Abstract
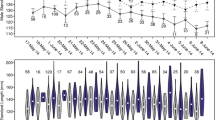
Mate choice is a key process in animals to optimize the fitness benefits of reproduction, and it is generally guided by phenotypic features of potential partners that mirror reproductive abilities. Assortative mating occurs when there is within-pair selection for specific functional traits that can confer fitness benefits. Assortative mating can be positive if mates are more similar, and negative if they are more dissimilar than expected by chance. Mate choice is particularly important in long-lived species with biparental care, such as procellariforms that form long term monogamous bonds. We assessed the mating strategy of a sexually dimorphic Mediterranean procellariform, the Scopoli’s Shearwater (Calonectris diomedea), by testing for assortative mating according to bill (in accordance with previous studies on a sister species) and tarsus size (proxy of body size). We found that shearwaters adopted a positive size-assortative mating by tarsus length, while mating for bill size was random. Moreover, tarsus length was positively correlated with the duration of incubation shifts, when individuals are fasting on eggs. The observed assortative mating could be the results of choice by similarity between individuals, likely because partners with similar relative size have similar tolerance to fasting. Alternatively, the observed pattern could be the product of mutual mate choice, with a selection for large size that could confer competitive abilities in nest selection, defense, foraging aggregations and fasting ability. While our data suggest strong assortative mating in the Scopoli’s Shearwater (R = 0.4), we cannot fully disentangle the multiple processes at play acting on mate choice.
Zusammenfassung
Größen-assortative Paarung bei einem langlebigen, monogamen Seevogel
Die Partnerwahl ist ein wichtiger Prozess bei Tieren, um die Fitnessvorteile der Fortpflanzung zu optimieren, und sie wird im Allgemeinen von phänotypischen Merkmalen potenzieller Partner geleitet, die die Fortpflanzungsfähigkeiten widerspiegeln. Assortative Paarung tritt auf, wenn innerhalb eines Paares eine Selektion auf bestimmte funktionale Merkmale stattfindet, die Fitnessvorteile bringen können. Assortative Paarung kann positiv sein, wenn sich die Partner ähnlicher sind, und negativ, wenn sie unterschiedlicher sind als zufällig erwartet. Die Partnerwahl ist besonders wichtig bei langlebigen Arten mit biparentaler Pflege, wie z. B. bei Sturmtauchern, die langfristige monogame Bindungen eingehen. Wir untersuchten die Paarungsstrategie eines geschlechtsdimorphen mediterranen Sturmtauchers, des Gelbschnabel-Sturmtauchers (Calonectris diomedea), indem wir auf assortative Paarung nach Schnabel- (in Übereinstimmung mit früheren Studien an einer Schwesterart) und Tarsuslänge (stellvertretend für die Körpergröße) prüften. Wir fanden, dass Sturmtaucher eine positive größen-assortative Paarung nach Tarsuslänge zeigen, während die Verpaarung nach Schnabellänge zufällig war. Darüber hinaus korrelierte die Tarsuslänge positiv mit der Dauer der Inkubationsschichten, wenn die Individuen auf den Eiern fasten. Die beobachtete assortative Paarung könnte das Ergebnis einer Auswahl nach Ähnlichkeit zwischen den Individuen sein, wahrscheinlich, weil Partner mit ähnlicher relativer Größe eine ähnliche Toleranz gegenüber dem Fasten haben. Alternativ könnte das beobachtete Muster das Ergebnis einer gegenseitigen Partnerwahl sein, bei der eine Selektion auf Größe erfolgt, die Wettbewerbsvorteile bei der Nestwahl, der Verteidigung, bei der Bildung von Futtergruppen und beim Fasten mit sich bringt. Während unsere Daten auf eine starke assortative Paarung beim Gelbschnabel-Sturmtaucher (R = 0,4) hindeuten, können wir die verschiedenen Prozesse, die bei der Partnerwahl eine Rolle spielen, nicht vollständig entschlüsseln.


