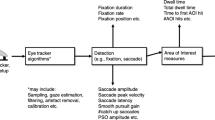
Abstract
Visual processing of the body movements of other animals is important for adaptive animal behaviors. It is widely known that animals can distinguish articulated animal movements even when they are just represented by points of light such that only information about biological motion is retained. However, the extent to which nonhuman great apes comprehend the underlying structural and physiological constraints affecting each moving body part, i.e., biomechanics, is still unclear. To address this, we examined the understanding of biomechanics in bonobos (Pan paniscus) and chimpanzees (Pan troglodytes), following a previous study on humans (Homo sapiens). Apes underwent eye tracking while viewing three-dimensional computer-generated (CG) animations of biomechanically possible or impossible elbow movements performed by a human, robot, or nonhuman ape. Overall, apes did not differentiate their gaze between possible and impossible movements of elbows. However, some apes looked at elbows for longer when viewing impossible vs. possible robot movements, which indicates that they may have had knowledge of biomechanics and that this knowledge could be extended to a novel agent. These mixed results make it difficult to draw a firm conclusion regarding the extent to which apes understand biomechanics. We discuss some methodological features that may be responsible for the results, as well as implications for future nonhuman animal studies involving the presentation of CG animations or measurement of gaze behaviors.






Similar content being viewed by others
Availability of data and material
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request. Note that relevant data are visualized in this published article (and in Supplementary file 1).
Code availability
The R code used during the current study is available from the corresponding author on reasonable request.
References
Avenanti A, Bolognini N, Maravita A, Aglioti SM (2007) Somatic and motor components of action simulation. Curr Biol 17:2129–2135. https://doi.org/10.1016/j.cub.2007.11.045
Bardi L, Regolin L, Simion F (2011) Biological motion preference in humans at birth: role of dynamic and configural properties. Dev Sci 14:353–359. https://doi.org/10.1111/j.1467-7687.2010.00985.x
Bardi L, Regolin L, Simion F (2014) The first time ever I saw your feet: inversion effect in newborns’ sensitivity to biological motion. Dev Psychol 50:986–993. https://doi.org/10.1037/a0034678
Bidet-Ildei C, Kitromilides E, Orliaguet J-P et al (2014) Preference for point-light human biological motion in newborns: contribution of translational displacement. Dev Psychol 50:113–120. https://doi.org/10.1037/a0032956
Blake R (1993) Cats perceive biological motion. Psychol Sci 4:54–57. https://doi.org/10.1111/j.1467-9280.1993.tb00557.x
Bradley MM, Sapigao RG, Lang PJ (2017) Sympathetic ANS modulation of pupil diameter in emotional scene perception: effects of hedonic content, brightness, and contrast. Psychophysiology 54:1419–1435. https://doi.org/10.1111/psyp.12890
Campbell MW, Carter JD, Proctor D et al (2009) Computer animations stimulate contagious yawning in chimpanzees. Proc R Soc B: Biol Sci 276:4255–4259. https://doi.org/10.1098/rspb.2009.1087
Chouinard-Thuly L, Gierszewski S, Rosenthal GG et al (2017) Technical and conceptual considerations for using animated stimuli in studies of animal behavior. Curr Zool 63:5–19. https://doi.org/10.1093/cz/zow104
Christie T, Slaughter V (2010) Movement contributes to infants’ recognition of the human form. Cognition 114:329–337. https://doi.org/10.1016/j.cognition.2009.10.004
