Abstract
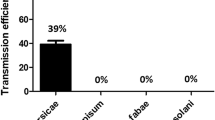
The aphid transmissibility and within-plant movement of two PVY isolates belonging to PVYO and PVYN strains were studied. Potato plants were infected by Myzus persicae either 1 week after emergence or 30 or 45 days after emergence. Infected leaves, petioles of infected leaves, and different segments of the stems of plants infected 1 week after emergence were ELISA-tested 7, 11 and 14 days after infection. Segments of the plants infected 30 or 45 days after emergence were ELISA tested 30 days after infection. Normalised ELISA values were used for statistical analyses. Virus infection of progeny tubers was assessed by post-harvest inspection. The ability of 15 aphid species to transmit the virus isolates was also examined. There was no difference in the aphid transmissibility of the two isolates 7 or 11 days after exposure to M. persicae. However, normalised ELISA values were significantly affected by both isolate and sampling date 11 and 14 days after M. persicae transmission. The level of infection of different stem segments was significantly affected by both isolate and growth stage of the plant at infection. The proportions of infected leaves, stem segments and progeny tubers were all significantly higher for PVYN than for PVYO. PVYN was more effectively transmitted by a number of different aphid species than was PVYO. Newly described vectors of PVYN include Schizaphis graminum, Aphis fabae cirsiacanthoides, Aphis spiraecola, Myzus ligustri, Aphis fabae, Aphis spiraephaga, Myzus cerasi, Macrosiphum rosae, Diuraphis noxia, Aphis pomi and Rhopalosiphum padi, while those of PVYO include Aphis fabae cirsiacanthoides, Myzus cerasi and Myzus ligustri.





Similar content being viewed by others
References
Beczner L, Horváth J, Romhányi I, Förster H (1984) Studies on the etiology of tuber necrotic ringspot disease in potato. Potato Res 27:339–352
Beemster ABR (1976) Translocation of the potato YN and YO in some potato varieties. Potato Res 19:169–172
Blanco-Urgoit B, Tribodet M, Leclere S, Ponz F, Perez de San Roman C, Legorburu FJ, Kerlan C (1998) Characterisation of potato virus Y (PVY) isolates from seed potato batches. Situation of the NTN, Wilga and Z isolates. Eur J Plant Pathol 104:811–819
Boonham N, Walsh K, Preston S, North J, Smith P, Barker I (2002) The detection of tuber necrotic isolates of potato virus Y, and the accurate discrimination of PVYC strains using RT-PCR. J Virol Meth 102:103–112
Chrzanowska M (1991) New isolates of necrotic strain of potato virus Y (PVYN) found recently in Poland. Potato Res 34:179–182
Clark MF, Adams AA (1977) Characteristics of the microplate method of enzyme/linked immunosorbent assay for the detection of plant viruses. J Gen Virol 34:475–483
Carrington JC, Kasschau DK, Mahajan SK, Schaad MC (1996) Cell-to-cell and long-distance transport of viruses in plants. Plant Cell 8:1669–1681
Cronin S, Verchot J, Haldeman-Cahill R, Schaad MC, Carrington JC (1995) Long-distance movement factor: a transport function of the potyvirus helper component proteinase. Plant Cell 7:549–559
De Bokx JA, Piron PGM (1977) Effect of temperature on symptom expression and relative virus concentration in potato plants infected with potato virus YN and YO. Potato Res 20:207–213
Draper MD, Pasche JS, Gudmestad NC (2002) Factors influencing PVY development and disease expression in three potato cultivars. Am J Potato Res 79:155–165
Gibson RW (1991) The development of mature plant resistance in four potato cultivars against aphid-inoculated potato virus YO and YN in four potato cultivars. Potato Res 34:205–210
Glais L, Tribodet M, Kerlan C (2002) Genomic variability in potato potyvirus Y (PVY): evidence that PVYNW and PVYNTN variants are single to multiple recombinants between PVYO and PVYN isolates. Arch Virol 147:363–378
Knoblauch M, van Bel AJE (1998) Sieve tubes in action. Plant Cell 10:35–50
Lalonde S, Boles E, Hellmann H, Barker L, Patrick JW, Frommer WB (1999) The dual function of sugar carriers: transport and sugar sensing. Plant Cell 11:707–726
Latorre BA, Flores V (1985) Strain identification and cross-protection of potato virus-Y affecting tobacco in Chile. Plant Dis 69:930–932
Maule AJ (1991) Virus movement in infected plants. Crit Rev Plant Sci 9:457–473
McDonald JG, Kristjansson GT (1993) Properties of strains of potato virus YN in North America. Plant Dis 77:87–89
Oparka KJ, Turgeon R (1999) Sieve elements and companion cells-traffic control centers of the phloem. Plant Cell 11:739–750
Robert Y, Woodford JAT, Ducray-Bourdin DG (2000) Some epidemiological approaches to the control of aphid-borne virus diseases in seed potato crops in northern Europe. Virus Res 71:33–47
Sáenz P, Salvador B, Simón-Mateo C, Kasschau KD, Carrington JC (2002) Host-specific involvement of the HC protein in the long-distance movemnent of potyviruses. J Virol 76:1922–1931
Sigvald R (1985) Mature-plant resistance of potato plants against potato virus YO (PVYO). Potato Res 28:135–143
Silva MS, Wellink J, Goldbach RW, van Lent JMV (2002) Phloem loading and unloading of Cowpea mosaic virus in Vigna unguiculata. J Gen Virol 83:1493–1504
Szirmai J (1958) A burgonya Y-vírusának érbarnulást okozó törzse dohánykulturákban. Növénytermelés 7:341–350
Valkonen JPT, Rokka VM (1988) Combination and expression of two virus resistance mechanisms in interspecific somatic hybrids of potato. Plant Sci 131:85–94
Viola R, Roberts AG, Haupt S, Gazzani S, Hancock RD (2001) Tuberization in potato involves a switch from apoplastic to symplatic phloem unloading. Plant Cell 13:385–398
Walsh K, North J, Barker I, Boonham N (2001) Detection of different strains of potato virus Y and their mixed infections using competitive fluorescent RT-PCR. J Virol Meth 91:167–173
Weidemann HL (1988) Importance and control of potato virus YN (PVYN) in seed potato production. Potato Res 31:85–94
Wu X, Weige D, Wigge PA (2002) Signaling in plants by intercellular RNA and protein movement. Genes Dev 16:151–158
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Basky, Z., Almási, A. Differences in aphid transmissibility and translocation between PVYN and PVYO isolates. J Pest Sci 78, 67–75 (2005). https://doi.org/10.1007/s10340-004-0070-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-004-0070-5