Abstract
Organisms differ in their susceptibility to ionizing radiation, although the ecological basis for such differences remain poorly understood. We hypothesized that ecological characteristics such as body size, diet, migration and pigments of plumage would predict the impact of radiation on abundance through effects on relative food consumption rates, free radicals and antioxidants. We made 2,398 breeding bird censuses and quantified the impact of radiation on abundance at Chernobyl and Fukushima providing statistical replication, but also analyses of interaction effects. The impact of radiation on abundance of birds was stronger at Fukushima than at Chernobyl. Species with small body size and hence relatively high food consumption rates were more negatively impacted. Secondary consumers showed stronger negative effects of radiation on abundance than herbivores, especially at Fukushima. There was no main effect of migration, but migrants were more negatively impacted at Chernobyl, while residents were more negatively impacted at Fukushima. Carotenoid and pheomelanin plumage pigments associated with antioxidant status showed stronger negative effects, especially at Chernobyl compared to Fukushima, while eumelanic coloration which is not related to antioxidant status did not show such an effect. These differences between Chernobyl and Fukuskima may reflect differences in duration of exposure, differences in radioactive isotopes and differences in accumulation of mutations.
Similar content being viewed by others
Introduction
There is considerable interspecific variation among organisms in their susceptibility to radiation effects (e.g., Asker et al. 2011; Romanovskaya et al. 2002; Dighton et al. 2008; Møller and Mousseau 2013). Why that is the case remains an open question. This may arise from differences in exposure and hence differences in the intensity of natural selection that have resulted in the evolution of mechanisms that sustain or even nullify such effects of ionizing radiation. This interesting scientific question has received little attention despite the fact that many species are tolerant to extreme levels of radiation (review in Møller and Mousseau 2013). A few studies have attempted to assess interspecific differences in susceptibility to radiation and interspecific differences in their ecological correlates (Møller and Mousseau 2007b, 2011b; Galván et al. 2011, 2014). Ionizing radiation produces free radicals and thus promotes the generation of reactive oxidative species that deplete antioxidant levels in animals (Riley 1994; Ivaniota et al. 1998; Neyfakh et al. 1998). Not surprisingly, interspecific variation in susceptibility to radiation can be directly linked to ecological traits that are associated with high rates of use of antioxidants such as long-distance migration, dispersal, allocation of antioxidants to coloration and allocation of antioxidants by mothers to their offspring (Møller and Mousseau 2007b). Field studies of individual birds in the vicinity of Chernobyl have shown increased rates of DNA damage at higher levels of background radiation (Bonisoli-Alquati et al. 2010b), and this effect can be linked directly to an increased level of oxidative stress caused by background radiation (Bonisoli-Alquati et al. 2010a).
Among the pigments responsible for integument color, melanins and carotenoids are some of the most prominent and widespread. Phaeomelanin- and carotenoid-based colors share a strong dependence on antioxidant status. Vertebrates synthesize two main forms of melanin: eumelanin and pheomelanin, with the latter producing lighter colors than the former. Glutathione (GSH) levels determine the melanin form that is synthesized, as melanogenesis can either lead to the production of eumelanin when the activity of tyrosinase is high and the ratio of cysteine:dopaquinone is low, or to the production of pheomelanin or even an absence of melanin synthesis under opposite conditions (Ozeki et al. 1997; Galván and Alonso-Alvarez 2009). Therefore, eumelanogenesis takes place when the levels of GSH are low and pheomelanogenesis when the levels of GSH are high, implying that pheomelanogenesis proceeds with higher levels of endogenous oxidative stress as compared to eumelanogenesis (Galván and Solano 2009). The evolutionary implication of this mechanism is that species in which natural selection has favored the development of pheomelanic traits may have a decreased capacity to combat oxidative stress as compared to species with eumelanic traits, as the maintenance of high GSH levels as required by pheomelanogenesis might be metabolically costly under adverse environmental conditions that generate oxidative stress and thus consume GSH resources (Galván and Alonso-Alvarez 2009; Galván and Solano 2009).
