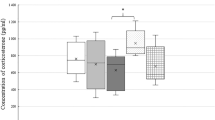
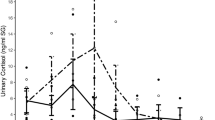
Abstract
Monitoring short-term changes of endocrine responses in non-human primates living in wild populations is a challenge. Saliva contains enzymes, steroids, and various analytes that can be broadly useful for helping us understand physiological responses to social and environmental sources of stress as well as other compromises to an individual’s health homeostasis. Salivary alpha-amylase and salivary cortisol are known to respond rapidly to stress, which can allow us to use these analytes to monitor stress on a time scale of minutes. We developed a non-invasive methodology for the collection of saliva and verified its applicability by conducting short-term interval sampling of focal individuals under varying social conditions in a group of semi-provisioned free-ranging Japanese macaques (Macaca fuscata) living endemically on the island of Koshima, Miyazaki prefecture, Japan. Monkeys were habituated to sampling by using ingestive attractants applied to cotton ropes. Their receptivity to chewing on the saliva sampling material provided and chewing time was recorded ad libitum during the initial habituation period of the troop to the sampling procedure. Focal animal behavior observations were conducted in conjunction with saliva sampling. Salivary analytes associated with stress, alpha-amylase and cortisol, were measured via enzyme immunoassay. Short-term changes in salivary alpha-amylase and salivary cortisol were examined in relation to short-term changes in social behavior, in particular aggression or grooming. We tested flow-rate effects in both analytes and found strong correlations between original sample results and their respective flow-rate transformed equivalents. Additionally, temperature effects on samples were tested and both analytes showed nearly the same values when stored at − 20, 4, and 30 °C conditions for 6 h; important information expected to facilitate future sampling in field conditions where freezer storage is uncertain. Our study shows that saliva can be repeatedly sampled non-invasively to investigate short-term changes in stress-associated markers in free-ranging Japanese macaques.






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author (NB) upon reasonable request.
References
Beltzer EK, Fortunato CK, Guaderrama MM, et al (2010) Salivary flow and alpha-amylase: collection technique, duration, and oral fluid type. Physiol Behav 101:289–296. https://doi.org/10.1016/j.physbeh.2010.05.016
Bosch JA (2014) The use of saliva markers in psychobiology: mechanisms and methods. In: Ligtenberg AJM, Veerman ECI (eds) Monogr Oral Sci. S. KARGER AG, Basel, pp 99–108
Bosch JA, Veerman ECI, de Geus EJ, Proctor GB (2011) α-Amylase as a reliable and convenient measure of sympathetic activity: don’t start salivating just yet! Psychoneuroendocrinology 36:449–453. https://doi.org/10.1016/j.psyneuen.2010.12.019
Broche N, Takeshita RSC, Mouri K et al. (2019) Salivary alpha-amylase enzyme is a non-invasive biomarker of acute stress in Japanese macaques (Macaca fuscata). Primates 60:547–558. https://doi.org/10.1007/s10329-019-00757-6
Chrousos GP (2009) Stress and disorders of the stress system. Nat Rev Endocrinol 5:374–381. https://doi.org/10.1038/nrendo.2009.106
Daniel M, Moore DS, Decker S et al. (2006) Associations among education, cortisol rhythm, and BMI in blue-collar women*. Obesity 14:327–335. https://doi.org/10.1038/oby.2006.42
Dorn LD, Kolko DJ, Susman EJ et al. (2009) Salivary gonadal and adrenal hormone differences in boys and girls with and without disruptive behavior disorders: contextual variants. Biol Psychol 81:31–39. https://doi.org/10.1016/j.biopsycho.2009.01.004
Eatough EM, Shirtcliff EA, Hanson JL, Pollak SD (2009) Hormonal reactivity to MRI scanning in adolescents. Psychoneuroendocrinology 34:1242–1246. https://doi.org/10.1016/j.psyneuen.2009.03.006
Garrett JR, Kidd A (1993) The innervation of salivary glands as revealed by morphological methods. Microsc Res Tech 26:75–91. https://doi.org/10.1002/jemt.1070260108
Godoy LD, Rossignoli MT, Delfino-Pereira P et al. (2018) A comprehensive overview on stress neurobiology: basic concepts and clinical implications. Front Behav Neurosci 12:127. https://doi.org/10.3389/fnbeh.2018.00127
