Abstract
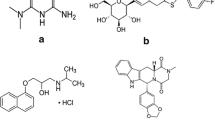
A simple, robust, and sensitive high-performance liquid chromatography tandem mass spectrometry method was developed and validated for determination of a new nonpeptidic glucose-like peptide-1 receptor (GLP-1R) agonist 1,3-bis(4-((p-butoxycarbonyl)amino)benzamido)-2,4-bis(m-methoxy-p-(thiophene-2-methanoyl)phenyl)-cyclobutane-1,3-dicarboxylic acid (Boc5) in rat plasma using wogonin as internal standard (IS). After a simple protein precipitation with methanol, chromatographic separation was achieved on an Atlantis T3 (100 mm × 4.6 mm, 3.5 μm) analytical column eluted by mobile phase consisting of methanol, acetonitrile, and 20 mM ammonium formate addition to 2% formic acid (39:39:22, v/v/v) at flow rate of 0.6 mL/min for 7 min. The quantitation analysis was performed using multiple reaction monitoring at the transitions of m/z 1,077.2 → 784.4 for Boc5 and m/z 285.1 → 270.4 for IS in positive ion mode with electrospray ionization source on an AB SCIEX API 4000 QTRAP mass spectrometer. Good linearity was achieved over the concentration range of 2–2,000 ng/mL with lower limit of quantification at 2 ng/mL. Intra- and interday precisions were less than 7.3 %, and accuracy ranged from −3.6 to 3.0 %. The mean recovery of Boc5 was 93.4–100.2 %, and the matrix effect of Boc5 was 88.0–93.8 %. The validated method was then successfully applied to a pharmacokinetic study of Boc5 in rats.



Similar content being viewed by others
References
Kieffer TJ, Habener JF (1999) Endocr Rev 20:876
Kim W, Egan JM (2008) Pharmacol Rev 60:470–512
Neumiller JJ (2009) Am Pharm Assoc 49:16
Meier JJ, Nauck MA, Kranz D, Holst JJ, Deacon CF, Gaeckler D, Schmidt WE, Gallwitz B (2004) Diabetes 53:654
Van Genugten RE, Van Raalte DH, Diamant M (2009) Diabetes Res Clin Pract 86:26–34
Croom KF, McCormack PL (2009) Drugs 69:1985
Lotfy M, Singh J, Kalász H, Tekes K, Adeghate E (2011) Open Med Chem J 5:82–92
Kieffer T, McIntosh CH, Pederson RA (1995) Endocrinology 136:85
Chen DS, Liao JY, Li N, Zhou CH, Liu Q, Wang GX, Zhang R, Zhang S, Lin LL, Chen KX, Xie X, Nan FJ, Young AA, Wang MW (2007) Proc Natl Acad Sci USA 104:943
Su HR, He M, Li HM, Liu Q, Wang J, Wang YQ, Gao WW, Zhou L, Liao JY, Young AA, Wang MW (2008) PLoS ONE 3:2892
Wang MW, Liu Q, Zhou CH (2010) Acta Pharmacol Sin 1026:31
Murphy KG, Bloom SR (2007) Proc Natl Acad Sci USA 104:689
FDA. Guidance for Industry, Bioanalytical Method Validation, FDA. http://www.fda.gov/cder/guidance
Grobosch T, Schwarze B, Stoecklein D, Binscheck T (2012) J Anal Toxicol 36(1):36–43
Uniyal GC, Bala S, Mathur AK, Kulkarni RN (2001) Phytochem Anal 12(3):206–210
Koufopantelis P, Georgakakou S, Kazanis M, Giaginis C, Margeli A, Papargiri S, Panderi I (2009) J Chromatogr B 877(30):3850–3856
Acknowledgments
This research was supported by Major State Basic Research Development Program of China (“973” Program) (no. 2010CB735602 and no. 2010CB933904).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, R., Fan, H., Gu, Y. et al. Determination of Boc5 in Rat Plasma by HPLC–MS/MS and Its Application to a Pharmacokinetic Study. Chromatographia 75, 721–727 (2012). https://doi.org/10.1007/s10337-012-2248-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10337-012-2248-y