GSH is one of the antioxidants most susceptible to radiation (Bump and Brown 1990; Navarro et al. 1997; Vartanian et al. 1994). Therefore, species which have the molecular basis to produce large amounts of pheomelanin may be more limited in their use of GSH to combat oxidative stress, and may thus be more susceptible to ionizing radiation than species in which melanogenesis has been selected to be directed toward the production of eumelanin (Galván et al. 2011). Adaptation to radiation in radioactively contaminated areas and naturally contaminated areas alike may result in increased glutathione levels and body condition, while oxidative stress and DNA damage has decreased with increasing background radiation (Galván et al. 2014). This apparent adaptation to radiation appears to be linked to larger amounts of pheomelanin and smaller amounts of eumelanin being costly, as shown from decreased glutathione levels, increased oxidative stress and DNA damage, and reduced body condition (Galván et al. 2014). Finally, pheomelanin has been shown to induce a change towards the production of less pro-oxidant forms of pheomelanin that may help acclimatize birds to radiation exposure (Galván et al. 2014). Indeed, levels of gluthathione and pheomelanin are significantly correlated (Galván et al. 2014).
Here, we report the results of analyses of unique data on the abundance of breeding birds at Fukushima, Japan and Chernobyl, Ukraine, in relation to background levels of radiation. There has been accumulation of mutations at Chernobyl since 1986 for more than 20 generations for many small birds that typically have very short lifespan, (Møller and Mousseau 2011b), while only a couple of generations have passed since the accident at Fukushima in 2011. Thus, we predicted significant differences in the association between abundance and radiation between Chernobyl and Fukushima for different species of birds, implying a significant area by ecological variable interaction.
The objectives of this study were to test (1) whether the effect of radiation reduced the abundance of birds; (2) whether this effect differed among species; (3) whether body mass, diet, migration and pigment-based coloration affected the abundance of birds; and (4) whether the effects of these predictors differed between Fukushima and Chernobyl, as predicted from their different histories of exposure to radioactive contaminants. Small-sized species have relatively higher metabolism, higher intake rates and hence higher ingestion of radionuclides. Similarly, species at higher trophic levels should suffer more from radioactive contamination as a result of bioaccumulation. Migratory birds through their high metabolic rate and use of antioxidants should differ in impact of radiation compared to residents. Indeed, negative impacts of radiation on abundance are stronger on the abundance of migrants than resident species (Møller and Mousseau 2007b). This is contrary to expectation because migrants spend less time in the contaminated breeding areas than residents. However, migrants deplete their stores of antioxidants during migration resulting in very low levels upon arrival (Ninni et al. 2004). This effect is detrimental because breeding birds start establishing territories, nest building and egg formation upon arrival, causing antioxidant levels to be severely depleted (Møller et al. 2005). Finally, there is a trade-off between allocation of antioxidants to production of feather color and self-maintenance in birds with carotenoid- and pheomelanin-based plumage. Therefore, any individual that allocated a large fraction of antioxidant resources to plumage coloration without consideration of future uses for protection against oxidative stress caused by radiation would be at a selective disadvantage. Likewise, such individuals would also risk increased rates of mutations due to the lack of protection against oxidative stress thus reducing survival prospects.
Methods
Study sites
The breeding bird census points were located at ca. 100-m intervals in forested areas west of the exclusion zone around the Fukushima Daiichi power plants in 2011–2014 (Fig. 1a) or in forested areas within the Chernobyl Exclusion Zone or adjacent areas, or in areas in southern Belarus around Gomel during the breeding seasons 2006–2009 (Fig. 1b). At least one local ornithologist participated in the censuses in Japan to confirm the identity of some difficult bird species.
Census methods
We adopted the point count census method, which provides reliable information on relative abundance of birds (Blondel et al. 1970; Møller 1983; Bibby et al. 2005; Voříšek et al. 2010). The method is based on an observer recording for a period of 5 min all birds seen and heard. Extensive national monitoring programs for breeding and wintering birds based on point counts take place in many different countries, and this effort is part of environmental monitoring by the European Union (Voříšek et al. 2010). This method has provided highly repeatable results for birds and other animals at Chernobyl (Møller and Mousseau 2011a). It consists of counts lasting 5 min during which the number of birds seen or heard is recorded. A.P.M. conducted these standard point counts during 29 May–9 June 2006, 1–11 June 2007, 29 May–5 June 2008, 1–6 June 2009 in the surroundings of Chernobyl (898 census points) and during 11–15 July 2011, 14–19 2012, 14–19 2013 and 11–16 2014 at Fukushima (1,500 census points). Thus, one single 5-min count was recorded for each point in each of the study years. The fact that one person made all counts eliminates any variance in results due to inter-observer variability. There are no bird census data from Chernobyl or Fukushima before the accidents, nor to the best of our knowledge have other scientists conducted bird censuses comparable to ours in the years following the accidents.
