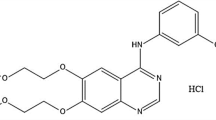
Abstract
A Quality by Design (QbD) systematic and analytical approach was used to develop a novel and sensitive Lenvatinib stability-indicating method. The ICH Q1A(R2) and Q3 guidelines were implemented to determine Lenvatinib degradation behavior under various environmental conditions. The QbD approach implementation has screening and optimization stages.The Placket–Burman design was used to assess primary parameters, and Response Surface Design (RSD) to optimize critical factors. The drug degradation was examined under different degradation conditions, including acidic, basic, oxidative, neutral, thermal, and photolytic conditions. Separation was achieved using a Shimadzu® C18 column (250 mm × 4.6 mm, particle size 5 µ) with the mobile phase consisted of Acetonitrile: 10 mM ammonium acetate at pH 3.5 (39:61, v/v) at a flow rate 0.8 mL/min. The run time was 20 min and the wavelength used was 245 nm. The drug found sensitive toward acid and base hydrolysis, resulting in the generation of five degradation products. These products were successfully identified using the optimized LC–MS compatible analytical method. The optimized method was found to be sensitive, reproducible, specific, and robust, with a linearity range of 10 to 60 mg/mL and a correlation coefficient (R2 = 0.9993). The greenness score of the analytical method was calculated, revealing that the developed method is environmentally friendly.
Graphical abstract








Similar content being viewed by others
Data Availability
All the data generated or analyzed during this study are included in this published article and as in form of supplementary data.
Abbreviations
- nm:
-
Nanometer
- ppm:
-
Parts per million
- °C:
-
Degree Celsius
- mL:
-
Milliliter
- µg:
-
Micron gram
- SIAM:
-
Stability-indicating assay method
- mg:
-
Milligram
- Conc.:
-
Concentration
- HPLC:
-
High-Performance Liquid Chromatography
- LC/MS–MS:
-
Liquid Chromatography–Mass Spectrometry
- HCl:
-
Hydrochloric acid
- NaOH:
-
Sodium Hydroxide
- MeOH:
-
Methanol
- ACN:
-
Acetonitrile
- pH:
-
Potential of Hydrogen
- Hrs:
-
Hours
- min:
-
Minute
- DOE:
-
Design of Experiment
- QbD:
-
Quality by Design
- H2O2 :
-
Hydrogen peroxide
- PBD:
-
Plackett–Burman Design
- RSD:
-
Response Surface Design
- ANOVA:
-
Analysis of variance
- LOD:
-
Limit of detection
- LOQ:
-
Limit of quantification
- ICH:
-
International Council for Harmonization
- DPs:
-
Degradation products
- CQAs:
-
Critical quality attributes
- CAAs:
-
Critical analytical attributes
- AMGS:
-
Analytical method greenness score
- CMAs:
-
Critical material attributes
- AGREE:
-
Appraisal of Guidelines, Research and Evaluation
- GAC:
-
Green analytical chemistry
- GAPI:
-
Green analytical process index
References
Zigart N, Casa Z (2020) Development of a stability-indicating analytical method for determination of venetoclax using AQbD principles. ACS Omega 5:17726
International council for Harmonization (2003) ICH Topic Q 1 A (R2) Stability Testing of new Drug Substances and Products Step 5 Note for guidance on stability testing: stability testing of new drug substances and products
Sehrawat R, Maithani M, Singh R (2010) Regulatory aspects in development of stability-indicating methods: a review. Chromatographia 72:1–6. https://doi.org/10.1365/S10337-010-1612-Z
Kalal DJ, Redasani VK (2022) Stability-indicating RP-HPLC method development and validation for estimation of Mupirocin calcium in bulk and in pharmaceutical formulation. Futur J Pharm Sci. https://doi.org/10.1186/S43094-022-00412-W
Bhavna BS, Ojha A (2022) International Council for Harmonisation (ICH) guidelines. Regulatory Affairs in the Pharmaceutical Industry. Elsevier, Amsterdam, pp 47–74
Cui Y, Li Y, Fan L et al (2021) UPLC-MS/MS method for the determination of Lenvatinib in rat plasma and its application to drug-drug interaction studies. J Pharm Biomed Anal 206:114360. https://doi.org/10.1016/J.JPBA.2021.114360
Tahara M, Kiyota N, Yamazaki T et al (2017) Lenvatinib for anaplastic thyroid cancer. Front Oncol. https://doi.org/10.3389/fonc.2017.00025
Chellapan S, Datta D, Kumar S, Uslu H (2022) Statistical modeling and optimization of itaconic acid reactive extraction using response surface methodology (RSM) and artificial neural network (ANN). Chem Data Collect 37:47–55. https://doi.org/10.1016/j.cdc.2021.100806