Cliff N (1993) Dominance statistics: ordinal analyses to answer ordinal questions. Psychol Bull 114:494–509. https://doi.org/10.1037/0033-2909.114.3.494
Costantini M, Galati G, Ferretti A et al (2005) Neural systems underlying observation of humanly impossible movements: an fMRI study. Cereb Cortex 15:1761–1767. https://doi.org/10.1093/cercor/bhi053
Craighero L, Leo I, Umiltà C, Simion F (2011) Newborns’ preference for goal-directed actions. Cognition 120:26–32. https://doi.org/10.1016/j.cognition.2011.02.011
De Agrò M, Rößler DC, Kim K, Shamble PS (2021) Perception of biological motion in point-light displays by jumping spiders. bioRxiv. https://doi.org/10.1101/2021.02.24.432515
Errington TM, Tan FE, Lomax J et al (2018) Replication studies. Retrieved from osf.io/p7ayb
Feng D, Cliff N (1995) Comparisons of power and size of d and t statistics under normal and nonnormal assumptions. University of Southern California, Los Angeles (Unpublished manuscript)
Forbes SH (2020) PupillometryR: an R package for preparing and analysing pupillometry data. J Open Source Software 5:2285. https://doi.org/10.21105/joss.02285
Gao J, Tomonaga M (2020a) Body perception in chimpanzees (Pan troglodytes): the effect of body structure changes. J Comp Psychol 134:222–231. https://doi.org/10.1037/com0000214
Gao J, Tomonaga M (2020b) How chimpanzees and children perceive other species’ bodies: comparing the expert effect. Dev Sci. https://doi.org/10.1111/desc.12975
Gao J, Kawakami F, Tomonaga M (2020) Body perception in chimpanzees and humans: the expert effect. Sci Rep 10:7148. https://doi.org/10.1038/s41598-020-63876-x
Geangu E, Senna I, Croci E, Turati C (2015) The effect of biomechanical properties of motion on infants’ perception of goal-directed grasping actions. J Exp Child Psychol 129:55–67. https://doi.org/10.1016/j.jecp.2014.08.005
Hattori Y, Kano F, Tomonaga M (2010) Differential sensitivity to conspecific and allospecific cues in chimpanzees and humans: a comparative eye-tracking study. Biol Let 6:610–613. https://doi.org/10.1098/rsbl.2010.0120
Hepach R, Vaish A, Kano F, Albiach-Serrano A, Benziad L, Call J, Tomasello M (2021) Chimpanzees’ (Pan troglodytes) internal arousal remains elevated if they cannot themselves help a conspecific. J Comp Psychol 135:196–207
Hess MR, Kromrey JD, Ferron JM et al (2005) Robust inference in meta-analysis: an empirical comparison of point and interval estimates using the standardized mean difference and cliff’s delta. Annual Meeting of the American Educational Research Association. Montreal, p 11−15
Hirata S, Fuwa K, Sugama K et al (2010) Facial perception of conspecifics: chimpanzees (Pan troglodytes) preferentially attend to proper orientation and open eyes. Anim Cogn 13:679–688. https://doi.org/10.1007/s10071-010-0316-y
Hopper LM, Gulli RA, Howard LH et al (2020) The application of noninvasive, restraint-free eye-tracking methods for use with nonhuman primates. Behav Res. https://doi.org/10.3758/s13428-020-01465-6
Hothorn T, Hornik K (2019) exactRankTests: exact distribution for rank and permutation tests. R package version 0.8–31. https://CRAN.R-project.org/package=exactRankTests
Iacoboni M, Woods RP, Brass M et al (1999) Cortical mechanisms of human imitation. Science 286:2526–2528. https://doi.org/10.1126/science.286.5449.2526
Iorns E, Tsui R, Denis A et al (2019) Meta-analysis. Retrieved from osf.io/c69jx
Johansson G (1973) Visual perception of biological motion and a model for its analysis. Percept Psychophys 14:201–211. https://doi.org/10.3758/BF03212378
