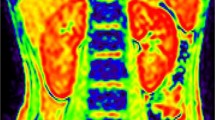
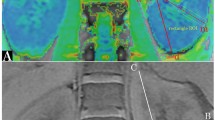
Abstract
Objective
Children are often sedated for renal blood oxygenation level-dependent (BOLD) magnetic resonance imaging (MRI) and may require low-dose oxygen administration. It is unclear whether low-dose oxygen administration affects results of BOLD MRI. We investigated the effect of low-dose oxygen administration on renal BOLD MRI and its variation by the presence or absence of renal disease.
Materials and methods
We retrospectively examined children undergoing MRI for renal disease between 2013 and 2020. Patients were divided into glomerulonephritis and non-glomerulonephritis groups; spin relaxation time (T2*) was determined using a 3.0 T MRI system.
Results
The study included 10 children (5 patients in each group); patient characteristics between the groups did not differ significantly. In the entire cohort, oxygen administration reduced mean spin relaxation rate (R2*) value in the medulla (p < 0.04). The mean R2* value decreased with oxygen administration in the non-glomerulonephritis group, whereas this was not observed in the glomerulonephritis group. The responses to oxygen administration of the two groups differed significantly in the cortex (p < 0.05) and medulla (p < 0.02).
Discussion
Low-dose oxygen administration affects the results of BOLD MRI. We suggest that understanding the fluctuations due to oxygen administration is useful in monitoring the disease activity of glomerulonephritis.




Similar content being viewed by others
Availability of data and materials
The datasets generated during and/or analyzed in the present study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Abbreviations
- ANOVA:
-
Analysis of variance
- BOLD:
-
Blood oxygenation level-dependent
- CAKUT:
-
Congenital anomalies of the kidney and urinary tract
- eGFR:
-
Estimated glomerular filtration rate
- MRI:
-
Magnetic resonance imaging
- RAS:
-
Renin–angiotensin system
- RN:
-
Reflux nephropathy
- ROI:
-
Region of interest
- TLCO:
-
Twelve-layer concentric objects
- VUR:
-
Vesicoureteral reflux
References
Ding J-J, Lin S-H, Huang J-L, Wu T-W, Hsia S-H, Lin J-J, Chou Y-C, Tseng M-H (2019) Risk factors for complications of percutaneous ultrasound-guided renal biopsy in children. Pediatr Nephrol 35(2):271–278
Prasad PV, Edelman RR, Epstein FH (1996) Noninvasive evaluation of intrarenal oxygenation with BOLD MRI. Circulation 94(12):3271–3275
Chen F, Li S, Sun D (2018) Methods of blood oxygen level-dependent magnetic resonance imaging analysis for evaluating renal oxygenation. Kidney Blood Press Res 43(2):378–388
Luo F, Liao Y, Cui K, Tao Y (2020) Noninvasive evaluation of renal oxygenation in children with chronic kidney disease using blood–oxygen-level-dependent magnetic resonance imaging. Pediatr Radiol 50(6):848–854
Chehade H, Milani B, Ansaloni A, Anex C, Bassi I, Piskunowicz M, Stuber M, Cachat F, Burnier M, Pruijm M (2016) Renal tissue oxygenation in children with chronic kidney disease due to vesicoureteral reflux. Pediatr Nephrol 31(11):2103–2111
Hall ME, Jordan JH, Juncos LA, Hundley WG, Hall JE (2018) BOLD magnetic resonance imaging in nephrology. Int J Nephrol Renovasc Dis 11:103–112
Niendorf T, Pohlmann A, Arakelyan K, Flemming B, Cantow K, Hentschel J, Grosenick D, Ladwig M, Reimann H, Klix S, Waiczies S, Seeliger E (2015) How bold is blood oxygenation level-dependent (BOLD) magnetic resonance imaging of the kidney? Opportunities, challenges and future directions. Acta Physiol (Oxf) 213(1):19–38