We directly tested the reliability of our counts by letting two persons independently perform counts, and the degree of consistency was high for both species richness, total abundance and abundance of individual species (details reported by Møller and Mousseau 2007a for Chernobyl; similar results exist for Fukushima: A. P. Møller, I. Nishiumi and T. A. Mousseau, unpublished data).
Abundance estimates can be affected by numerous confounding variables (Voříšek et al. 2010), and, therefore, it is important to control such variables statistically to assess the underlying relationship between radiation and species richness and abundance. We classified habitats (agricultural habitats with grassland or shrub, deciduous forest, or coniferous forest) and estimated to the nearest 10 % ground cover by herbs, shrub, trees, agricultural habitat, deciduous forest and coniferous forest within a distance of 50 m from the census points. We recorded altitude to the nearest foot (c.30 cm), using GPS. Weather conditions can affect animal activity and hence census results (Voříšek et al. 2010), and we recorded cloud cover at the start of each point count (to the nearest eighth), temperature (°C), and wind force (Beaufort). For each census point, we recorded time of day when the count was started (to the nearest minute). Because activity may show a curvilinear relationship with time of day, with high levels of bird activity in the morning and to a lesser extent in the evening (Voříšek et al. 2010), we also included time squared as an explanatory variable.
Background radiation
We measured radiation in the field and cross-validated these measurements with those reported by the Ukrainian Ministry of Emergencies. Once having finished the 5-min census, we measured α, β, and γ radiation levels at ground level directly in the field at each point where were censused invertebrates using a hand-held dosimeter (Model: Inspector; SE International, Summertown, TN, USA). We measured levels two–three times at each site and averaged the results. We cross-validated our measurements in Ukraine against data from the governmental measurements published by Shestopalov (1996), estimated as the mid-point of the ranges published. This analysis revealed a very strong positive relationship [linear regression on log–log transformed data: F = 1546.49, df = 1, 252, r 2 = 0.86, P < 0.0001, slope (SE) = 1.28 (0.10)], suggesting that our field estimates of radiation provided reliable measurements of levels of radiation among sites.
At Fukushima, we used the same dosimeters, and the measurements were cross-validated with readings with a dosimeter that had been recently calibrated and certified to be accurate by the factory (International Medcom, Sebastopol, CA, USA). We cross-validated tests at Fukushima by comparing our own measurements with measurements obtained at the same locations with a TCS 171-ALOKA used by Japanese authorities. Again, there was a very strong positive relationship [linear regression on log–log transformed data: F = 2427.97, df = 1, 20, r 2 = 0.99, P < 0.0001, slope (SE) = 1.120 (0.023)]. We have made extensive measurements of internal dose in hundreds of birds at Chernobyl and found very strong positive correlations between internal dose and background radiation level (A. P. Møller and T. A. Mousseau, unpublished).
Ecological variables
Body mass
We extracted mean body mass of males and females during the breeding season from Cramp and Perrins (1977–1994), Lislevand et al. (2007) and del Hoyo et al. (1992–2011). Body mass was estimated as the mean value of the means for males and females.
Diet
We scored the diet of different species on a two-level classification based on information in Cramp and Perrins (1977–1994) and del Hoyo et al. (1992–2011): (0) mainly plant material, especially outside the breeding season; and (1) primary predators eating herbivores such as insects and spiders, but also vertebrates.
Migration distance
We estimated migration distance as the difference in latitude between the mean of the northernmost and the southernmost breeding distribution and the mean of the northernmost and the southernmost winter distribution, relying on information in Cramp and Perrins (1977–1994) and del Hoyo et al. (1992–2011). A strict classification of species as either residents or migrants resulted in similar conclusions (unpublished data).
Carotenoid-based coloration
We only considered pigment-based coloration in this study because we did not have information on structural color for the species considered. The extent of plumage patches colored by carotenoids was determined by examining illustrations of adult breeding birds in Cramp and Perrins (1977–1994) and Wild Bird Society of Japan (1982). We considered colors that were yellow, orange and red to be caused by carotenoids (Tella et al. 2004; Olson and Owens 2005). We assigned scores that ranged from 0 (total lack of carotenoid-based color) to 5 (all carotenoid-based).