Kano F, Call J (2014) Cross-species variation in gaze following and conspecific preference among great apes, human infants and adults. Anim Behav 91:137–150. https://doi.org/10.1016/j.anbehav.2014.03.011
Kano F, Call J (2017) Great ape social attention. In: Watanabe S, Hofman M, Shimizu T (eds) Evolution of the brain, cognition, and emotion in vertebrates. Springer, Tokyo, pp 187–206. https://doi.org/10.1007/978-4-431-56559-8_9
Kano F, Tomonaga M (2009) How chimpanzees look at pictures: a comparative eye-tracking study. Proc R Soc B: Biol Sci 276:1949–1955. https://doi.org/10.1098/rspb.2008.1811
Kano F, Hirata S, Call J, Tomonaga M (2011) The visual strategy specific to humans among hominids: a study using the gap–overlap paradigm. Vis Res 51:2348–2355. https://doi.org/10.1016/J.VISRES.2011.09.006
Kim J, Afshari A, Sengupta R et al (2018) Replication study: melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Elife 7:e39944. https://doi.org/10.7554/eLife.39944
Komori N, Katayama J, Kitazaki M, Itakura S (2006) Do infants prefer possible human movements? The 5th International Conference of the Cognitive Science. Vancouver, pp 143–144
Kraskov A, Dancause N, Quallo MM et al (2009) Corticospinal neurons in macaque ventral premotor cortex with mirror properties: a potential mechanism for action suppression? Neuron 64:922–930. https://doi.org/10.1016/j.neuron.2009.12.010
Kret ME, Prochazkova E, Sterck EHM, Clay Z (2020) Emotional expressions in human and non-human great apes. Neurosci Biobehav Rev. https://doi.org/10.1016/j.neubiorev.2020.01.027
Kromrey JD, Hogarty KY, Ferron JM et al (2005) Robustness in meta-analysis: an empirical comparison of point and interval estimates of standardized mean differences and Cliff’s delta. Joint Statistical Meetings. Minneapolis, pp 7−11
Long JD, Feng D, Cliff N (2003) Ordinal analysis of behavioral data. In: Schinka JA, Velicer WF (eds) Handbook of psychology: research methods in psychology, vol 2. Wiley, Hoboken, NJ, pp 635–661. https://doi.org/10.1002/0471264385.wei0225
Longhi E, Senna I, Bolognini N et al (2015) Discrimination of biomechanically possible and impossible hand movements at birth. Child Dev 86:632–641. https://doi.org/10.1111/cdev.12329
Massen JJM, Behrens F, Martin JS et al (2019) A comparative approach to affect and cooperation. Neurosci Biobehav Rev 107:370–387. https://doi.org/10.1016/j.neubiorev.2019.09.027
Matsuno T, Fujita K (2018) Body inversion effect in monkeys. PLoS ONE 13:e0204353. https://doi.org/10.1371/journal.pone.0204353
Mori M (1970) Bukimi no tani (the uncanny valley). Energy 7:33–35
Mori M, MacDorman KF, Kageki N (2012) The uncanny valley (from the field). IEEE Robot Autom Magaz 19:98–100. https://doi.org/10.1109/MRA.2012.2192811
Morita T, Katayama N, Kitazaki M, Itakura S (2010) Development of perception of human and robot body movement. J Robot Soc Japan 28:463–469. https://doi.org/10.7210/jrsj.28.463
Morita T, Slaughter V, Katayama N et al (2012) Infant and adult perceptions of possible and impossible body movements: an eye-tracking study. J Exp Child Psychol 113:401–414. https://doi.org/10.1016/j.jecp.2012.07.003
Nakayasu T, Watanabe E (2014) Biological motion stimuli are attractive to medaka fish. Anim Cogn 17:559–575. https://doi.org/10.1007/s10071-013-0687-y
Nakayasu T, Yasugi M, Shiraishi S et al (2017) Three-dimensional computer graphic animations for studying social approach behaviour in medaka fish: effects of systematic manipulation of morphological and motion cues. PLoS ONE 12:e0175059. https://doi.org/10.1371/journal.pone.0175059
Orquin JL, Holmqvist K (2018) Threats to the validity of eye-movement research in psychology. Behav Res 50:1645–1656. https://doi.org/10.3758/s13428-017-0998-z
