Abstract
Purpose
The purpose of this prospective study was to assess physiologic changes in the renal corticomedullary 23Na-concentration ([23Na]) gradient with 23Na-MRI at 3.0T in patients with central diabetes insipidus (CDI) before and after intranasal administration of 20 μg desmopressin (DDAVP).
Methods and materials
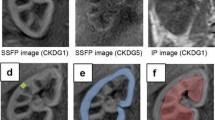
Four patients with CDI (all male, mean age 60.2 years) were included in this IRB-approved study. For 23Na-imaging, a 3D density adapted, radial GRE-sequence (TE = 0.55 ms; TR = 120 ms; projections = 8,000; spatial resolution = 5 × 5 × 5 mm3) was used in combination with a dedicated 23Na-coil and reference phantoms. The corticomedullary [23Na] gradient (in mmol/L/mm) was calculated pixel-by-pixel along a linear region-of-interest (ROI) spanning from the renal cortex in the direction of the medulla. Mean ± SDs of [23Na] were calculated for each patient as well as for the entire group.
Results
Mean [23Na] increased along the corticomedullary gradient from the cortex (pre-DDAVP 38.0 ± 6.3 mmol/L vs. post-DDAVP 30.7 ± 3.5 mmol/L) to the medulla (pre-DDAVP 71.6 ± 14.8 mmol/L vs. post-DDAVP 59.7 ± 10.8 mmol/L). The overall mean decrease of [23Na] after DDAVP administration was 17.1 ± 1.1 %.
Conclusion
23Na-MRI with state-of-the-art techniques at 3T depicts the physiologic renal response to the administration of desmopressin in patients with central diabetes insipidus.


Similar content being viewed by others
References
Di Iorgi N, Napoli F, Allegri AE, Olivieri I, Bertelli E, Gallizia A, Rossi A, Maghnie M (2012) Diabetes insipidus—diagnosis and management. Horm Res Paediatr 77(2):69–84
Makaryus AN, McFarlane SI (2006) Diabetes insipidus: diagnosis and treatment of a complex disease. Cleve Clin J Med 73(1):65–71
Caldwell HK, Young WSI (2006) Oxytocin and vasopressin: genetics and behavioral implications. In: Lajtha A, Lim R (eds) Handbook of neurochemistry and molecular neurobiology: neuroactive proteins and peptides, 3rd edn. Springer, Berlin, pp 573–607
Agarwal SK, Gupta A (2008) Aquaporins: the renal water channels. Indian J Nephrol 18(3):95–100
Tomita K, Pisano JJ, Knepper MA (1985) Control of sodium and potassium transport in the cortical collecting duct of the rat. Effects of bradykinin, vasopressin, and deoxycorticosterone. J Clin Invest 76(1):132–136
Harris AS (1989) Clinical experience with desmopressin: efficacy and safety in central diabetes insipidus and other conditions. J Pediatr 114(4 Pt 2):711–718
Kosman ME (1978) Evaluation of a new antidiuretic agent, desmopressin acetate (DDAVP). JAMA 240(17):1896–1897
Kapic E, Becic F, Todic M (2005) Modern approach in treatment of diabetes insipidus. Bosn J Basic Med Sci 5(2):38–42
Vande Walle J, Stockner M, Raes A, Norgaard JP (2007) Desmopressin 30 years in clinical use: a safety review. Curr Drug Saf 2(3):232–238
Kim RJ, Malattia C, Allen M, Moshang T, Maghnie M (2004) Vasopressin and desmopressin in central diabetes insipidus: adverse effects and clinical considerations. Pediatr Endocrinol Rev 2(Suppl 1):115–123
Lam KS, Wat MS, Choi KL, Ip TP, Pang RW, Kumana CR (1996) Pharmacokinetics, pharmacodynamics, long-term efficacy and safety of oral 1-deamino-8-d-arginine vasopressin in adult patients with central diabetes insipidus. Br J Clin Pharmacol 42(3):379–385
Maghnie M, Cosi G, Genovese E, Manca-Bitti ML, Cohen A, Zecca S, Tinelli C, Gallucci M, Bernasconie S, Boscherini B, Severi F, Arico M (2000) Central diabetes insipidus in children and young adults. N Engl J Med 343(14):998–1007
Tisdall M, Crocker M, Watkiss J, Smith M (2006) Disturbances of sodium in critically ill adult neurologic patients: a clinical review. J Neurosurg Anesthesiol 18(1):57–63
Sigounas DG, Sharpless JL, Cheng DM, Johnson TG, Senior BA, Ewend MG (2008) Predictors and incidence of central diabetes insipidus after endoscopic pituitary surgery. Neurosurgery 62(1):71–78
Argyropoulou MI, Kiortsis DN (2005) MRI of the hypothalamic-pituitary axis in children. Pediatr Radiol 35(11):1045–1055
Fujisawa I (2004) Magnetic resonance imaging of the hypothalamic-neurohypophyseal system. J Neuroendocrinol 16(4):297–302
