Abstract
Objective
New systemic therapies are needed to improve the prognosis of patients with advanced-stage hepatocellular carcinoma. The study was conducted to determine the efficacy and safety of viscum fraxini-2 in advanced Hepatocellular carcinoma.
Methods
A phase II study with a two-stage design that enrolled a total of 120 patients with chemotherapy naïve advanced hepatocellular carcinoma. The mistletoe preparation for the study is an aqueous injectable solution that contains one milliliter of viscum fraxini. Two ampoules of viscum fraxini were administered subcutaneously once weekly.
Results
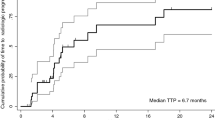
Chronic hepatitis C virus infection was the predominant cause of liver disease (60%) in studied cases. According to the response evaluation criteria in solid tumors 24 patients (20%) achieved objective response (including 2 complete responses) and 40 patients (33.3%) achieved stable disease. The median progression free survival for all patients was 4 months (range 1–28 months; 95% CI 3.3:4.7 months). The median overall survival for all patients was 8 months (range 1–28 months; 95% CI 6.3:9.7 months). The median survival for patients who achieved responsive or stable disease was 16 months. The toxicity was generally mild and well tolerated, mainly in the form of local reaction and fever. There were no drug related discontinuation or toxic deaths.
Conclusion
Viscum fraxini-2 is an effective, safe treatment for patients with advanced hepatocellular carcinoma. Further randomized controlled trials are recommended.
Similar content being viewed by others
References
Varela M, Sala M, Llovet JM, Bruix J. Treatment of hepatocellular carcinoma: is there an optimal strategy? Cancer Treat Rev, 2003, 29: 99–104.
Llovet JM, Fuster J, Bruix J. The Barcelona approach: diagnosis, staging, and treatment of hepatocellular carcinoma. Liver Transpl, 2004, 10: S115–120.
Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet, 2003, 362: 1907–1917.
Thomas MB. Systemic therapy for hepatocellular carcinoma. Cancer J, 2008, 14: 123–127.
Simonetti RG, Liberati A, Angiolini C, et al. Treatment of hepatocellular carcinoma: a systematic review of randomized controlled trials. Ann Oncol, 1997, 8: 117–136.
Mathurin P, Rixe O, Carbonell N, et al. Overview of medical treatments in unresectable hepatocellular carcinoma — an impossible meta-analysis? Aliment Pharmacol Ther, 1998, 12: 111–126.
Martin RC 2nd, Jarnagin WR. Randomized clinical trials in hepatocellular carcinoma and biliary cancer. Surg Oncol Clin N Am, 2002, 11: 193–205.
Llovet JM, Ricci S, Mazzaferro V, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med, 2008, 359: 378–390.
Becker H. Botany of European mistletoe (Viscum album L). Oncology, 1986, 43Suppl 1: 2–7.
Kienle GS, Berrino F, Bussing A, et al. Mistletoe in cancer — a systematic review on controlled clinical trials. Eur J Med Res, 2003, 8: 109–119.
Bussing A, Schietzel M. Apoptosis-inducing properties of Viscum album L. extracts from different host trees, correlate with their content of toxic mistletoe lectins. Anticancer Res, 1999, 19: 23–28.
Park WB, Lyu SY, Kim JH, et al. Inhibition of tumor growth and metastasis by Korean mistletoe lectin is associated with apoptosis and antiangiogenesis. Cancer Biother Radiopharm, 2001, 16: 439–447.
Kienle GS, Glockmann A, Schink M, et al. Extracts in breast and gynaecological cancers: a systematic review of clinical and preclinical research. J Exp Clin Cancer Res, 2009, 28: 79.
Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst, 2000, 92: 205–216.
Simon R. Optimal two-stage designs for phase II clinical trials. Control Clin Trials, 1989, 10: 1–10.
Mohmad NH, El-Zawahry HM, Mokbtar NM, et al. Review of epidemiologic and clinicopathologic features of 403 hepatocellular carcinoma patients. J Egyptian Nat Cancer Inst, 2000, 12: 87–93.
Abdel-Wahab M, El-Ghawalby N, Mostafa M, et al. Epidemiology of hepatocellular carcinoma in lower Egypt, Mansoura Gastroenterology Center. Hepatogastroenterology, 2007, 54: 157–162.
Elluru SR, VAN Huyen JP, Delignat S, et al. Antiangiogenic properties of viscum album extracts are associated with endothelial cytotoxicity. Anticancer Res, 2009, 29: 2945–2950.
Harmsma M, Gromme M, Ummelen M, et al. Ramaekers, Differential effects of Viscum album extract IscadorQu on cell cycle progression and apoptosis in cancer cells. Int J Oncol, 2004, 25: 1521–1529.
Sagar SM, Yance D, Wong RK. Natural health products that inhibit angiogenesis: a potential source for investigational new agents to treat cancer-Part 2. Curr Oncol, 2006, 13: 99–107.
Lyu SY, Choi SH, Park WB. Korean mistletoe lectin-induced apoptosis in hepatocarcinoma cells is associated with inhibition of telomerase via mitochondrial controlled pathway independent of p53. Arch Pharm Res, 2002, 25: 93–101.
Kim WH, Park WB, Gao B, et al. Critical role of reactive oxygen species and mitochondrial membrane potential in Korean mistletoe lectin-induced apoptosis in human hepatocarcinoma cells. Mol Pharmacol, 2004, 66: 1383–1396.
Harmsma M, Ummelen M, Dignef W, et al. Ramaekers, Effects of mistletoe (Viscum album L.) extracts Iscador on cell cycle and survival of tumor cells. Arzneimittelforschung, 2006, 56: 474–482.
Horneber MA, Bueschel G, Huber R, et al. Mistletoe therapy in oncology. Cochrane Database Syst Rev, 2008, 2: CD003297.
Chao Y, Chan WK, Birkhofer MJ, et al. Phase II and pharmacokinetic study of paclitaxel therapy for unresectable hepatocellular carcinoma patients. Br J Cancer, 1998, 78: 34–39.
Rougier P, Ducreux M, Kerr D, et al. A phase II study of raltitrexed (’Tomudex’) in patients with hepatocellular carcinoma. Ann Oncol, 1997, 8: 500–502.
Stuart K, Tessitore J, Rudy J, et al. A Phase II trial of nolatrexed dihydrochloride in patients with advanced hepatocellular carcinoma. Cancer, 1999, 86: 410–414.
Yeo W, Mok TS, Zee B, et al. A randomized phase III study of doxorubicin versus cisplatin/interferon alpha-2b/doxorubicin/fluorouracil (PIAF) combination chemotherapy for unresectable hepatocellular carcinoma. J Natl Cancer Inst, 2005, 97: 1532–1538.
Mabed M, El-Helw L, Shamaa S. Phase II study of viscum fraxini-2 in patients with advanced hepatocellular carcinoma. Br J Cancer, 2004, 90: 65–69.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ebrahim, M.A., El-Hadaad, H.A., Alemam, O.A. et al. Efficacy and safety of viscum fraxini-2 in advanced hepatocellular carcinoma: a phase II study. Chin. -Ger. J. Clin. Oncol. 9, 452–458 (2010). https://doi.org/10.1007/s10330-010-0629-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10330-010-0629-y