Papadourakis V, Raos V (2017) Evidence for the representation of movement kinematics in the discharge of F5 mirror neurons during the observation of transitive and intransitive actions. J Neurophysiol 118:3215–3229. https://doi.org/10.1152/jn.00816.2016
Papadourakis V, Raos V (2019) Neurons in the macaque dorsal premotor cortex respond to execution and observation of actions. Cereb Cortex 29:4223–4237. https://doi.org/10.1093/cercor/bhy304
Peinkhofer C, Knudsen GM, Moretti R, Kondziella D (2019) Cortical modulation of pupillary function: systematic review. PeerJ 7:e6882. https://doi.org/10.7717/peerj.6882
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Regolin L, Tommasi L, Vallortigara G (2000) Visual perception of biological motion in newly hatched chicks as revealed by an imprinting procedure. Anim Cogn 3:53–60. https://doi.org/10.1007/s100710050050
Reid V, Belsky J, Johnson M (2005) Infant perception of human action: toward a developmental cognitive neuroscience of individual differences. Cogn Brain Behav 9:193–210
Reid VM, Hoehl S, Landt J, Striano T (2008) Human infants dissociate structural and dynamic information in biological motion: evidence from neural systems. Soc Cogn Affect Neurosci 3:161–167. https://doi.org/10.1093/scan/nsn008
Riečanský I, Lamm C (2019) The role of sensorimotor processes in pain empathy. Brain Topogr 32:965–976. https://doi.org/10.1007/s10548-019-00738-4
Rizzolatti G, Craighero L (2004) The mirror-neuron system. Annu Rev Neurosci 27:169–192. https://doi.org/10.1146/annurev.neuro.27.070203.144230
Rizzolatti G, Sinigaglia C (2010) The functional role of the parieto-frontal mirror circuit: interpretations and misinterpretations. Nat Rev Neurosci 11:264–274. https://doi.org/10.1038/nrn2805
Rizzolatti G, Cattaneo L, Fabbri-Destro M, Rozzi S (2014) Cortical mechanisms underlying the organization of goal-directed actions and mirror neuron-based action understanding. Physiol Rev 94:655–706. https://doi.org/10.1152/physrev.00009.2013
Rogmann JJ (2013) orddom: ordinal dominance statistics. R package version 3.1. https://CRAN.R-project.org/package=orddom
Romani M, Cesari P, Urgesi C et al (2005) Motor facilitation of the human cortico-spinal system during observation of bio-mechanically impossible movements. Neuroimage 26:755–763. https://doi.org/10.1016/j.neuroimage.2005.02.027
Senna I, Addabbo M, Bolognini N et al (2017) Infants’ visual recognition of pincer grip emerges between 9 and 12 months of age. Infancy 22:389–402. https://doi.org/10.1111/infa.12163
Simion F, Regolin L, Bulf H (2008) A predisposition for biological motion in the newborn baby. Proc Natl Acad Sci 105:809–813. https://doi.org/10.1073/pnas.0707021105
Southgate V, Johnson MH, Csibra G (2008) Infants attribute goals even to biomechanically impossible actions. Cognition 107:1059–1069
Steckenfinger SA, Ghazanfar AA (2009) Monkey visual behavior falls into the uncanny valley. Proc Natl Acad Sci 106:18362–18366. https://doi.org/10.1073/pnas.0910063106
Tomonaga M (2001) Visual search for biological motion patterns in chimpanzees (Pan troglodytes). Psychologia 644:46–59
Vallortigara G, Regolin L (2006) Gravity bias in the interpretation of biological motion by inexperienced chicks. Curr Biol 16:R279–R280. https://doi.org/10.1016/j.cub.2006.03.052
Vallortigara G, Regolin L, Marconato F (2005) Visually inexperienced chicks exhibit spontaneous preference for biological motion patterns. PLoS Biol 3:e208. https://doi.org/10.1371/journal.pbio.0030208
van der Meer AL (1997) Keeping the arm in the limelight: advanced visual control of arm movements in neonates. Eur J Paediatr Neurol 1:103–108. https://doi.org/10.1016/S1090-3798(97)80040-2
