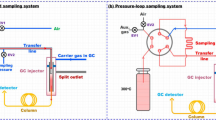
Abstract
Static headspace analysis is of value for the detection of volatile compounds present in biological and environmental samples in trace amounts. However, trace compound detection has typically relied on splitless inlet mode which generates wide non-Gaussian peaks when using ordinary methylpolysiloxane columns without resort to specialty columns such as those using a polyethylene glycol stationary phase for alcohols. Solvent vent settings were investigated as an alternative approach that would enable large volume injections necessary for high sensitivity. This inlet configuration was supported by tandem quadrupole mass spectrometric compound detection which provided low backgrounds and limited extraneous compound interference. The method was studied with diverse compounds of toxicological significance, including phosphine, dioxane, dichloromethane, ethyl acetate and simple alcohols such as methanol, ethanol, and isopropanol. Despite differences in initial temperature settings for inlet and oven required by each of these analytes, Solvent vent mode in each case introduced significant advantages in terms of chromatographic peak shape, separation, and sensitivity. Most significantly, the method enabled separation of methanol, ethanol, and isopropanol in contrast to Splitless mode which resulted in significant compound overlap. The effects of changes to crucial gas chromatographic and Solvent Vent parameters were investigated including injection volume, helium carrier gas flow rate, applicability to quantitation, injection needle dwell time, needle residence time, carryover, headspace incubation temperature, oven and inlet temperatures and timing of GC oven temperature ramping. A proof of principle was developed involving analyses of complex fire accelerants gasoline and carburetor cleaner where ~ 30 peaks could be separated from one another by application of the espoused principles.







Similar content being viewed by others
Availability of Data and Material
Additional information pertinent to this work is included as Supplementary Material.
References
Kolb B, Ettre LS (2006) Static headspace-gas chromatography: theory and practice, 2nd edn. Wiley-Interscience, Hoboken, N.J.
Snow NH, Slack GC (2002) Head-space analysis in modern gas chromatography. Trends Anal Chem 21:608–617. https://doi.org/10.1016/S0165-9936(02)00802-6
Powers J, Mohal B, Brannon S (2014) GC headspace applications for VOC: the challenges of obtaining accurate measurements with decreasing VOC levels. Coat Tech 11:38–45
Grove D, Miller-Atkins G, Melillo C, Rieder F, Kurada S, Rotroff DM, Tonelli AR, Dweik RA (2020) Comparison of volatile organic compound profiles in exhaled breath versus plasma headspace in different diseases. J Breath Res 14:036003
Davoli E, Bastone A, Bianchi G, Salmona M, Diomede L (2017) A simple headspace gas chromatography/mass spectrometry method for the quantitative determination of the release of the antioxidants butylated hydroxyanisole and butylated hydroxytoluene from chewing gum. Rapid Commun Mass Spectrom 31:859–864
Ghosh C, Singh V, Grandy J, Pawliszyn J (2020) Development and validation of a headspace needle-trap method for rapid quantitative estimation of butylated hydroxytoluene from cosmetics by hand-portable GC-MS. RSC Adv 10:6671–6677
Fakhari AR, Hasheminasab KS, Baghdadi M, Khakpour A (2012) A simple and rapid method based on direct transfer of headspace vapor into the GC injector: application for determination of BTEX compounds in water and wastewater samples. Anal Methods 4:1996–2001
Lim SK, Shin HS, Yoon KS, Kwack SJ, Um YM, Hyeon JH, Kwak HM, Kim JY, Kim TY, Kim YJ, Roh TH, Lim DS, Shin MK, Choi SM, Kim HS, Lee B-M (2014) Risk assessment of volatile organic compounds benzene, toluene, ethylbenzene, and xylene (BTEX) in consumer products. J Toxicol Environ Health Part A 77:1502–1521
Fang H, Chen J, Tian Y, Liu Y, Li H, Cheng H (2020) Chemometrics characterization of volatile changes in processed bayberry juice versus intact fruit during storage by headspace solid-phase micro-extraction combined with GC-MS. J Food Process Preserv 44:e14444
Del Barrio M-A, Hu J, Zhou P, Cauchon N (2006) Simultaneous determination of formic acid and formaldehyde in pharmaceutical excipients using headspace GC/MS. J Pharm Biomed Anal 41:738–743
Pena J, Laespada MEF, Pinto CG, Pavon JLP (2021) Multiple headspace sampling coupled to a programmed temperature vaporizer—gas chromatograph-mass spectrometer for the determination of polycyclic aromatic hydrocarbons in water and saliva. Microchem J 161(105822):1–8
Watanabe K, Hasegawa K, Yamagishi I, Nozawa H, Suzuki O (2009) Simple analysis of naphthalene in human whole blood and urine by headspace capillary gas chromatography with large-volume injection. Forensic Toxicol 27:98–102