Melanin-based coloration
We used a method for assigning coloration to melanin developed by (Galván et al. 2011). Information on melanin-based plumage coloration was obtained by examining illustrations of bird species censused at Chernobyl and Fukushima in Cramp and Perrins (1977–1994) and Wild Bird Society of Japan (1982). Eumelanic and pheomelanic traits are generally of distinctive colors, the former being responsible for black and gray colors and the latter for yellowish, reddish, chestnut and brown colors (Toral et al. 2008). Eumelanin and pheomelanin normally occur simultaneously in tissues (Ozeki et al. 1997), but the darker colors conferred by eumelanin (Toral et al. 2008) make evident the lower content of this pigment in chestnut and brown as compared to black and gray colors (Galván and Alonso-Alvarez 2009). Furthermore, many bird species have feather melanin contents of high purity (>90 % of either eumelanin or pheomelanin; McGraw and Wakamatsu 2004; J.J. Negro, personal communication). Therefore, we considered that black and gray plumage colors were predominantly generated by eumelanin, while chestnut and brown colors were predominantly generated by pheomelanin. We did not consider conspicuous yellow or red colorations assumed to be generated by other pigments (i.e. carotenoids), unless chemically identified as melanin-based by Toral et al. (2008). Although a rough approximation to the real proportion of eumelanic and pheomelanic plumage, the assumption that black-gray colors are eumelanic and brown-chestnut colors are pheomelanic should be adequate for comparative purposes (Owens and Hartley 1998). Thus, we quantified the proportion of melanic plumage parts by examining illustrations in Cramp and Perrins (1977–1994) and and Wild Bird Society of Japan (1982). Illustrations of both resting and flying adult birds in breeding plumage were examined. The method used by Beauchamp and Heeb (2001) and Galván (2008) was followed to obtain estimates of the proportion of eu- and pheomelanic color present in the plumage of each species, assigning scores that ranged from 0 (total lack of melanic color) to 5 (all melanic). When a species was sexually dichromatic regarding melanin-based coloration, eumelanic and pheomelanic scores were the average obtained for males and females. When a species had different subspecies or color morphs differing in extent or type of melanin-based coloration, we used the nominate subspecies or the most common morph, respectively. It must be noted that eu- and pheomelanic color patches can coexist in the same feathers, and thus the sum of both color scores, in a species that presents both color types, is not always necessarily 5, but higher values are also possible.
All interspecific data are reported in Appendix 1 in the electronic supplementary material.
Statistical analyses
Body mass, migration distance and radiation level were log10-transformed and coverage with agricultural land, herbs, shrub and trees, deciduous forest, coniferous forest and cloud cover were square-root arcsine-transformed before analyses.
We quantified the relationship between abundance of different species and level of radiation by estimating the Kendall rank-order correlation for the relationship between abundance and log10-transformed radiation while including all potentially confounding variables in the statistical models (coverage by herbs, shrub, trees, agricultural habitat, deciduous forest and coniferous forest, altitude, cloud cover, temperature, wind force, time of day and time of day squared). We used non-parametric rank order correlations because the assumptions of parametric analyses were violated. We have previously shown that statistical control for the confounding variables listed above did not affect the conclusions of analyses (Møller and Mousseau 2011a). Therefore, we used the Kendall rank-order correlations from analyses of abundance in relation to radiation rather than partial rank-order correlations in the subsequent analysis. In this model, we included slope as the response variable and body mass, diet, migration distance and area and the two-way interactions between body mass, diet, migration distance and area as predictors. We did not include other interactions because there were no a priori predictions.
It is a common underlying assumption of most statistical analyses that each data point provides equally precise information about the deterministic part of total process variation, i.e. the standard deviation of the error term is constant over all values of the predictor variable(s) (Sokal and Rohlf 1995). Because estimates of slopes depend on sample sizes, and because sample sizes vary considerably among bird species, such variation can have serious consequences for conclusions (Garamszegi and Møller 2010, 2011). The standard solution to this problem is to weight each observation by sampling effort in order to use all data relative to their importance, by giving each datum a weight that reflects its degree of precision due to sampling effort (Draper and Smith 1981; Neter et al. 1996; Garamszegi and Møller 2010). Therefore, we weighted the statistical model by sample size. All analyses were made with JMP (SAS 2012).