Xu X, Fang W, Ling H, Chai W, Chen K (2010) Diffusion-weighted MR imaging of kidneys in patients with chronic kidney disease: initial study. Eur Radiol 20(4):978–983
Thoeny HC, Zumstein D, Simon-Zoula S, Eisenberger U, De Keyzer F, Hofmann L, Vock P, Boesch C, Frey FJ, Vermathen P (2006) Functional evaluation of transplanted kidneys with diffusion-weighted and BOLD MR imaging: initial experience. Radiology 241(3):812–821
Michaely HJ, Metzger L, Haneder S, Hansmann J, Schoenberg SO, Attenberger UI (2012) Renal BOLD-MRI does not reflect renal function in chronic kidney disease. Kidney Int 81(7):684–689
Haneder S, Konstandin S, Morelli JN, Nagel AM, Zoellner FG, Schad LR, Schoenberg SO, Michaely HJ (2011) Quantitative and qualitative 23Na MR imaging of the human kidneys at 3 T: before and after a water load. Radiology 260(3):857–865
Maril N, Rosen Y, Reynolds GH, Ivanishev A, Ngo L, Lenkinski RE (2006) Sodium MRI of the human kidney at 3 Tesla. Magn Reson Med 56(6):1229–1234
Rosen Y, Lenkinski RE (2009) Sodium MRI of a human transplanted kidney. Acad Radiol 16(7):886–889
Haneder S, Michaely HJ, Schoenberg SO, Konstandin S, Schad LR, Siebenlist K, Wertz H, Wenz F, Lohr F, Boda-Heggemann J (2012) Assessment of renal function after conformal radiotherapy and intensity-modulated radiotherapy by functional (1)H-MRI and (23)Na-MRI. Strahlenther Onkol 188(12):1146–1154
Lanzman RS, Ljimani A, Pentang G, Zgoura P, Zenginli H, Kröpil P, Heusch P, Schek J, Miese FR, Blondin D, Antoch G, Wittsack HJ (2013) Kidney transplant: functional assessment with diffusion-tensor MR imaging at 3T. Radiology 266(1):218–225
Gaudiano C, Clementi V, Busato F, Corcioni B, Orrei MG, Ferramosca E, Fabbri E, Berardi P, Santoro A, Golfieri R (2013) Diffusion tensor imaging and tractography of the kidneys: assessment of chronic parenchymal diseases. Eur Radiol
Artz NS, Sadowski EA, Wentland AL, Grist TM, Seo S, Djamali A, Fain SB (2011) Arterial spin labeling MRI for assessment of perfusion in native and transplanted kidneys. Magn Reson Imaging 29(1):74–82
Maeda M, Seo Y, Murakami M et al (1990) Sodium-23 MR imaging of the kidney in guinea pig at 2.1 T, following arterial, venous, and ureteral ligation. Magn Reson Med 16(3):361–367
Maril N, Margalit R, Rosen S, Heyman SN, Degani H (2006) Detection of evolving acute tubular necrosis with renal 23Na MRI: studies in rats. Kidney Int 69(4):765–768
Lanz T, Mayer M, Robson MD, Neubauer S, Ruff J, Weisser A (2007) An 8 channel 23Na heart array for application at 3T. In: Proceedings of the 15th scientific meeting, International society for magnetic resonance in medicine, Berlin, p 247
Haneder S, Konstandin S, Morelli JN, Schad LR, Schoenberg SO, Michaely HJ (2013) Assessment of the renal cortico-medullary 23Na-gradient using isotropic data sets. Acad Radiol 20:407–413
Maril N, Margalit R, Mispelter J, Degani H (2004) Functional sodium magnetic resonance imaging of the intact rat kidney. Kidney Int 65(3):927–935
Maril N, Margalit R, Mispelter J, Degani H (2005) Sodium magnetic resonance imaging of diuresis: spatial and kinetic response. Magn Reson Med 53(3):545–552
Lote C (1994) Principles of renal physiology. Chapman & Hall, London
Joukhadar C, Schenk B, Kaehler ST, Kollenz CJ, Bauer P, Mueller M, Eichler H-G (2003) A replicate study design for testing bioequivalence: a case study on two desmopressin nasal spray preparations. Eur J Clin Pharmacol 59(8–9):631–636
Vilhardt H, Lundin S (1986) Biological effect and plasma concentrations of DDAVP after intranasal and peroral administration to humans. Gen Pharmacol 17(4):481–483
Acknowledgments
We thank Dr. Armin Nagel for providing the modified, density-adapted 23Na-sequence. Parts of this study were supported by a research grant from Deutsche Forschungsgemeinschaft (DFG) SCHO 710/2-2.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Haneder, S., Michaely, H.J., Konstandin, S. et al. 3T Renal 23Na-MRI: effects of desmopressin in patients with central diabetes insipidus. Magn Reson Mater Phy 27, 47–52 (2014). https://doi.org/10.1007/s10334-013-0377-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10334-013-0377-4