Letavernier JF, Aubert M, Ripoche G, Pellerin F (1985) Study of solvent residues of plastic material and coatings in drugs using head-space gas-chromatography. Ann Pharm Fr 43:117–122
Cundy KV, Willard KE, Valeri LJ, Shanholtzer CJ, Singh J, Peterson LR (1991) Comparison of traditional gas chromatography (GC), headspace GC, and the microbial identification library GC system for the identification of Clostridium difficile. J Clin Microbiol 29:260–263
Jerkovic I, Marijanovic Z (2009) A short review of headspace extraction and ultrasonic solvent extraction for honey volatiles fingerprinting. Croat J Food Sci Technol 1:28–34
Watanabe K, Fujita H, Hasegawa K, Gonmori K, Suzuki O (2011) GC/MS with post-column switching for large volume injection of headspace samples: sensitive determination of volatile organic compounds in human whole blood and urine. Anal Chem 83:1475–1479
Brachet A, de Saint Laumer J-Y, Chaintreau A (2005) Automated multiple headspace extraction procedure: adsorption modeling and determination of air-to-water partition coefficients. Anal Chem 77:3053–3059
Maruti A, Duran-Guerrero E, Barroso CG, Castro R (2018) Optimization of a multiple headspace sorptive extraction method coupled to gas chromatography-mass spectrometry for the determination of volatile compounds in macroalgae. J Chromatogr A 1551:41–51
Sanchez-Garcia F, Mirzayeva A, Roldan A, Castro R, Palacios V, Garcia-Barroso C, Duran-Guerrero E (2019) Evolution of volatile compounds and sensory characteristics of edible green seaweed (Ulva rigida) during storage at different temperatures. J Sci Food Agric 99:5475–5482
Kostic M, Stanojevic J, Tacic A, Gligorijevic N, Nikolic L, Nikolic V, Igic M, Vasic MB (2020) Determination of residual monomer content in dental acrylic polymers and effect after tissues implantation. Biotechnol Biotechnol Equip 34:254–263
Gemme S, Santoro M (2016) Static headspace analysis of residual solvents in flexible packaging and quantitation with multiple headspace extraction following EN 13628-1: 2002. Thermo Scientific Application Note 10396. https://assets.thermofisher.com/TFS-Assets/CMD/Application-Notes/AN-10396-GC-Flexible-Packaging-Solvents-Headspace-AN10396-EN.pdf. Accessed 4/7/21.
Schuberth J (1996) A full evaporation headspace technique with capillary GC and ITD: a means for quantitating volatile organic compounds in biological samples. J Chromatogr Sci 34:314–319. https://doi.org/10.1093/chromsci/34.7.314
Shapira A, Berman P, Futoran K, Guberman O, Meiri D (2019) Tandem mass spectrometric quantification of 93 terpenoids in cannabis using static headspace injections. Anal Chem 91:11425–11432
Dei Cas M, Arnoldi S, Monguzzi L, Casagni E, Morano C, Vieira de Manincor E, Bolchi C, Pallavicini M, Gambaro V, Roda G (2021) Characterization of chemotype-dependent terpenoids profile in cannabis by headspace gas-chromatography coupled to time-of-flight mass spectrometry. J Pharm Biomed Anal 203:114180
LabCompare (2013) Blood-alcohol analysis by HS-GC using 0.15-i.d. columns. https://www.labcompare.com/10-Featured-Articles/146904-Blood-Alcohol-Analysis-by-HS-GC-Using-0-15-i-d-Columns/. Accessed 4/7/21.
Chun H-J, Poklis JL, Poklis A, Wolf CE (2016) Development and validation of a method for alcohol analysis in brain tissue by headspace gas chromatography with flame ionization detector. J Anal Toxicol 40:653–658
Lara-lbeas I, Rodríguez-Cuevas A, Andrikopoulou C, Person V, Baldas L, Colin S, Le Calvé S (2019) Sub-ppb level detection of BTEX gaseous mixtures with a compact prototype GC equipped with a preconcentration unit. Micromach (Basel) 10:1–13
Dewulf J, Van Langenhove H, Wittmann G (2002) Analysis of volatile organic compounds using gas chromatography. TrAC Trends Anal Chem 21s:637–646
Rahman MM, El-Aty AMA, Choi J-H, Shin H-C, Shin SC, Shim J-H (2015) Basic overview on gas chromatography columns. In: Anderson JL, Berthod A, Estévez VP, Stalcup AM (eds) Analytical separation science, 1st edn. Wiley-VCH Verlag GmbH & Co, KGaA, Weinheim, pp 823–834
Lehner AF, Johnson M, Buchweitz JP (2020) Phosphine detection in veterinary samples using headspace gas chromatography/tandem mass spectrometry with multiple reaction monitoring. Rapid Commun Mass Spectrom 34:e8738
Kitteringham NR, Jenkins RE, Lane CS, Elliott VL, Park BK (2009) Multiple reaction monitoring for quantitative biomarker analysis in proteomics and metabolomics. J Chromatogr B Analyt Technol Biomed Life Sci 877:1229–1239
US Department of Health and Human Services (USDHHS) (2000) Toxicological profile for methylene chloride. https://www.atsdr.cdc.gov/toxprofiles/tp14.pdf (Accessed April 22, 2021).