Heintz MR, Santymire RM, Parr LA, Lonsdorf EV (2011) Validation of a cortisol enzyme immunoassay and characterization of salivary cortisol circadian rhythm in chimpanzees (Pan troglodytes). Am J Primatol 73:903–908. https://doi.org/10.1002/ajp.20960
Higham JP (2016) Field endocrinology of nonhuman primates: past, present, and future. Horm Behav 84:145–155. https://doi.org/10.1016/j.yhbeh.2016.07.001
Higham JP, Vitale AB, Rivera AM et al. (2010) Measuring salivary analytes from free-ranging monkeys. Physiol Behav 101:601–607. https://doi.org/10.1016/j.physbeh.2010.09.003
Inoue E, Inoue-Murayama M, Takenaka O, Nishida T (2007) Wild chimpanzee infant urine and saliva sampled noninvasively usable for DNA analyses. Primates 48:156–159. https://doi.org/10.1007/s10329-006-0017-y
Ishizuka S, Kawamoto Y, Toda K, Furuichi T (2019) Bonobos’ saliva remaining on the pith of terrestrial herbaceous vegetation can serve as non-invasive wild genetic resources. Primates 60:7–13. https://doi.org/10.1007/s10329-018-00704-x
Jacobsen N, Hensten-Pettersen A (1976) Biochemical characteristics of certain salivary glycoproteins from Cercopithecus aethiops. Arch Oral Biol 21:611–615. https://doi.org/10.1016/0003-9969(76)90031-5
Kaufman E, Lamster IB (2002) The diagnostic applications of saliva—a review. Crit Rev Oral Biol Med 13:197–212. https://doi.org/10.1177/154411130201300209
Koolhaas JM, Bartolomucci A, Buwalda B et al. (2011) Stress revisited: a critical evaluation of the stress concept. Neurosci Biobehav Rev 35:1291–1301. https://doi.org/10.1016/j.neubiorev.2011.02.003
Kuebler U, von Känel R, Heimgartner N, et al (2014) Norepinephrine infusion with and without alpha-adrenergic blockade by phentolamine increases salivary alpha amylase in healthy men. Psychoneuroendocrinology 49:290–298. https://doi.org/10.1016/j.psyneuen.2014.07.023
Lutz CK, Tiefenbacher S, Jorgensen MJ et al. (2000) Techniques for collecting saliva from awake, unrestrained, adult monkeys for cortisol assay. Am J Primatol 52:93–99. https://doi.org/10.1002/1098-2345(200010)52:2%3c93::AID-AJP3%3e3.0.CO;2-B
Mandalaywala TM, Petrullo LA, Parker KJ et al. (2017) Vigilance for threat accounts for inter-individual variation in physiological responses to adversity in rhesus macaques: a cognition × environment approach. Dev Psychobiol 59:1031–1038. https://doi.org/10.1002/dev.21572
McEwen BS, Akil H (2020) Revisiting the stress concept: implications for affective disorders. J Neurosci 40:12–21. https://doi.org/10.1523/JNEUROSCI.0733-19.2019
Nagato T, Tandler B (1986) Ultrastructure of the submandibular gland in 2 species of macaques. Cells Tiss Organs 126:255–262. https://doi.org/10.1159/000146228
Nater UM, Rohleder N (2009) Salivary alpha-amylase as a non-invasive biomarker for the sympathetic nervous system: current state of research. Psychoneuroendocrinology 34:486–496. https://doi.org/10.1016/j.psyneuen.2009.01.014
Nater UM, Rohleder N, Schlotz W et al. (2007) Determinants of the diurnal course of salivary alpha-amylase. Psychoneuroendocrinology 32:392–401. https://doi.org/10.1016/j.psyneuen.2007.02.007
Novak MA, Hamel AF, Kelly BJ et al. (2013) Stress, the HPA axis, and nonhuman primate well-being: a review. Appl Ani Behav Sci 143:135–149. https://doi.org/10.1016/j.applanim.2012.10.012
Paul JC, David JC (1958) Acetylcholine and adrenaline-noradrenaline sensitivity in denervated parotid gland of monkey. Indian J Psychol 2:437–445
Petrakova L, Boy K, Mittmann L, et al (2017) Salivary alpha-amylase and noradrenaline responses to corticotropinreleasing hormone administration in humans. Biol Psychol 127:34–39. https://doi.org/10.1016/j.biopsycho.2017.04.016
Petrullo LA, Mandalaywala TM, Parker KJ et al. (2016) Effects of early life adversity on cortisol/salivary alpha-amylase symmetry in free-ranging juvenile rhesus macaques. Horm Behav 86:78–84. https://doi.org/10.1016/j.yhbeh.2016.05.004
Proctor GB, Carpenter GH (2007) Regulation of salivary gland function by autonomic nerves. Auton Neurosci 133:3–18. https://doi.org/10.1016/j.autneu.2006.10.006
Quissell DO (1993) Steroid hormone analysis in human saliva. Ann NY Acad Sci 694:143–145. https://doi.org/10.1111/j.1749-6632.1993.tb18348.x
R Core Team. (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/.