Results
We investigated the impact of low-dose radiation on the abundance of 154 species of birds using 2,398 standardized point counts, which resulted in records of 15,811 individual birds. A total of 14 species are common to Chernobyl and Fukushima allowing for comparison of the effect of radiation on abundance of the same species in two different environments (Møller et al. 2012a). The three species showing the strongest negative correlated with level of background radiation level at Chernobyl were Yellowhammer Emberiza citrinella, Chaffinch Fringilla coelebs and Blackcap Sylvia atricapilla. At Fukushima they were Tree Sparrow Passer montanus, Carrion Crow Corvus corone and Barn Swallow Hirundo rustica. The mean Kendall rank-order correlation between abundance and radiation was −0.109 (SE = 0.012), n = 154 species, differing significantly from the expected value zero in the absence of any effect of radiation (t = 9.18, df = 153, P < 0.0001). The mean correlation was −0.047 (0.008) at Chernobyl, but almost three-fold stronger at −0.150 (SE = 0.022) at Fukushima, with both the mean and the variance being significantly larger at Fukushima than at Chernobyl (Welch ANOVA: F = 19.04, df = 1, 11,735, P < 0.0001; Levene’s test, F = 21.73, df = 1, 152, P < 0.0001).
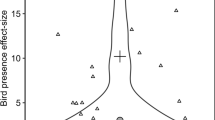
The statistical model explaining variation in the correlations between abundance and background radiation for different species accounted for 64 % of the variance (Table 1). Five out of seven main effects were significant as were five out of six interaction effects between area and ecological variables (Table 1). The correlations between abundance and radiation level were more negative in species with smaller body mass, and this effect differed significantly between Chernobyl and Fukushima as shown by the significant interaction (Fig. 2; Table 1). Carnivorous species showed more strongly negative correlations than herbivorous species and this effect differed between areas with a larger difference at Fukushima than at Chernobyl (Fig. 3; Table 1). Migration distance did not show a significant main effect, but the migration effect differed between Chernobyl and Fukushima with a stronger negative effect in migrants at Chernobyl, and a stronger negative effect in residents at Fukushima (Fig. 4; Table 1).
Plumage color pigments showed different relationships with impact of radiation on abundance. Species with carotenoid-based coloration showed a weaker negative effect than species without carotenoid-based coloration (Table 1). The interaction between carotenoids and area was significant with a stronger negative effect in species with than without carotenoid-based plumage at Chernobyl, and a weaker negative effect in species with than without such plumage at Fukushima (Fig. 5a). There was only a weak and non-significant main effect of eumelanin and no significant effect of the interaction (Fig. 5b; Table 1). There was no significant main effect of pheomelanin (Table 1). A strong interaction between pheomelanin and area implies that at Chernobyl species with pheomelanin-based coloration experienced stronger negative effects of radiation than species without such coloration, while at Fukushima species with pheomelanin showed weaker negative effects than species without pheomelanin in their plumage (Fig. 5c).
Discussion
The main findings of this extensive study of breeding birds at Chernobyl and Fukushima were a negative impact of ionizing radiation on abundance and a number of ecological characteristics explaining variation in heterogeneity of this effect. We found significant main effects of body mass, diet, migration and feather pigments on the impact of radiation on abundance. Importantly, there were significant interactions between area and ecological characteristics, implying that the effects of radiation were not the same at Chernobyl and Fukushima. We will briefly discuss these findings.
Body size is an important correlate of life history, physiology and ecology (Bennett and Owens 2002). Small species eat disproportionately more for their body size than large species due to their surface to volume ratio. Therefore, we should expect small species to ingest disproportionately more radionuclides than large species. Among bird species at Fukushima, there was a stronger negative relationship between abundance and radiation in small-bodied species than among large species, and a similar relationship was found at Chernobyl with a significant interaction between radiation and area. Higher metabolism and higher rates of ingestion by small species should also result in greater production of free radicals and hence greater competition for anti-oxidants between use for metabolism and use due to radiation exposure.
Diet and its interaction with study area was a significant predictor of impact of radiation on abundance. We predicted and found an effect of bioaccumulation of radionuclides in the food web (Voitovich and Afonin 2002; Yakushev et al. 1999). Animals at higher trophic levels generally have higher levels of radionuclide concentrations than animals at lower levels (e.g., Kryshev and Ryabov 1990; Kryshev et al. 1992; Smith et al. 2002). Here, we found a stronger negative effect in secondary consumers compared to herbivores, and the interaction between diet and area was statistically significant, with a stronger effect at Fukushima compared to Chernobyl.
Long-distance migration involves high metabolic effort and extensive use of antioxidants. Møller and Mousseau (2007b) have previously reported for birds from Chernobyl that species with longer migrations had a lower abundance at high radiation levels. Here, we were unable to document a significant main effect of migration on the impact of radiation on abundance. However, we did show a stronger negative impact of radiation on migrants at Chernobyl, but on residents at Fukushima. The Japanese and the European avifaunas differ in their migration distance because more than twice as many species of birds at Fukushima were residents (54 % of 57 species) as the species at Chernobyl (21 % of 97 species). Furthermore, Western Palearctic species migrate longer distances and cross more inhospitable deserts than Eastern Palearctic migrants, while Japanese migrants have the possibility to stop at any of the many archipelagos during migration. Thus, it appears that it is whether or not a species is migrating rather than the actual migration distance that is the important predictor of the effects of radiation on abundance.