Al-Salama ZT, Syed YY, Scott LJ (2019) Lenvatinib: a review in hepatocellular carcinoma. Drugs 79:665–674. https://doi.org/10.1007/S40265-019-01116-X
Blessy M, Patel RD, Prajapati PN, Agrawal YK (2014) Development of forced degradation and stability indicating studies of drugs—a review. J Pharm Anal 4:159–165. https://doi.org/10.1016/j.jpha.2013.09.003
Motzer RJ, Taylor MH, Evans TRJ et al (2022) Lenvatinib dose, efficacy, and safety in the treatment of multiple malignancies. Expert Rev Anticancer Ther 22:383–400. https://doi.org/10.1080/14737140.2022.2039123
Peterson JJ, Lief K (2009) The ICH Q8 definition of design space: a comparison of the overlapping means and the Bayesian predictive approaches. AIChE Annu Meet Conf Proc. https://doi.org/10.1198/SBR.2009.08065
Park G, Kim MK, Go SH et al (2022) Analytical quality by design (AQbD) approach to the development of analytical procedures for medicinal plants. Plants 11:2960
Del Vecchio RJ (2007) Design of experiments. Handbook of vinyl formulating, 2nd edn. Elsevier, Amsterdam, pp 515–527
Muchakayala SK, Katari NK, Saripella KK et al (2022) Implementation of analytical quality by design and green chemistry principles to develop an ultra-high performance liquid chromatography method for the determination of Fluocinolone Acetonide impurities from its drug substance and topical oil formulations. J Chromatogr A 1679:463380. https://doi.org/10.1016/j.chroma.2022.463380
Beydoun A, Dupont S, Zhou D et al (2020) Current role of carbamazepine and oxcarbazepine in the management of epilepsy. Seizure Eur J Epilepsy 83:251–263. https://doi.org/10.1016/j.seizure.2020.10.018
Sajid M, Talanta JP-W (2022) Green analytical chemistry metrics: a review. Talanta. https://doi.org/10.1016/j.talanta.2021.123046
Lopez-Lorente A, Chemistry FP-P (2022) The ten principles of green sample preparation. TrAC Trends Anal Chem 148:116530
Becker J, Manske C, Chemistry SRG and S (2022) Undefined Green chemistry and sustainability metrics in the pharmaceutical manufacturing sector. Elsevier, Amsterdam
Dhage SD, Shisodiya KK (2013) Application of green chemistry in susstainable development. Int Res J Pharm 4:1–4. https://doi.org/10.7897/2230-8407.04701
El-Sayed HM, Abdellatef HE, Hendawy HAM et al (2023) DoE-enhanced development and validation of eco-friendly RP-HPLC method for analysis of safinamide and its precursor impurity: QbD approach. Microchem J 190:108730. https://doi.org/10.1016/j.microc.2023.108730
Muchakayala SK, Katari NK, Saripella KK et al (2022) AQbD based green UPLC method to determine mycophenolate mofetil impurities and Identification of degradation products by QToF LCMS. Sci Rep 121(12):1–18. https://doi.org/10.1038/s41598-022-22998-0
Soliman SS, Sedik GA, Elghobashy MR et al (2022) Greenness assessment profile of a QbD screen-printed sensor for real-time monitoring of sodium valproate. Microchem J 182:107859. https://doi.org/10.1016/j.microc.2022.107859
Mohamed D, Fouad MM (2020) Application of NEMI, analytical eco-scale and GAPI tools for greenness assessment of three developed chromatographic methods for quantification of sulfadiazine and trimethoprim in bovine meat and chicken muscles: comparison to greenness profile of reporte. Microchem J. https://doi.org/10.1016/j.microc.2020.104873
Sheldon R, Bode M, Chemistry SAG, S, 2022 U (2022) Metrics of green chemistry: waste minimization. Curr Opin Green Sustain Chem. https://doi.org/10.1016/j.cogsc.2021.100569
Ferreira S, Brito T, Santana A et al (2022) Greenness of procedures using NADES in the preparation of vegetal samples: comparison of five green metrics. Talanta Open 6:100131
Bandla J, Ganapaty S (2018) New stability-indicating ultra performance liquid chromatography method development and validation of lenvatinib mesylate in bulk drug and pharmaceutical dosage forms. Asian J Pharm Clin Res 11:140–143. https://doi.org/10.22159/ajpcr.2018.v11i9.26766
Bang PP, Bhatt HG (2023) Development of green RP- and green NP-HPTLC methods for estimation of lenvatinib and comparative evaluation by AGREE. ACS Sustain Chem Eng 11:2249–2263. https://doi.org/10.1021/ACSSUSCHEMENG.2C05767
