Abstract
Objective
The aim of the study was to test the expressions of Livin and insulin-like growth factor II (IGF-II) in colorectal cancers and discuss their significance of carcinogenesis and progression in colorectal cancers, to provide a new target and theory basement for cancer therapy.
Methods
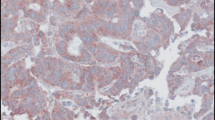
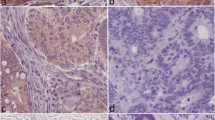
The expressions of Livin and IGF-II were detected by immunohistochemistry SABC in 60 cases of colorectal cancer and their associated colorectal tissues, and the associations between the expression levels of Livin and IGF-II and clinical pathological characteristics were analyzed.
Results
The positive rates of Livin and IGF-II in colorectal carcinomas were 68.33% and 63.33% respectively, higher than those of para-carcinomatous normal tissues and colorectal adenomas. There were no significant correlations between the expressions of Livin, IGF-II in colorectal carcinomas and the patient’s age, sex, tumor size and location, while distant metastasis, Dukes stage, histological type, lymph node metastasis, and whether the tumors have received radiotherapy and chemotherapy were significantly correlated with the expressions of Livin, IGF-II in colorectal carcinomas.
Conclusion
Livin and IGF-II play important roles in carcinogenesis and progression of colorectal cancer. This study showed obvious correlation between the expressions of Livin and IGF-II in colorectal cancers. It could be used as the reference for further researches. Livin is hopeful to be a new molecular target in cancer therapy.
Similar content being viewed by others
References
Kasof GM, Gomes BC. Livin, a novel inhibitor of apoptosis protein family member. Biol Chem, 2001, 276: 3238–3246.
Ashhab Y, Alian A, Polliack A, et al. Two splicing variants of a new inhibitor of apoptosis gene with different biological properties and tissue distribution pattern. FEBS Letters, 2001, 95: 56–60.
Nachmias B, Ashhab Y, Ben-yehuda D. The inhibitor of apoptosis protein family (IAPs): an emerging therapeutic target in cancer. Semin Cancer Biol, 2004, 14: 231–243.
Kempkenstefen C, Hinz S, Cluistoph F, et al. Expression of the apoptosis inhibitor Livin in renal cell carcinomas: correlations with pathology and outcome. Tumour Biol, 2007, 28: 132–136.
Xiang Y, Yao H, Wang S, et al. Prognostic value of survivin and Livin in nasopharyngeal carcinoma. Laryngoscope, 2006, 116: 126–129.
Sun JG, Liao RX, Chen ZT, et al. Gene transfection of Livin isoforms into A549 cell line and its effect on cell growth and sensitivity to chemotherapy and radiotherapy. Chin J Tuberc Respir Dis (Chinese), 2005, 28: 836–840.
Crnkovic-Mertons I, Hoppe-Seyler F, Butz K. Induction of apoptosis in tumor cells by siRNA-mediated silencing of the Livin/ML-IAP/KIAP gene. Oncogene, 2003, 22: 8330–8336.
Zhang Q, Xiong J, Jin Y, et al. CC chemokine ligand 25 enhances resistance to apoptosis in CD4+ T cells from patients with T-cell-lineage acute and chronic lymhocytie leukemia by means of Livin activation. Cancer Res, 2004, 64: 7579–7587.
Zhang Q, Xiong J, Jin Y, et al. Selectively frequent expression of CXCR5 enhances resistance to apoptosis in CD8+ CD34+ T cells from patients with T-cell-lineage acute lymphocytic leukemia. Oncogene, 2005, 24: 573–584.
Li HG, Chen YS, Chen G, et al. Expression of Livin in tissues of lung cancer and its correlation with the expression of caspase-3. Chinese-German J Clin Oncol, 2008, 7: 383–386.
Toretsky JA, Helman LJ. Involvement of IGF-II in human cancer. En docrinol, 1996, 149: 367–372.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bao, G., Zhang, C. & Wang, G. Expressions of Livin and IGF-II in colorectal carcinoma and their relationship. Chin. -Ger. J. Clin. Oncol. 9, 470–474 (2010). https://doi.org/10.1007/s10330-010-0636-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10330-010-0636-z