Similar content being viewed by others
Data availability
Data used for the analyses are available upon reasonable request from the corresponding author.
References
Andersson M, Simmons LW (2006) Sexual selection and mate choice. Trend Ecol Evol 21:296–302. https://doi.org/10.1016/j.tree.2006.03.015
Baccetti N, Capizzi D, Corbi F, Massa B, Nissardi S, Spano G, Sposimo P (2009) Breeding shearwaters on Italian islands: population size, island selection and co-existence with their main alien predator, the black rat. Riv Ital Orn 78:83–100
Baldauf SA, Kullmann H, Schroth SH, Thünken T, Bakker T (2009) You can’t always get what you want: size assortative mating by mutual mate choice as a resolution of sexual conflict. BMC Evol Biol 9:1–9. https://doi.org/10.1186/1471-2148-9-129
Barbraud C, Chastel O (1999) Early body condition and hatching success in the snow petrel Pagodroma nivea. Polar Biol 21:1–4. https://doi.org/10.1007/s003000050326
Bateman AJ (1948) Intra-sexual selection in Drosophila. Heredity 2:349–368. https://doi.org/10.1038/hdy.1948.21
Bateson PPG (1983) Mate Choice. Cambridge University Press, Cambridge
Bouwhuis S, Vedder O, Becker PH (2015) Sex-specific pathways of parental age effects on offspring lifetime reproductive success in a long-lived seabird. Evolution 69:1760–1771. https://doi.org/10.1111/evo.12692
Bried J, Jouventin P (2001) Site and mate choice in seabirds: an evolutionary approach. In: Schreiber EA, Burger J (eds) Biology of marine birds. CRC Press, Boca Raton, pp 263–305
Bried J, Pontier D, Jouventin P (2003) Mate fidelity in monogamous birds: a re-examination of the Procellariiformes. Anim Behav 65:235–246. https://doi.org/10.1006/anbe.2002.2045
Bried J, Andris M, Dubois MP, Jarne P (2021) Decreased selectivity during mate choice in a small-sized population of a long-lived seabird. J Avian Biol 52:1–13. https://doi.org/10.1111/jav.02837
Burley N (1983) The meaning of assortative mating. Ethol Sociobiol 4:191–203. https://doi.org/10.1016/0162-3095(83)90009-2
Cachia-Zammit R, Borg J (1987) Notes on the breeding biology of the Cory’s shearwater in the Maltese Islands. Il-Merill 24:1–9
Caillaud MC, Via S (2000) Specialized feeding behavior influences both ecological specialization and assortative mating in sympatric host races of pea aphids. Am Nat 156:606–621. https://doi.org/10.1086/316991
Çakmak E, Akın Pekşen Ç, Bilgin CC (2017) Comparison of three different primer sets for sexing birds. J Vet Diagn Invest 29:59–63. https://doi.org/10.1177/1040638716675197
Campioni L, Dell’Omo G, Vizzini S, De Pascalis F, Badalamenti F, Massa B, Rubolini D, Cecere JG (2022) Year-round variation in the isotopic niche of Scopoli’s shearwater (Calonectris diomedea) breeding in contrasting sea regions of the Mediterranean Sea. Mar Environ Res 178:105650–8. https://doi.org/10.1016/j.marenvres.2022.105650
Cecere JG, Catoni C, Maggini I, Imperio S, Gaibani G (2013) Movement patterns and habitat use during incubation and chick-rearing of Cory’ s shearwaters (Calonectris diomedea ) ( Aves : Vertebrata ) from Central Mediterranean: influence of seascape and breeding stage. Ita J Zool 80:82–89. https://doi.org/10.1080/11250003.2012.710654
Cecere JG, Gaibani G, Imperio S (2014) Effects of environmental variability and offspring growth on the movement ecology of breeding Scopoli’s shearwater Calonectris diomedea. Curr Zool 60:622–630. https://doi.org/10.1093/czoolo/60.5.622