The slope of the relationship between abundance and radiation was significantly related to whether plumage color was based on carotenoids and pheomelanin. At Fukushima, species without carotenoids in their plumage had lower abundance at high radiation levels, while that was not the case for species with carotenoid-based plumage color. We have previously reported for Chernobyl that species with carotenoid-based plumage are more strongly negatively impacted (Møller and Mousseau 2007b), as we report here. The slope of the relationship between abundance and background radiation differed between species with and without plumage containing carotenoids and phaeomelanin, but not eumelanin, as we a priori had predicted. The two former kinds of pigment-based coloration impose a trade-off between antioxidant use for maintenance caused by radiation and use of antioxidant pigments allocated to plumage (Møller and Mousseau 2007b; Galván et al. 2011). Interestingly, these effects differed between Fukushima and Chernobyl for carotenoid- and phaeomelanin-based coloration, but not for eumelanin coloration. The main difference between Fukushima and Chernobyl is the duration of chronic exposure to radiation (3 vs. 28 years), the duration of accumulation of deleterious mutations (Møller and Mousseau 2011b) and differences in composition of radionuclides (cesium at Fukushima and strontium, cesium and plutonium at Chernobyl). Galván et al. (2014) have previously suggested and provided evidence for pheomelanin playing an active role in resistance to radiation.
We have previously investigated the importance of similarity in effect of ionizing radiation on abundance due to common phylogenetic descent, showing that conclusions are independent of phylogeny (Møller and Mousseau 2007b; Galván et al. 2011, 2014). Hence, this justifies the absence of phylogenetic analyses in the present study.
There may be many causes for differences in population trends of birds. Here, we have attempted to control statistically for a number of such factors. We have previously shown that a habitat preference for human proximity and other factors did not account for differences in population trends among species (Møller et al. 2012a, b).
In conclusion, we have provided extensive information on the impact of radiation on the abundance of breeding birds at Chernobyl and Fukushima, and the ecological characteristics associated with such impacts generally confirmed findings from previous studies at Chernobyl, although most characteristics showed significant differences between Chernobyl and Fukushima. This level of replication suggests that birds may constitute a useful model system for investigating the impact of radiation and the evolution of radiation-resistance in vertebrates.
References
Asker D, Awad TS, McLandsborough L, Beppu T, Ueda K (2011) Deinococcus depolymerans sp. nov., a gamma- and UV-radiation-resistant bacterium, isolated from a naturally radioactive site. Int J Syst Evol Microbiol 61:1448–1453
Beauchamp G, Heeb P (2001) Social foraging and the evolution of white plumage. Evol Ecol Res 3:703–720
Bennett PM, Owens IPF (2002) Evolutionary ecology of birds. Oxford University Press, Oxford
Bibby CJ, Hill DA, Burgess ND, Mustoe S (2005) Bird census techniques. Academic, London
Blondel J, Ferry C, Frochot B (1970) La méthode des indices ponctuels d’abondance (I. P. A.) au des relevés d’avifaune par “stations d’ecoute”. Alauda 38:55–71
Bonisoli-Alquati A, Mousseau TA, Møller AP, Caprioli M, Saino N (2010a) Increased oxidative stress in barn swallows from the Chernobyl region. Comp Biochem Physiol A 155:205–210
Bonisoli-Alquati A, Voris A, Mousseau TA, Møller AP, Saino N, Wyatt M (2010b) DNA damage in barn swallows (Hirundo rustica) from the Chernobyl region detected by use of the Comet assay. Comp Biochem Physiol C 151:271–277
Bump EA, Brown JM (1990) Role of glutathione in the radiation response of mammalian cells in vitro and in vivo. Pharmacol Therapeut 47:117–136
Cramp S, Perrins CM (1977–1994) The birds of the Western Palearctic, vol 1–9. Oxford University Press, Oxford