Sultana M, Res DR (2020) Stability indicating method development and validation for simultaneous estimation of dasatinib and lenvatinib by using uplc in pharmaceutical dosage form. World J Pharm 9:1548–1560. https://doi.org/10.20959/wjpr202014-18875
Patel M, Kothari C (2020) Comprehensive stability-indicating method development of Avanafil Phosphodiesterase type 5 inhibitor using advanced quality-by-design approach. J Anal Sci Technol 11:29. https://doi.org/10.1186/s40543-020-00228-4
Gundecha S, Patel M, Mayur YC (2022) An application of quality by design and analytical greenness assessment approach for the development of erlotinib stability indicating method. Chromatographia 85:575–588. https://doi.org/10.1007/S10337-022-04167-7
Raposo F, Ibelli-Bianco C (2020) Performance parameters for analytical method validation: controversies and discrepancies among numerous guidelines. TrAC Trends Anal Chem 129:115913
Mishra V, Thakur S, Patil A, Shukla A (2018) Quality by design (QbD) approaches in current pharmaceutical set-up. Expert Opin Drug Deliv 15:737–758. https://doi.org/10.1080/17425247.2018.1504768
Patel KY, Dedania ZR, Dedania RR, Patel U (2021) QbD approach to HPLC method development and validation of ceftriaxone sodium. Futur J Pharm Sci. https://doi.org/10.1186/S43094-021-00286-4
Zagalo DM, Silva BMA, Silva C et al (2022) A quality by design (QbD) approach in pharmaceutical development of lipid-based nanosystems: a systematic review. J Drug Deliv Sci Technol. https://doi.org/10.1016/j.jddst.2022.103207
Palandurkar K, Bhandre R, Boddu SHS et al (2023) Quality risk assessment and DoE—practiced validated stability-indicating chromatographic method for quantification of Rivaroxaban in bulk and tablet dosage form. Acta Chromatogr 35:10–20. https://doi.org/10.1556/1326.2021.00978
Raska CS, Bennett TS, Goodberlet SA (2010) Risk-based analytical method transfer: application to large multi-product transfers. Anal Chem 82:5932–5936. https://doi.org/10.1021/ac1008892
Deidda R, Orlandini S, Hubert P, Hubert C (2018) Risk-based approach for method development in pharmaceutical quality control context: a critical review. J Pharm Biomed Anal 161:110–121. https://doi.org/10.1016/j.jpba.2018.07.050
Chaudhari SR, Shirkhedkar AA (2020) Application of Plackett-Burman and central composite designs for screening and optimization of factor influencing the chromatographic conditions of HPTLC method for quantification of efonidipine hydrochloride. J Anal Sci Technol. https://doi.org/10.1186/S40543-020-00246-2
Valmorida JS, Castillo-Israel KAT (2018) Application of Plackett-Burman experimental design in the development of muffin using adlay flour. IOP Conf Ser Earth Environ Sci. https://doi.org/10.1088/1755-1315/102/1/012081
Januardi WE (2020) A review of response surface methodology approach in supply chain management. ACM Int Conf Proc Ser. https://doi.org/10.1145/3400934.3400993
Myers RH, Montgomery DC, Geoffrey Vining G et al (2004) Response surface methodology: a retrospective and literature survey. J Qual Technol 36:53–78
de Oliveira LG, de Paiva AP, Balestrassi PP et al (2019) Response surface methodology for advanced manufacturing technology optimization: theoretical fundamentals, practical guidelines, and survey literature review. Int J Adv Manuf Technol 104:1785–1837. https://doi.org/10.1007/S00170-019-03809-9
Mark H (2003) Application of an improved procedure for testing the linearity of analytical methods to pharmaceutical analysis. J Pharm Biomed Anal 33:7–20. https://doi.org/10.1016/S0731-7085(03)00346-7
Mollica JA, Ahuja S, Cohen J (1978) Stability of pharmaceuticals. J Pharm Sci 67:443–465. https://doi.org/10.1002/jps.2600670405
Moema D, Makwakwa T, BG-J of F (2023) undefined by high pressure liquid chromatography: Greenness assessment using national environmental methods index label (NEMI), green analytical procedure index (GAPI). Elsevier, Amsterdam
Acknowledgements
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
All authors read and approved the final version of the manuscript for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tiwari, P., Patel, M. Advancing Quality and Environmental Responsibility: A Stability-Indicating LC Method Development for Lenvatinib Through QbD and Green Chemistry. Chromatographia 86, 579–593 (2023). https://doi.org/10.1007/s10337-023-04271-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-023-04271-2