Cecere JG, Catoni C, Gaibani G, Geraldes P, Celada C, Imperio S (2015) Commercial fisheries, inter-colony competition and sea depth affect foraging location of breeding Scopoli’s Shearwaters Calonectris diomedea. Ibis 157:284–298. https://doi.org/10.1111/ibi.12235
Chaurand T, Weimerskirch H (1994) Incubation routine, body mass regulation and egg neglect in the blue petrel Halobaena caerulea. Ibis 136:285–290. https://doi.org/10.1111/j.1474-919X.1994.tb01097.x
Cianchetti-Benedetti M, Dell’Omo G, Russo T, Catoni C, Quillfeldt P (2018) Interactions between commercial fishing vessels and a pelagic seabird in the southern Mediterranean Sea. BMC Ecol 18:1–10. https://doi.org/10.1186/s12898-018-0212-x
Cramp S, Simmons KEL (1977) Handbook of the birds of Europe, the Middle East, and North Africa: the birds of the western Palearctic. Ostrich to Ducks, vol 1. Oxford University Press, Oxford
Curé C, Aubin T, Mathevon N (2009) Acoustic convergence and divergence in two sympatric burrowing nocturnal seabirds. Biol J Linn Soc 96:115–134. https://doi.org/10.1111/j.1095-8312.2008.01104.x
Curé C, Mathevon N, Aubin T (2016) Mate vocal recognition in the Scopoli’s shearwater Calonectris diomedea: do females and males share the same acoustic code? Behav Processes 128:96–102. https://doi.org/10.1016/j.beproc.2016.04.013
De Pascalis F, Imperio S, Benvenuti A, Catoni C, Rubolini D, Cecere JG (2020) Sex-specific foraging behaviour is affected by wind conditions in a sexually size dimorphic seabird. Anim Behav 166:207–218. https://doi.org/10.1016/j.anbehav.2020.05.014
Diedenhofen B, Musch J (2015) Cocor: a comprehensive solution for the statistical comparison of correlations. PLoS One 10:1–12. https://doi.org/10.1371/journal.pone.0121945
Einoder LD, Page B, Goldsworthy SD (2008) Sexual size dimorphism and assortative mating in the short-tailed shearwater Puffinus Tenuirostris. Mar Ornithol 36:167–173
Fransson T, Jansson L, Kolehmainen T, Kroon C, Wenninger T (2017) EURING list of longevity records for European birds. http://www.euring.org/data_and_codes/longevity.htm. Accessed 9 May 2022
Gillies N, Padget O, Syposz M, Bond S, Guilford T (2022) Resource allocation underlies parental decision-making during incubation in the Manx Shearwater. Ornithology 139:1–15. https://doi.org/10.1093/ornithology/ukac006
González-Medina E, Castillo-Guerrero JA, Masero JA, Fernández G (2020) Mate selection based on labile traits affects short-term fitness in a long-lived seabird. Proc R Soc Lond B. https://doi.org/10.1098/rspb.2019.2578
González-Solís J, Croxall JP, Wood AG (2000) Sexual dimorphism and sexual segregation in foraging strategies of northern giant petrels, Macronectes halli, during incubation. Oikos 90:390–398. https://doi.org/10.1034/j.1600-0706.2000.900220.x
Grémillet D, Péron C, Pons JB, Ouni R, Authier M, Thévenet M, Fort J (2014) Irreplaceable area extends marine conservation hotspot off Tunisia: insights from GPS-tracking Scopoli’s shearwaters from the largest seabird colony in the Mediterranean. Mar Biol 161:2669–2680. https://doi.org/10.1007/s00227-014-2538-z
Hedrick PW, Smith DW, Stahler DR (2016) Negative-assortative mating for color in wolves. Evolution 70:757–766. https://doi.org/10.1111/evo.12906
Hedrick PW, Tuttle EM, Gonser RA (2018) Negative-assortative mating in the white-throated sparrow. J Hered 109:223–231. https://doi.org/10.1093/jhered/esx086
Helfenstein F, Danchin E, Wagner RH (2004) Assortative mating and sexual size dimorphism in Black-legged Kittiwakes. Waterbirds 27:350–354. https://doi.org/10.1675/1524-4695(2004)027[0350:AMASSD]2.0.CO;2