del Hoyo J, Elliott A, Sagatal J, Christie DA (eds) (1992–2011) Handbook of the birds of the World. Lynx, Barcelona
Dighton J, Tugay T, Zhdanova N (2008) Fungi and ionizing radiation from radionuclides. FEMS Microbiol Lett 281:109–120
Draper NR, Smith H (1981) Applied regression analysis, 2nd edn. Wiley, New York
Galván I (2008) The importance of white on black: unmelanized plumage proportion predicts display complexity in birds. Behav Ecol Sociobiol 63:303–311
Galván I, Alonso-Alvarez C (2009) The expression of melanin-based plumage is separately modulated by exogenous oxidative stress and a melanocortin. Proc R Soc Lond B 276:3089–3097
Galván I, Solano F (2009) The evolution of eu- and pheomelanic traits may respond to an economy of pigments related to environmental oxidative stress. Pigment Cell Melanoma Res 22:339–342
Galván I, Mousseau TA, Møller AP (2011) Bird population declines due to radiation exposure at Chernobyl are stronger in species with pheomelanin-based coloration. Oecologia 165:827–835
Galván I, Bonisoli-Alquati A, Jenkinson S, Ghanem G, Wakamatsu K, Mousseau TA, Møller AP (2014) Chronic exposure to low-dose radiation at Chernobyl favors adaptation to oxidative stress in birds. Funct Ecol 28:1387–1403
Garamszegi LZ, Møller AP (2010) Effects of sample size and intraspecific variation in phylogenetic comparative studies: a meta-analytic review. Biol Rev 85:797–805
Garamszegi LZ, Møller AP (2011) Nonrandom variation in within-species sample size and missing data in phylogenetic comparative studies. Syst Biol 60:676–680
Ivaniota L, Dubchak AS, Tyshchenko VK (1998) Free radical oxidation of lipids and antioxidant system of blood in infertile women in a radioactive environment. Ukr Biochim Zh 70:132–135 (in Russian)
Kryshev II, Ryabov IN (1990) About the efficiency of trophic level in the accumulation of Cs-137 in fish of the Chernobyl NPP cooling pond. In: Ryabov IN, Ryabtsev IA (eds) Biological and radioecological aspects of the consequences of the Chernobyl accident. USSR Academy of Sciences, Moscow, pp 116–121
Kryshev I, Alexakhin R, Makhonko K (1992) Radioecological consequences of the Chernobyl accident. Nuclear Society, Moscow
Lislevand T, Figuerola J, Székely T (2007) Avian body sizes in relation to fecundity, mating system, display behavior, and resource sharing. Ecology 88:1605
McGraw KJ, Wakamatsu K (2004) Melanin basis of ornamental feather colors in male Zebra Finches. Condor 106:686–690
Møller AP (1983) Methods for monitoring bird populations in the Nordic countries. Nordic Council of Ministers, Oslo
Møller AP, Mousseau TA (2007a) Species richness and abundance of birds in relation to radiation at Chernobyl. Biol Lett 3:483–486
Møller AP, Mousseau TA (2007b) Determinants of interspecific variation in population declines of birds from exposure to radiation at Chernobyl. J Appl Ecol 44:909–919
Møller AP, Mousseau TA (2011a) Efficiency of bio-indicators for low-level radiation under field conditions. Ecol Indic 11:424–430
Møller AP, Mousseau TA (2011b) Conservation consequences of Chernobyl and other nuclear accidents. Biol Cons 144:2787–2798
Møller AP, Mousseau TA (2013) The effects of natural variation in background radioactivity on humans, animals and other organisms. Biol Rev 88:226–254
Møller AP, Surai PF, Mousseau TA (2005) Antioxidants, radiation and mutation in barn swallows from Chernobyl. Proc R Soc Lond B 272:247–253
Møller AP, Hagiwara A, Matsui S, Kasahara S, Kawatsu K, Nishiumi I, Suzuki H, Ueda K, Mousseau TA (2012a) Abundance of birds in Fukushima as judged from Chernobyl. Environ Poll 164:36–39
Møller AP, Nishiumi I, Suzuki H, Ueda K, Mousseau TA (2012b) Differences in effects of radiation on abundance of animals in Fukushima and Chernobyl. Ecol Indic 24:75–81
Navarro J, Obrador E, Pellicer JA, Asensi M, Viña J, Estrela JM (1997) Blood glutathione as an index of radiation-induced oxidative stress in mice and humans. Free Radic Biol Med 22:1203–1209
Neter J, Kutner MH, Nachtsheim CJ, Wasserman W (1996) Applied linear statistical models. Irwin, Chicago
Neyfakh EA, Alimbekova AI, Ivanenko GF (1998) Radiation-induced lipoperoxidative stress in children coupled with deficit of essential antioxidants. Biochim (Moscow) 63:977–987
