Abstract
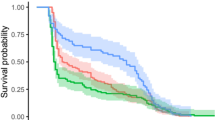
Many arthropod predators and parasitoids exhibit either stage-specific or lifetime omnivory, in that they include extra-floral nectar, floral nectar, honeydew or pollen in their immature and/or adult diet. Access to these plant-derived foods can enhance pest suppression by increasing both the individual fitness and local density of natural enemies. Commercial products such as Amino-Feed®, Envirofeast®, and Pred-Feed® can be applied to crops to act as artificial-plant-derived foods. In laboratory and glasshouse experiments we examined the influence of carbohydrate and protein rich Amino-Feed UV® or Amino-Feed, respectively, on the fitness of a predatory nabid bug Nabis kinbergii Reuter (Hemiptera: Nabidae) and bollworm pupal parasitoid Ichneumon promissorius (Erichson) (Hymenoptera: Ichneumonidae). Under the chosen conditions, the provision of either wet or dry residues of Amino-Feed UV had no discernable effect on immediate or longer-term survival and immature development times of N. kinbergii. In contrast, the provision of honey, Amino-Feed plus extrafloral nectar, and extrafloral nectar alone had a marked effect on the longevity of I. promissorius, indicating that they were limited by at least carbohydrates as an energy source, but probably not protein. Compared with a water only diet, the provision of Amino-Feed plus extrafloral nectar increased the longevity of males and females of I. promissorius by 3.0- and 2.4-fold, respectively. Not only did female parasitoids live longer when provided food, but the total number of eggs laid and timing of deposition was affected by diet under the chosen conditions. Notably, females in the water and honey treatments deposited greater numbers of eggs earlier in the trial, but this trend was unable to be sustained over their lifetime. Egg numbers in these treatments subsequently fell below the levels achieved by females in the Amino-Feed plus extrafloral nectar and cotton extrafloral nectar only treatments. Furthermore, there were times when the inclusion of the Amino-Feed was beneficial compared with cotton extrafloral nectar only. Artificial food supplements and plant-derived foods are worthy of further investigation because they have potential to improve the ecosystem service of biological pest control in targeted agroecosystems by providing natural enemies with an alternative source of nutrition, particularly during periods of prey/host scarcity.



Similar content being viewed by others
References
Baker HG, Baker I (1983) A brief historical review of the chemistry of floral nectar. In: Bentley B, Elias T (eds) The biology of nectaries. Columbia University Press, New York, pp 126–152
Bartlett BR (1962) The ingestion of dry sugars by adult entomophagous insects and the use of this feeding habit for measuring the moisture needs of parasites. J Econ Entomol 55:749–753
Ben Saad AA, Bishop GW (1976) Effect of artificial honeydews on insect communities in potato fields. Environ Entomol 5:453–457
Coll M, Guershon M (2002) Omnivory in terrestrial arthropods: mixing plant and prey diets. Ann Rev Entomol 47:267–297
Eubanks MD, Denno RF (2000) Healthy food versus fast food: the effects of prey quality and mobility on prey selection by a generalist predator and indirect interactions among prey species. Ecol Entomol 25:140–146
Evans EW (2000) Egg production in response to combined alternative foods by the predator Coccinella transversalis. Entomol Exp Appl 94:141–147
Fink U, Völkl W (1995) The effect of abiotic factors on foraging and oviposition success of the aphid parasitoid, Aphidius rosae. Oecologia 103:371–378
Georgiev G (2005) Bioecological characteristics of Bracon intercessor Nees (Hymenoptera: Braconidae) as a parasitoid of the poplar clearwing moth, Paranthrene tabaniformis (Rott.) (Lepidoptera: Sesiidae) in Bulgaria. J Pest Sci 78:161–165
Godfray HCJ (1994) Parasitoids: behavioral and evolutionary ecology. Princeton University Press, Princeton
Grant JF, Shepard M (1985) Techniques for evaluating predators for control of insect pests. J Agric Entomol 2:99–116
Gurr GM, Wratten SD (1999) ‘Integrated biological control’: a proposal for enhancing success in biological control. Int J Pest Manage 45:81–84
Hagen KS (1986) Ecosystem analysis: plant cultivars (HPR), entomophagous species and food supplements. In: Boethel DJ, Eikenbary RD (eds) Interactions of plant resistance and parasitoids and predators of insects. Horwood, Chichester, pp 151–197
Heimpel GE, Jervis MA (2005) Does floral nectar improve biological control by parasitoids? In: Wäckers FL, van Rijn PCJ, Bruin J (eds) Plant-provided food for carnivorous insects: a protective mutualism and its applications. Cambridge University Press, Cambridge, pp 267–304
Heimpel GE, Rosenheim JA (1998) Egg limitation in parasitoids: a review of the evidence and a case study. Biol Control 11:160–168