Ilahiane L, Boano G, Pavia M, Pellegrino I, Grussu M, Voelker G, Galimberti A (2020) Completing the genetic puzzle of the reed warbler complex: insights from Italy. Bird Study 67:440–447. https://doi.org/10.1080/00063657.2021.1927980
Ilahiane L, De Pascalis F, Pisu D, Pala D, Ferrairo F, Cucco M, Rubolini D, Cecere JG, Pellegrino I (2022) No evidence of avian malaria in two Mediterranean endemic seabirds. Mar Ornithol 50:13–17
Jiang Y, Bolnick DI, Kirkpatrick M (2013) Assortative mating in animals. Am Nat. https://doi.org/10.1086/670160
Kvarnemo C (2018) Why do some animals mate with one partner rather than many? A review of causes and consequences of monogamy. Biol Rev 93:1795–1812. https://doi.org/10.1111/brv.12421
Lüdecke D, Ben-Shachar M, Patil I, Waggoner P, Makowski D (2021) Performance: an R package for assessment, comparison and testing of statistical models. J Open Source Softw 6:3139. https://doi.org/10.21105/joss.03139
Lynch M, Walsh B (1998) Genetics and analysis of quantitative traits. Sinauer, Sunderland
Mock DW, Fujioka M (1990) Monogamy and long-term pair bonding in vertebrates. Trend Ecol Evol 5:39–43. https://doi.org/10.1016/0169-5347(90)90045-F
Morinay J, De Pascalis F, Catoni C, Benvenuti A, Imperio S, Rubolini D, Cecere JG (2022) Assessing important conservation areas for colonial species from individual tracking data: an evaluation of the effects of colony structure and temporal heterogeneity in movement patterns. Front Mar Sci 9:854826. https://doi.org/10.3389/fmars.2022.854826
Mougin JL (2000) Pairing in the Cory’s Shearwater (Calonectris diomedea) of Selvagem Grande. J Ornithol 141:319–326. https://doi.org/10.1007/BF02462241
Nava CP, Kim SY, Magalhães MC, Neves V (2014) Do Cory’s shearwaters Calonectris borealis choose mates based on size? J Ornithol 155:869–875. https://doi.org/10.1007/s10336-014-1070-8
Navarro J, González-Solís J, Viscor G (2007) Nutritional and feeding ecology in Cory’s shearwater Calonectris diomedea during breeding. Mar Ecol Prog Ser 351:261–271. https://doi.org/10.3354/meps07115
Peck DR, Erwin CA, Congdon BC (2006) Variation in egg-size in the Wedge-tailed Shearwater (Puffinus pacificus). Emu 106:227–231. https://doi.org/10.1071/MU05037
Pinaud D, Weimerskirch H (2002) Ultimate and proximate factors affecting the breeding performance of a marine top-predator. Oikos 99:141–150
Podos J (2010) Acoustic discrimination of sympatric morphs in Darwin’s finches: a behavioural mechanism for assortative mating? Philos Trans R Soc 365:1031–1039. https://doi.org/10.1098/rstb.2009.0289
R Core Team (2021) R: language and environment or statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 06 July 2021
Ramos JA, Moniz Z, Solá E, Monteiro LR (2003) Reproductive measures and chick provisioning of Cory’s Shearwater Calonectris diomedea borealis in the Azores. Bird Study 50:47–54. https://doi.org/10.1080/00063650309461289
Rosenthal GG (2017) Mate choice: the evolution of sexual decision making from microbes to humans. Princeton University Pres, Princeton
Rubolini D, Maggini I, Ambrosini R, Imperio S, Paiva VH, Gaibani G, Saino N, Cecere JG (2015) The effect of moonlight on Scopoli’s Shearwater Calonectris diomedea colony attendance patterns and nocturnal foraging: a test of the foraging efficiency hypothesis. Ethology 121:284–299. https://doi.org/10.1111/eth.12338
Rull IL, Nicolás L, Neri-Vera N, Argáez V, Martínez M, Torres R (2016) Assortative mating by multiple skin color traits in a seabird with cryptic sexual dichromatism. J Ornithol 157:1049–1062. https://doi.org/10.1007/s10336-016-1352-4