Ninni P, de Lope F, Saino N, Haussy C, Møller AP (2004) Antioxidants and condition-dependence of arrival date in a migratory passerine. Oikos 105:55–64
Olson V, Owens IPF (2005) Interspecific variation in the use of carotenoid-based coloration in birds: diet, life history and phylogeny. J Evol Biol 18:1534–1546
Owens IPF, Hartley IR (1998) Sexual dimorphism in birds: why are there so many different forms of dimorphism? Proc R Soc Lond B 265:397–407
Ozeki H, Ito S, Wakamatsu K, Ishiguro I (1997) Chemical characterization of pheomelanogenesis starting from dihydroxyphenylalanine or tyrosine and cysteine. Effects of tyrosinase and cysteine concentrations and reaction time. Biochim Biophys Acta 1336:539–548
Riley PA (1994) Free radicals in biology: oxidative stress and the effects of ionizing radiation. Int J Radiat Biol 65:27–33
Romanovskaya VA, Rokitko PV, Mikheev AN, Gushcha NI, Malashenko YR, Chernaya NA (2002) The effect of γ-radiation and desiccation on the viability of the soil bacteria isolated from the alienated zone. Microbiology 71:608–613
SAS Institute Inc (2012) JMP. SAS Institute, Cary
Shestopalov VM (1996) Atlas of Chernobyl exclusion zone. Ukrainian Academy of Science, Kiev
Smith MH, Oleksyk TK, Tsyusko O (2002) Effects of trophic position and ecosystem type on the form of the frequency distribution of radiocesium at Chornobyl and nuclear sites in the United States. In: Proc Int Symp: transfer of radionuclides in biosphere: prediction and assessment, Mito, Japan, pp 37–48, December 18–19, 2002
Sokal RR, Rohlf FJ (1995) Biometry. Freeman, New York
Tella JL, Figuerola J, Negro JJ, Blanco G, Rodríguez-Estrella R, Forero MG, Blazquez MC, Green AJ, Hiraldo F (2004) Ecological, morphological and phylogenetic correlates of interspecific variation in plasma carotenoid concentration in birds. J Evol Biol 17:156–164
Toral GM, Figuerola J, Negro JJ (2008) Multiple ways to become red: pigment identification in red feathers using spectrometry. Comp Biochem Physiol B 150:147–152
Vartanian LS, Gurevich S, Kozachenko AI, Nagler LG, Burlakova EB (1994) Age-related peculiarities of effect of low dose ionizing radiation on blood antioxidant enzyme system status in Chernobyl’s accident liquidation participant. Adv Gerontol 14:48–54 (in Russian)
Voitovich AM, Afonin VYu (2002) DNA damages and radionuclide accumulation in wild small vertebrates. In: Environmental radioactivity in the Arctic and Antarctic, Proceedings of the 5th International Conference, St. Petersburg, Russia, pp 340–343, 16–20
Voříšek P, Klvanova A, Wotton S, Gregory RD (2010) A best practice guide for wild bird monitoring schemes. European Union, Bruxelles
Wild Bird Society of Japan (1982) A field guide to the birds of Japan. Wild Bird Society of Japan, Tokyo
Yakushev BI, Budkevich TA, Zabolotny AI, Mironov V, Kudryashov VP (1999) Contamination of vegetation in Belarus by transuranium radionuclides due to Chernobyl NPP accident. In: Goossens LHJ (ed) Proc 9th Ann Conf “Risk analysis: Facing the new millennium”, October 10–13, 1999. Delft University Press, Rotterdam, pp 841–844
Acknowledgments
We gratefully acknowledge logistic support and help by Professor A. Hagiwara, T. Kanagawa, K. Kawai, Professor K. Kawatsu and A.M. Smith in Japan. We are especially grateful to Namie-cho and the people of Fukushima Prefecture who permitted and supported us to conduct this study in the field. We gratefully acknowledge support from the US National Science Foundation, the University of South Carolina College of Arts and Sciences, CNRS (France), the Samuel Freeman Charitable Trust, Qiagen GmbH, JSPS KAKENHI Grant Number 26440254, and anonymous gifts from individuals in Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Matthysen.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Møller, A.P., Mousseau, T.A., Nishiumi, I. et al. Ecological differences in response of bird species to radioactivity from Chernobyl and Fukushima. J Ornithol 156 (Suppl 1), 287–296 (2015). https://doi.org/10.1007/s10336-015-1173-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10336-015-1173-x