Heimpel GE, Rosenheim JA, Mangel M (1997) Predation on adult Aphytis parasitoids in the field. Oecologia 110:346–352
Hoferer S, Wäckers FL, Dorn S (2000) Measuring CO2 respiration rates in the parasitoid Cotesia glomerata. Mitteilungen der Deutschen Gesellschaft für allgemeine und angewandte Entomologie 12:555–558
Jervis MA, Kidd NAC, Fitton MG, Huddleston T, Dawah HA (1993) Flower-visiting by hymenopteran parasitoids. J Nat Hist 2:67–105
Jervis MA, Heimpel GE, Ferns PN, Harvey JA, Kidd NA (2001) Life-history strategies in parasitoid wasps: a comparative analysis of “ovigeny”. J Anim Ecol 70:442–458
Johnson M-L, Pearce S, Wade M, Davies A, Silberbauer L, Gregg P, Zalucki M (2000) Review of beneficials in cotton farming systems. Report to the Australian cotton research and development corporation, Narrabri, NSW, Australia, pp 86
Landis DA, Wratten SD, Gurr GM (2000) Habitat management to conserve natural enemies of arthropod pests in agriculture. Ann Rev Entomol 45:175–202
Lavandero B, Wratten SD, Shishehbor P, Worner S (2005) Enhancing the effectiveness of the parasitoid Diadegma semiclausum (Helen): movement after use of nectar in the field. Biol Control 34:152–158
Lee JC, Heimpel GE (2007) Sugar feeding reduces short-term activity of a parasitoid wasp. Physiol Entomol 32:99–103
Leius K (1961) Influence of food on fecundity and longevity of adults of Itoplectis conquisitor (Say) (Hymenoptera: Ichneumonidae). Can Entomol 93:771–780
McCullagh P, Nelder JA (1989) Generalized linear models. Chapman & Hall, London
McEwen PK, Jervis MA, Kidd NAC (1996) The influence of an artificial food supplement on larval and adult performance in the green lacewing Chrysoperla carnea (Stephens). Int J Pest Manage 42:25–27
Mensah R, Singleton A (2002) Use of food sprays in cotton systems: what do we know? In: Proceedings of the 11th Australian cotton conference, Brisbane, QLD, 13–15 August, pp 359–375
Mensah RK, Singleton A (2003) Optimum timing and placement of a supplementary food spray Envirofeast® for the establishment of predatory insects of Helicoverpa spp. in cotton systems in Australia. Int J Pest Manage 49:163–168
Polis GA, Strong DR (1996) Food web complexity and community dynamics. Am Nat 147:813–846
Pontin DR, Wade MR, Kehrli P, Wratten SD (2006) Attractiveness of single and multiple species flower patches to beneficial arthropods in agroecosystems. Ann Appl Biol 148:39–47
Šidák Z (1967) Rectangular confidence regions for the means of multivariate normal distributions. J Am Stat Assoc 62:626–633
Steppuhn A, Wäckers FL (2004) HPLC sugar analysis reveals the nutritional state and the feeding history of parasitoids. Funct Ecol 18:812–819
Wäckers FL (2000) Do oligosaccharides reduce the suitability of honeydew for predators and parasitoids? A further facet to the function of insect-synthesized honeydew sugars. Oikos 90:197–201
Wäckers FL, van Rijn PCJ (2005) Food for protection: an introduction. In: Wäckers FL, van Rijn PCJ, Bruin J (eds) Plant-provided food for carnivorous insects: a protective mutualism and its applications. Cambridge University Press, Cambridge, pp 1–14
Wade MR, Wratten SD (2007) Excised or intact inflorescences? Methodological effects on parasitoid wasp longevity. Biol Control 40:347–354
Wade MR, Zalucki MP, Wratten SD, Robinson KA (2008). Conservation biological control of arthropods using artificial food sprays: Current status and future challenges. Biol Control doi:10.1016/j.biocontrol.2007.10.024(in press)
Winkler K, Wäckers FL, Bukovinszkine-Kiss G, van Lenteren JC (2006) Nectar resources are vital for Diadegma semiclausum fecundity under field conditions. Basic Appl Ecol 7:133–140
Zehnder G, Gurr GM, Kuehne S, Wade MR, Wratten SD, Wyss E (2007) Arthropod pest management in organic crops. Ann Rev Entomol 52:57–80
Acknowledgments
We are grateful to Sue Maclean for providing the bollworm materials, Erin Conze of Agrichem for supplying the Amino-Feed UV, and Allan Lisle and Kerry Bell for statistical advice. Thanks to Nancy Schellhorn, Felix Wäckers and two anonymous reviewers for their valuable comments on an earlier version of the manuscript. Principal funding was provided by the Australian Cotton Research and Development Corporation (postgraduate research project UQ29C) and an in-kind contribution from the Australian Grains Research and Development Corporation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by M. Traugott.
Rights and permissions
About this article
Cite this article
Wade, M.R., Hopkinson, J.E. & Zalucki, M.P. Influence of food supplementation on the fitness of two biological control agents: a predatory nabid bug and a bollworm pupal parasitoid. J Pest Sci 81, 99–107 (2008). https://doi.org/10.1007/s10340-007-0191-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-007-0191-8