predator and slugs over five years. Ecology 83:137–147. https://doi.org/10.1890/0012-9658(2002)083[0137:DOTRBA]2.0.CO;2
Symondson WOC, Sunderland KD, Greenstone MH (2002b) Can generalist predators be effective biocontrol Agents? Annu Rev Entomol 47:561–594
Tamburini G, De Simone S, Sigura M et al (2016) Conservation tillage mitigates the negative effect of landscape simplification on biological control. J Appl Ecol 53:233–241. https://doi.org/10.1111/1365-2664.12544
Traugott M, Bell JR, Raso L et al (2012) Generalist predators disrupt parasitoid aphid control by direct and coincidental intraguild predation. B Entomol Res 102:239–247. https://doi.org/10.1017/S0007485311000551
Tscharntke T, Bommarco R, Clough Y et al (2007) Conservation biological control and enemy diversity on a landscape scale. Biol Control 43:294–309. https://doi.org/10.1016/j.biocontrol.2007.08.006
Tschumi M, Albrecht M, Bärtschi C et al (2016) Perennial, species-rich wildflower strips enhance pest control and crop yield. Agric Ecosyst Environ 220:97–103. https://doi.org/10.1016/j.agee.2016.01.001
Winder L (1990) Predation of the cereal aphid Sitobion avenae by polyphagous predators on the ground. Ecol Entomol 15:105–110. https://doi.org/10.1111/j.1365-2311.1990.tb00789.x
Wood SA, Karp DS, DeClerck F et al (2015) Functional traits in agriculture: agrobiodiversity and ecosystem services. Trends Ecol Evol 30:531–539. https://doi.org/10.1016/j.tree.2015.06.013
Zadoks JC (1985) On the conceptual basis of crop loss assessment: the threshold theory. Annu Rev Phytopathol 23:455–473. https://doi.org/10.1146/annurev.py.23.090185.002323
Acknowledgements
We thank the team at the “UE 0787 Domaine Expérimental de la Motte” in Le Rheu and the team at the experimental station “Terre d’Essais” in Pleumeur-Gautier for supplying and managing the fields used in our experiments. We also thank Vianney Estorgues for helpful information about the agronomy of brassicaceous vegetables. We thank Dr. Leigh Gebbie for the English revision of this manuscript. Finally, we thank the two anonymous reviewers for the detailed comments and suggestions that greatly improved the manuscript.
Funding
This work was supported by the Région Bretagne (Grant n° 0461/ COH14000/00001042) and the Chaire AEI (Grant n° 2016 0190).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Consent for publication
All authors gave written informed consent for publication.
Ethics approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Communicated by Mattias Jonsson.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mesmin, X., Maret, M., Vincent, M. et al. Biological control at work: demonstrating the complementary effects of natural enemies on two contrasting pests and the damage they cause. J Pest Sci 95, 653–667 (2022). https://doi.org/10.1007/s10340-021-01426-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-021-01426-8