Prasad PV, Epstein FH (1999) Changes in renal medullary pO2 during water diuresis as evaluated by blood oxygenation level-dependent magnetic resonance imaging: effects of aging and cyclooxygenase inhibition. Kidney Int 55(1):294–298
Siddiqi L, Hoogduin H, Visser F, Leiner T, Mali WP, Blankestijn PJ (2014) Inhibition of the renin-angiotensin system affects kidney tissue oxygenation evaluated by magnetic resonance imaging in patients with chronic kidney disease. J Clin Hypertens (Greenwich) 16(3):214–218
Bane O, Mendichovszky IA, Milani B, Dekkers IA, Deux JF, Eckerbom P, Grenier N, Hall ME, Inoue T, Laustsen C, Lerman LO, Liu C, Morrell G, Pedersen M, Pruijm M, Sadowski EA, Seeliger E, Sharma K, Thoeny H, Vermathen P, Wang ZJ, Serafin Z, Zhang JL, Francis ST, Sourbron S, Pohlmann A, Fain SB, Prasad PV (2020) Consensus-based technical recommendations for clinical translation of renal BOLD MRI. MAGMA 33(1):199–215
Boss A, Martirosian P, Jehs MC, Dietz K, Alber M, Rossi C, Claussen CD, Schick F (2009) Influence of oxygen and carbogen breathing on renal oxygenation measured by T2*-weighted imaging at 3.0 T. NMR Biomed 22(6):638–645
Donati OF, Nanz D, Serra AL, Boss A (2012) Quantitative BOLD response of the renal medulla to hyperoxic challenge at 1.5 T and 3.0 T. NMR Biomed 25(10):1133–1138
Khatir DS, Pedersen M, Jespersen B, Buus NH (2014) Reproducibility of MRI renal artery blood flow and BOLD measurements in patients with chronic kidney disease and healthy controls. J Magn Reson Imaging 40(5):1091–1098
Zheng Z, Wang Y, Yan T, Jia J, Li D, Wei L, Shang W, Shi H (2020) Detection of renal hypoxia configuration in patients with lupus nephritis: a primary study using blood oxygen level-dependent MR imaging. Abdom Radiol (NY). https://doi.org/10.1007/s00261-020-02794-y
Milani B, Ansaloni A, Sousa-Guimaraes S, Vakilzadeh N, Piskunowicz M, Vogt B, Stuber M, Burnier M, Pruijm M (2017) Reduction of cortical oxygenation in chronic kidney disease: evidence obtained with a new analysis method of blood oxygenation level-dependent magnetic resonance imaging. Nephrol Dial Transplant 32(12):2097–2105
Pedersen M, Dissing TH, Morkenborg J, Stodkilde-Jorgensen H, Hansen LH, Pedersen LB, Grenier N, Frokiaer J (2005) Validation of quantitative BOLD MRI measurements in kidney: application to unilateral ureteral obstruction. Kidney Int 67(6):2305–2312
Pohlmann A, Arakelyan K, Hentschel J, Cantow K, Flemming B, Ladwig M, Waiczies S, Seeliger E, Niendorf T (2014) Detailing the relation between renal T2* and renal tissue pO2 using an integrated approach of parametric magnetic resonance imaging and invasive physiological measurements. Invest Radiol 49(8):547–560
Xin-Long P, Jing-Xia X, Jian-Yu L, Song W, Xin-Kui T (2012) A preliminary study of blood–oxygen-level-dependent MRI in patients with chronic kidney disease. Magn Reson Imaging 30(3):330–335
Lassen UV, Thaysen JH (1961) Correlation between sodium transport and oxygen consumption in isolated renal tissue. Biochim Biophys Acta 47:616–618
Herthelius M, Berg U (1999) Renal function during and after childhood acute poststreptococcal glomerulonephritis. Pediatr Nephrol 13(9):907–911
Li X, Xu X, Zhang Q, Ren H, Zhang W, Liu Y, Yan F, Chen N (2014) Diffusion weighted imaging and blood oxygen level-dependent MR imaging of kidneys in patients with lupus nephritis. J Transl Med 12:295
Nishino T, Takahashi K, Ono S, Mimaki M (2021) Blood oxygen level-dependent imaging for evaluating C3 glomerulonephritis. Pediatri Int. https://doi.org/10.1111/ped.14605
Shi H, Yan T, Li D, Jia J, Shang W, Wei L, Zheng Z (2017) Detection of renal hypoxia in lupus nephritis using blood oxygen level-dependent MR imaging: a multiple correspondence analysis. Kidney Blood Press Res 42(1):123–135