Association of Official Racing Chemists (2016) AORC guidelines for the minimum criteria for identification by chromatography and mass spectrometry. http://www.aorc-online.org/documents/aorc-ms-criteria-modified-23-aug-16/ (Accessed Dec 15, 2021).
Moosavi S, Ghassabian S (2018) Linearity of calibration curves for analytical methods: a review of criteria for assessment of method reliability. In: Stauffer M (ed) Calibration and validation of analytical methods: a sampling of current approaches. Intechopen, London, pp 109–127
USDHHS (2011) Report on carcinogens, twelfth edition. https://web.archive.org/web/20140425233701/http://ntp.niehs.nih.gov/ntp/roc/twelfth/roc12.pdf (Accessed April 22, 2021).
Rusyn I, Bataller R (2013) Alcohol and toxicity. J Hepatol 59:387–388
Kraut JA, Mullins ME (2018) Toxic alcohols. N Engl J Med 378:270–280
Restek (2000) A technical guide for static headspace analysis using GC. https://d1lqgfmy9cwjff.cloudfront.net/csi/pdf/e/rk67.pdf (Accessed April 23, 2021).
Smith ER, Wojciechowski M (1937) Boiling point—composition diagram of the system dioxane-water. J Res Natl Bur Stand 18:461–465
Agilent (2009) Agilent multimode inlet: large volume injection tutorial. https://www.agilent.com/cs/library/usermanuals/Public/G3510-90020.pdf (Accessed April 23, 2021).
Klee MS (1990) GC inlets—an Introduction. Hewlett-Packard Co, Avondale
Grob K (2001) Split and splitless injection for quantitative gas chromatography: concepts, processes, practical guidelines, sources of error (4th, completely revised edition). Wiley, Hoboken
Gómez-Ruiz JÁ, Cordeiro F, López P, Wenzl T (2009) Optimisation and validation of programmed temperature vaporization (PTV) injection in solvent vent mode for the analysis of the 15 + 1 EU-priority PAHs by GC–MS. Talanta 80:643–650
Piparo M, Flamant L, Jousset G, Cardinael P, Giusti P (2020) Careful investigations of PTV injection parameters for the analysis of vacuum gas oil by high-temperature comprehensive GC x GC. Energy Fuels 34:12010–12017
Pena J, Fernandez Laespada ME, Garcia Pinto C, Perez Pavon JL, Moreno Cordero B (2019) Multiple headspace sampling coupled to a programmed temperature vaporizer to improve sensitivity in headspace-gas chromatography. Determination of aldehydes. J Chromatogr B Anal Technol Biomed Life Sci 1133:121824
Perez Anton A, Ramos AG, del Nogal SM, Pavon JLP, Cordero BM, Pozas APC (2016) Headspace-programmed temperature vaporization-mass spectrometry for the rapid determination of possible volatile biomarkers of lung cancer in urine. Anal Bioanal Chem 408:5239–5246
Sales C, Poma G, Malarvannan G, Portoles T, Beltran J, Covaci A (2017) Simultaneous determination of dechloranes, polybrominated diphenyl ethers and novel brominated flame retardants in food and serum. Anal Bioanal Chem 409:4507–4515
Shimadzu (2022) A comparison study of different capillary columns for analysis of alcohol congeners in alcoholic beverages. https://www.shimadzu.com/an/sites/shimadzu.com.an/files/pim/pim_document_file/applications/application_note/13335/apc116006.pdf (Accessed April 23, 2021).
Pundlik MD, Sitharaman B, Kaur I (2001) Gas chromatographic determination of 1,4-dioxane at low parts-per-million levels in glycols. J Chromatogr Sci 39:73–76
Wahab MF, Armstrong DW, Patel DC (2017) Peak shapes and their measurements: the need and the concept behind total peak shape analysis. LCGC North America 35: 846–853. https://alfresco-static-files.s3.amazonaws.com/alfresco_images/pharma/2019/01/07/383b8ddf-6673-4ecc-b13e-f71a743b3e36/LCGC_NAmerica_December2017.pdf (Accessed May 13, 2021).
Funding
This research did not receive any specific Grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no financial interests in the outcome of this research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lehner, A.F., Buchweitz, J.P. Benefits and Malefits of Solvent Vent Mode in Combination with Tandem Mass Spectrometry for Static Headspace Analysis of Organic Solvents by Gas Chromatography. Chromatographia 85, 315–331 (2022). https://doi.org/10.1007/s10337-022-04135-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-022-04135-1