Abstract
Objective
The purpose of this study was to assess the curative effect and adverse reaction of preoperative induction chemotherapy with gemcitabine combined with cisplatin for locally advanced non-small cell lung cancer (NSCLC).
Methods
This prospective randomized controlled trial included 115 patients with locally advanced NSCLC were randomly divided into experimental and control groups and were treated from January 2007 to January 2010. The experimental group of 63 cases was treated with two cycles of induction chemotherapy before operation, radical surgery had been performed about three weeks after completion of chemotherapy, followed by received two cycles of chemotherapy. And the control group (52 cases) was treated at first with radical surgery, then treated with four cycles of chemotherapy. Two groups of the cases received routine thoracic radiotherapy with a total dose of 45 Gy. One cycle of gemcitabine combined with cisplatin regimen included gemcitabine 1000 mg/m2 on day 1 and day 8 and cisplatin 25 mg/m2 on day 1, day 2 and day 3 by intravenous infusion, with 21 days as one cycle. The tumor recurrence was evaluated by chest CT and abdominal B-ultrasound. Efficacy and toxicity results were compared by two groups.
Results
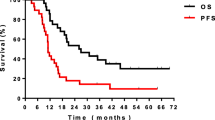
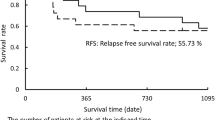
All patients were followed up for three months to two years. The surgical stage of the experimental group reduced, two-years disease-free survival and postoperative recovery in the experimental group were better than in the control group, the difference was statistical significant. Toxicity and side effect after chemotherapy were mainly bone marrow suppression and gastrointestinal reactions, other complications included thrombocytopenia, leukopenia, anemia, liver and kidney dysfunction were no significant difference in two groups.
Conclusion
Preoperative induction chemotherapy with gemcitabine combined with cisplatin for locally advanced lung cancer can reduce the surgical staging and extend the postoperative disease-free survival.
Similar content being viewed by others
References
Jemal A, Bray F, Center MM, et al. Global cancer statistics. CA Cancer J Clin, 2011, 61: 69–90.
Nakajima E, Katou H. Adjuvant chemotherapy for resectable nonsmall cell lung cancer (NSCLC). Kyobu Geka, 2008, 61: 4–8.
Spasova I, Petera J, Hytych V. The role of neoadjuvant chemotherapy in marginally resectable or unresectable stage III non-small cell lung cancer. Neoplasma, 2002, 49: 189–196.
Watanabe H, Yamamoto S, Kunitoh H, et al. Tumor response to chemotherapy: The validity and reproducibility of RECIST guidelines in NSCLC patients. Cancer Sci, 2003, 94: 1015–1020.
Garrido P, Gonzalez-Larriba JL, Insa A, et al. Long-term survival associated with complete resection after induction chemotherapy in stage IIIA (N2) and IIIB (T4N0-1) non small-cell lung cancer patients: the Spanish Lung Cancer Group Trial 9901. J Clin Oncol, 2007, 25: 4736–4742.
Ardizzoni A, Boni L, Tiseo M, et al. Cisplatin-versus carboplatin-based chemotherapy in first-line treatment of advanced non-small-cell lung cancer: an individual patient data meta-analysis. J Natl Cancer Inst, 2007, 99: 847–857.
Kosmidis PA, Dimopoulos MA, Syrigos K, et al. Gemcitabine versus gemcitabine-carboplatin for patients with advanced non-small cell lung cancer and a performance status of 2: a prospective randomized phase II study of the Hellenic Cooperative Oncology Group. J Thorac Oncol, 2007, 2: 135–140.
Szkorupa M, Klein J, Bohanes T, et al. Neoadjuvant chemotherapy and surgical treatment in advanced stages of non-small cell lung cancer. Rozhl Chir, 2011, 90: 433–439.
Higgins KA, Chino JP, Ready N, et al. Persistent N2 disease after neoadjuvant chemotherapy for non-small-cell lung cancer. J Thorac Cardiovasc Surg, 2011, 142: 1175–1179.
Doddoli C, Thomas P, Thirion X, et al. Postoperative complications in relation with induction therapy for lung cancer. Eur J Cardiothorac Surg, 2001, 20: 385–390.
Rivera C, Jougon J, Dahan M, et al. Are postoperative consequences of neoadjuvant chemotherapy for non-small cell lung cancer more severe in elderly patients? Lung Cancer, 2012, 76: 216–221.
Gridelli C, Gallo C, Shepherd FA, et al. Gemcitabine plus vinorelbine compared with cisplatin plus vinorelbine or cisplatin plus gemcitabine for advanced non-small-cell lung cancer: a phase III trial of the Italian GEMVIN Investigators and the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol, 2003, 21: 3025–3034.
Sun ZH, Zheng HM, Zhang LY. Gemcitabine plus carboplatin used as induction regimen for elderly patients with locally advanced unresectable non-small cell lung cancer. Chinese-German J Clin Oncol, 2011, 10: 85–87.
Cai YG, Li M, Xie X. Clinical observation of docetaxel plus cisplatin versus gemcitabine plus cisplatin in the treatment of patients with advanced non-small-cell lung cancer. Chinese-German J Clin Oncol, 2011, 10: 472–475.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by a grant from the Research Foundation of Education Bureau of Hubei Province, China (No. B20112116).
Rights and permissions
About this article
Cite this article
Li, Q., Wang, J., Zhang, J. et al. Clinical analysis of preoperative induction chemotherapy with gemcitabine combined with cisplatin for locally advanced non-small cell lung cancer. Chin. -Ger. J. Clin. Oncol. 11, 266–270 (2012). https://doi.org/10.1007/s10330-012-0985-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10330-012-0985-x