Zheng Z, Shi H, Ma H, Li F, Zhang J, Zhang Y (2014) Renal oxygenation characteristics in healthy native kidneys: assessment with blood oxygen level-dependent magnetic resonance imaging. Nephron Physiol 128(3–4):47–54
Pruijm M, Mendichovszky IA, Liss P, Van der Niepen P, Textor SC, Lerman LO, Krediet CTP, Caroli A, Burnier M, Prasad PV (2018) Renal blood oxygenation level-dependent magnetic resonance imaging to measure renal tissue oxygenation: a statement paper and systematic review. Nephrol Dial Transplant 33(suppl 2):ii22–ii28
Eckardt KU, Bernhardt WM, Weidemann A, Warnecke C, Rosenberger C, Wiesener MS, Willam C (2005) Role of hypoxia in the pathogenesis of renal disease. Kidney Int Suppl. https://doi.org/10.1111/j.1523-1755.2005.09909.x(99):S46-51
Cardona-Grau D, Kogan BA (2015) Update on multicystic dysplastic kidney. Curr Urol Rep 16(10):67
Brenner BM, Meyer TW, Hostetter TH (1982) Dietary protein intake and the progressive nature of kidney disease: the role of hemodynamically mediated glomerular injury in the pathogenesis of progressive glomerular sclerosis in aging, renal ablation, and intrinsic renal disease. N Engl J Med 307(11):652–659
van der Bel R, Coolen BF, Nederveen AJ, Potters WV, Verberne HJ, Vogt L, Stroes ES, Krediet CT (2016) Magnetic resonance imaging-derived renal oxygenation and perfusion during continuous, steady-state angiotensin-II Infusion in healthy humans. J Am Heart Assoc 5(3):e003185
Acknowledgements
The authors would also like to thank Dr. Bastien Milani of the Service of Nephrology and Hypertension, Centre hospitalier universitaire vaudois, for his technical advice.
Funding
This study received no specific grants from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
The authors TN and KT contributed equally to this work. Study conception and design; TN: study conception and design, acquisition of data, analysis and interpretation of data, drafting of manuscript, and critical revision. KT: study conception and design, acquisition of data, analysis and interpretation of data, and critical revision. SO: study conception and design, and critical revision. MM: study conception and design, and critical revision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
The study was approved by the Teikyo University Ethical Review Board for Medical and Health Research Involving Human Subjects (Approval No. 20-195-2). All the procedures in studies involving human participants were performed in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent to participate/consent for publication
The study plan outline was presented on Teikyo University website, where patients and their guardians could ask questions about the study and opt out from the use of their data. Informed consent to participate and publication was obtained on the day before MRI. The study was explained to all patients and their parents/guardians in plain language with explanatory documents, and written consent for participation in research and publication was obtained from the parents/guardians of all subjects. At the same time, it was clarified that the parents/guardians had the right to refuse participation and withdraw consent at will.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nishino, T., Takahashi, K., Ono, S. et al. Effects of low-dose oxygen administration on renal blood oxygenation level-dependent MRI in children with glomerulonephritis. Magn Reson Mater Phy 34, 823–831 (2021). https://doi.org/10.1007/s10334-021-00945-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10334-021-00945-w