Sapolsky RM, Romero LM, Munck AU (2000) How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions 1. Endocr Rev 21:55–89
Simons ND, Lorenz JG, Sheeran LK et al. (2012) Noninvasive saliva collection for DNA analyses from free-ranging Tibetan macaques (Macaca Thibetana): saliva collection from Tibetan macaques. Am J Primatol 74:1064–1070. https://doi.org/10.1002/ajp.22062
Smiley T, Spelman L, Lukasik-Braum M et al. (2010) Noninvasive saliva collection techniques for free-ranging mountain gorillas and captive eastern gorillas. J Zoo Wildl Med 41:201–209. https://doi.org/10.1638/2009-0015R.1
Smiley Evans T, Barry PA, Gilardi KV, et al (2015) Optimization of a novel non-invasive oral sampling technique for zoonotic pathogen surveillance in nonhuman primates. PLOS Negl Trop Dis 9:e0003813. https://doi.org/10.1371/journal.pntd.0003813
Spencer RL, Deak T (2017) A users guide to HPA axis research. Physiol Behav 178:43–65. https://doi.org/10.1016/j.physbeh.2016.11.014
Sugiyama Y (1976) Life History of Male Japanese Monkeys. In: Advances in the Study of Behavior. Elsevier, pp 255–284. https://doi.org/10.1016/S0065-3454(08)60169-2
Suzuki J, Ohkura S, Terao K (2002) Baseline and stress levels of cortisol in conscious and unrestrained Japanese macaques (Macaca fuscata). J Med Primatol 31:340–344. https://doi.org/10.1034/j.1600-0684.2002.01011.x
Thoma MV, Kirschbaum C, Wolf JM, Rohleder N (2012) Acute stress responses in salivary alpha-amylase predict increases of plasma norepinephrine. Biol Psychol 91:342–348. https://doi.org/10.1016/j.biopsycho.2012.07.008
Toyoda A, Matsudaira K, Maruhashi T et al. (2021) Highly versatile, non-invasive method for collecting buccal DNA from free-ranging non-human primates. J Trop Biol Conserv 18:251–267
van der Maarel MJEC, van der Veen B, Uitdehaag JCM et al. (2002) Properties and applications of starch-converting enzymes of the α-amylase family. J Biotechnol 94:137–155. https://doi.org/10.1016/S0168-1656(01)00407-2
Warren CM, van den Brink RL, Nieuwenhuis S, Bosch JA (2017) Norepinephrine transporter blocker atomoxetine increases salivary alpha amylase. Psychoneuroendocrinology 78:233–236. https://doi.org/10.1016/j.psyneuen.2017.01.029
Watanabe K (2008) A Review of 50 Years of Research on the Japanese monkeys of Koshima: status and dominance. In: Matsuzawa T (ed) Primate origins of human cognition and behavior. Springer Japan, Tokyo, pp 405–417
Acknowledgements
We thank the two anonymous reviewers for their constructive comments. Ikuma Adachi kindly assisted the study by providing needed field equipment. Financial support was provided by the Japanese Ministry of Education, Culture, Sports, Science and Technology (#160383 to NB), the Leading Graduate Program in Primatology and Wildlife Science (PWS) of Kyoto University, the Graduate School of Science, Kyoto University, and from the Kyoto University Wildlife Research Center Joint Use funding (No. 2020-B-3 to NB).
Funding
Japan Ministry of Education, Culture, Sports, Science and Technology, #160383, Nelson Broche
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The research described here was approved (permit no. 2019–001) by the Field Research Committee of the Center for the Evolutionary Origins of Human Behavior (formerly the Primate Research Institute), Kyoto University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Broche, N., Mouri, K., Suzumura, T. et al. Measuring short-term changes in stress-associated salivary analytes in free-ranging Japanese macaques (Macaca fuscata). Primates 64, 439–449 (2023). https://doi.org/10.1007/s10329-023-01067-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10329-023-01067-8