Ryan MJ, Akre KL, Kirkpatrick M (2007) Mate choice. Curr Biol 17:313–316. https://doi.org/10.1016/j.cub.2007.02.002
Schneider J, Fromhage L (2010) Monogynous mating strategies in spiders. In: Kappler P (ed) Animal behaviour: evolution and mechanisms. Springer, Berlin, pp 441–464
Schreiber EA, Burger J (2001) Seabirds in the marine environment. In: Schreiber EA, Burger J (eds) Biology of marine birds. CRC Press, Boca Raton, pp 263–305
Senar JC, Pascual J (1997) Keel and tarsus length may provide a good predictor of avian body size. Ardea 85:269–274
Seyer Y, Gauthier G, Bernatchez L, Therrien JF (2020) Sexing a monomorphic plumage seabird using morphometrics and assortative mating. Waterbirds 42:380–392. https://doi.org/10.1675/063.042.0403
Stoffel MA, Nakagawa S, Schielzeth H (2017) rptR: repeatability estimation and variance decomposition by generalized linear mixed-effects models. Methods Ecol Evol 8:1639–1644. https://doi.org/10.1111/2041-210X.12797
Taborsky B, Guyer L, Taborsky M (2009) Size-assortative mating in the absence of mate choice. Anim Behav 77:439–448. https://doi.org/10.1016/j.anbehav.2008.10.020
Wang D, Forstmeier W, Valcu M, Dingemanse NJ, Bulla M, Both C et al (2019) Scrutinizing assortative mating in birds. PLoS Biol 17:3000156. https://doi.org/10.1371/journal.pbio.3000156
Werner AC, Paiva VH, Ramos JA (2014) On the “real estate market”: individual quality and the foraging ecology of male Cory’s Shearwaters. Auk 131:265–274. https://doi.org/10.1642/AUK-13-172.1
Acknowledgements
We thank Alessandro Mazzoleni, Federica Ferrario, Eugenio Carlon, Giacomo dell’Omo, Bruno Massa and Pierandrea Brichetti for the help during fieldwork, and Stefano Focardi and Andrea Pilastro for the useful discussion on preliminary results. A special thanks to Yuri Donno, Mirko Ugo, Antonella Gaio, and Lorenzo Muzzu for the logistic support provided at the La Maddalena Archipelago. We are grateful to three anonymous referees for the valuable suggestions provided. Fieldwork in Linosa 2008 was carried out within the framework of the agreement between LIPU and Ministry for the Ecological Transition (formerly MATTM) signed in 2007; fieldwork in Tremiti and La Maddalena 2011-2013 was funded by LIPU UK; fieldwork in Maddalena 2018–2019–2020–2021 was carried out within the framework of two agreements between Italian Institute for Environmental Protection and Research (ISPRA) and Parco Nazionale dell'Arcipelago di La Maddalena signed in 2018 and 2019 respectively. LC was supported by Marine and Environmental Sciences Center (MARE) (UIDB/04292/2020 and UIDP/04292/2020).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
Our work was undertaken following the international, national and institutional ethical guidelines on the care and use of captured animals. Birds were caught, ringed, handle, and equipped with leg-mounted geolocators by the Italian Institute for Environmental Protection and Research (ISPRA), under the authorization of Law 157/1992 [Art.4(1) and Art 7(5)], which regulates research on wild bird species in Italy.
Additional information
Communicated by C. Barbraud.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Visalli, F., De Pascalis, F., Morinay, J. et al. Size-assortative mating in a long-lived monogamous seabird. J Ornithol 164, 659–667 (2023). https://doi.org/10.1007/s10336-023-02063-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-023-02063-x