Abstract
Objective
The aim of this study was to compare the efficacies and safeties of the combination of docetaxel-carboplatin with the combination of non docetaxel-carboplatin as first-line chemotherapy for advanced epithelial ovarian, primary peritoneal or fallopian tube cancers.
Methods
Relevant articles were identified from MEDLINE (1993–2010), EMBASE (1980–2010), MEDION, the Cochrane library, Science Citation Index Expanded databases, hand searching of reference lists from primary articles and reviews, conference abstracts and contact with experts in the field. The review included 5 relevant primary studies (1430 women). Data was extracted for study characteristics and quality. Bivariate random-effect model metaanalysis was used to estimate diagnostic accuracy of the various index tests. A quantitative meta-analysis was carried out by two reviewers based on the inclusion criteria from all available studies.
Results
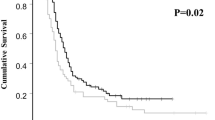
The frequency of the subgroup analysis of toxicity showed that toxicity action of combination of docetaxel-carboplatin was more severe than that of non docetaxel-carboplatin group (OR = 1.33, 95% CI = 1.13–1.56, P = 0.0005), whereas that of clinical responses was equivalent in comparison combination of docetaxel-carboplatin with combination of paclitaxel-carboplatin or docetaxel-cisplatin (OR = 1.0, 95% CI = 0.87–1.16, P = 0.95). There were heterogeneity (χ2 = 79.36, P < 0.00001) and inconsistency (83.6%) in toxicity analysis among the trials, while neither heterogeneity (χ2 = 3.21, P = 0.99) nor inconsistency (I 2 = 0%) in clinical responses among the trials.
Conclusion
The safety of combination of docetaxel-carboplatin is less than that of combination of paclitaxelcarboplatin or docetaxel-cisplatin. However, the clinical responses of combination of docetaxel-carboplatin are comparable with combination of paclitaxel-carboplatin or docetaxel-cisplatin.
Similar content being viewed by others
References
Decker DG, Fleming TR, Malkasian GD Jr, et al. Cyclophosphamide plus cis-platinum in combination: treatment program for stage III or IV ovarian carcinoma. Obstet Gynecol, 1982, 60: 481–487.
McGuire WP, Hoskins WJ, Brady MF, et al. Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. N Engl J Med, 1996, 334: 1–6.
Piccart MJ, Bertelsen K, James K, et al. Randomized intergroup trial of cisplatin-paclitaxel versus cisplatin-cyclophosphamide in women with advanced epithelial ovarian cancer: three-year results. J Natl Cancer Inst, 2000, 92: 699–708.
Ozols RF, Bundy BN, Greer BE, et al. Phase III trial of carboplatin and paclitaxel compared with cisplatin and paclitaxel in patients with optimally resected stage III ovarian cancer: a Gynecologic Oncology Group study. J Clin Oncol, 2003, 21: 3194–3200.
du Bois A, Lück HJ, Meier W, et al. A randomized clinical trial of cisplatin/paclitaxel versus carboplatin/paclitaxel as first-line treatment of ovarian cancer. J Natl Cancer Inst, 2003, 95: 1320–1329.
Neijt JP, Engelholm SA, Tuxen MK, et al. Exploratory phase III study of paclitaxel and cisplatin versus paclitaxel and carboplatin in advanced ovarian cancer. J Clin Oncol, 2000, 18: 3084–3092.
Gu MJ, Shi W. Current research and management of ovarian cancer in China. Chinese-German J Clin Oncol, 2002, 1: 212–218.
Vasey PA, Jayson GC, Gordon A, et al. Phase III randomized trial of docetaxel-carboplatin versus paclitaxel-carboplatin as first-line chemotherapy for ovarian carcinoma. J Natl Cancer Inst, 2004, 96: 1682–1691.
Hsu Y, Sood AK, Sorosky JI. Docetaxel versus paclitaxel for adjuvant treatment of ovarian cancer: case-control analysis of toxicity. Am J Clin Oncol, 2004, 27: 14–18.
Minagawa Y, Kigawa J, Kanamori Y, et al. Feasibility study comparing docetaxel-cisplatin versus docetaxel-carboplatin as first-line chemotherapy for ovarian cancer. Gynecol Oncol, 2006, 101: 495–498.
Mori T, Hosokawa K, Kinoshita Y, et al. A pilot study of docetaxelcarboplatin versus paclitaxel-carboplatin in Japanese patients with epithelial ovarian cancer. Int J Clin Oncol, 2007, 12: 205–211.
Markman J, Zanotti K, Webster K, et al. Experience with the management of neutropenia in gynecologic cancer patients receiving carboplatin-based chemotherapy. Gynecol Oncol, 2004, 92: 592–595.
Guastalla JP, Pujade-Lauraine E, Weber B, et al. Efficacy and safety of the paclitaxel and carboplatin combination in patients with previously treated advanced ovarian carcinoma. A multicenter GINECO (Group d’Investigateurs Nationaux pour l’Etude des Cancers Ovariens) phase II study. Ann Oncol, 1998, 9: 37–43.
McGuire WP 3rd, Markman M. Primary ovarian cancer chemotherapy: current standards of care. Br J Cancer, 2003, 89 Suppl 3: S3–8.
Kaye SB, Piccart M, Aapro M, et al. Phase II trials of docetaxel (Taxotere) in advanced ovarian cancer — an updated overview. Eur J Cancer, 1997, 33: 2167–2170.
Fung-Kee-Fung M, Oliver T, Elit L, et al. Optimal chemotherapy treatment for women with recurrent ovarian cancer. Curr Oncol, 2007, 14: 195–208.
Misawa H, Ochiai K, Yasuda M, et al. Combination chemotherapy with docetaxel and carboplatin for epithelial ovarian cancer. Nippon Rinsho (Japanese), 2004, 62Suppl 10: 546–549.
Furugen Y, Nishihara S, Kurokawa A, et al. Combination chemotherapy with docetaxel + carboplatin in the treatment of cancer of the ovary and fallopian tube. Gan To Kagaku Ryoho (Japanese), 2004, 31: 1205–1208.
Pfisterer J, du Bois A, Wagner U, et al. Docetaxel and carboplatin as first-line chemotherapy in patients with advanced gynecological tumors. A phase I/II trial of the Arbeitsgemeinschaft Gynäkologische Onkologie (AGO-OVAR) Ovarian Cancer Study Group. Gynecol Oncol, 2004, 92: 949–956.
Vasey PA, Atkinson R, Coleman R, et al. Docetaxel-carboplatin as first line chemotherapy for epithelial ovarian cancer. Br J Cancer, 2001, 84: 170–178.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, H., Rao, Y. & Qu, P. Combination of docetaxel-carboplatin for adjuvant chemotherapy of epithelial ovarian, primary peritoneal and fallopian tube cancers: a meta-analysis. Chin. -Ger. J. Clin. Oncol. 9, 475–481 (2010). https://doi.org/10.1007/s10330-010-0652-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10330-010-0652-z