van der Meer AL, van der Weel FR, Lee DN (1995) The functional significance of arm movements in neonates. Science 267:693–695. https://doi.org/10.1126/science.7839147
Viechtbauer W (2010) Conducting meta-analyses in R with the metafor package. J Stat Soft 36:1–48. https://doi.org/10.18637/jss.v036.i03
Watanabe S, Troje NF (2006) Towards a “virtual pigeon”: a new technique for investigating avian social perception. Anim Cogn 9:271–279. https://doi.org/10.1007/s10071-006-0048-1
White B, Castle P, Held R (1964) Observations on the development of visually-directed reaching. Child Dev 35:349–364. https://doi.org/10.2307/1126701
Yoshida H, Smith LB (2008) What’s in view for toddlers? Using a head camera to study visual experience. Infancy 13:229–248. https://doi.org/10.1080/15250000802004437
Acknowledgments
We would like to especially thank Mr. H. Ishimoto and Mr. K. Nagamachi at the Department of Computer Science and Engineering, Toyohashi University of Technology, for making the high-quality 3D animations. Further, we are grateful to Dr. N. Morimura, Dr. F. Kano, and all the staff at KS; Ms. H. Bando, Mr. M. Sato, Dr. Y. Yamanashi, and Dr. M. Tanaka at KCZ; and Ms. E. Ichino, Dr. Y. Kawaguchi, and members of the Language & Intelligence section of the PRI, for their kind help in conducting the current study. We also thank Dr. T. Matsuzawa for financial support. We thank Sydney Koke, MFA, from Edanz (https://jp.edanz.com/ac), for editing a draft of this manuscript. Finally, we are grateful to the editor and anonymous reviewers for their constructive feedback during the review process.
Funding
This study was supported financially by the Japan Society for the Promotion of Science, Ministry of Education, Culture, Sports, Science and Technology, to YS (grant no. 19J22889), MT (grant nos. 15H05709, 18H04194), SH (grant nos. 26245069, 18H05524), and TM (grant no. 16H06283, LDG-U04); the Cooperative Research Program of the PRI; and the Great Ape Information Network. These funding sources had no involvement in the study design, collection, analysis and interpretation of data, manuscript writing, or in the decision to submit the article.
Author information
Authors and Affiliations
Contributions
Conceptualization: YS, MK, SI, SH. Methodology: MK. Formal analysis, investigation, data curation, writing—original draft, and visualization: YS. Resources: MK, TM, YS, MT, SH. Writing—review and editing: MK, SI, TM, YS, MT, SH. Supervision: SI, SH. Project administration: SH. Funding acquisition: SH, YS.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
The animal husbandry and research protocols complied with the Guide for Animal Research Ethics of the Wildlife Research Center, Kyoto University for the KS experiments (chimpanzees, no. WRC-2018-KS006A; bonobos, no. WRC-2018-KS007A), the Research Ethics Guideline of KCZ for the KCZ experiments (no. 2019-KCZ-001), and the Guidelines for the Care and Use of Laboratory Primates of the PRI for the PRI experiments (no. 2019-001).
Consent to participate
Not applicable.
Consent for publication
All authors approved the submission of the article. The funding sources had no involvement in the decision to submit this article for submission.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Supplementary file2 (AVI 590553 KB)
Supplementary file3 (AVI 590544 KB)
Supplementary file4 (AVI 599677 KB)
Supplementary file5 (AVI 630122 KB)
Supplementary file6 (AVI 651423 KB)
Supplementary file7 (AVI 602722 KB)
Supplementary file8 (AVI 663601 KB)
Supplementary file9 (AVI 602722 KB)
About this article
Cite this article
Sato, Y., Kitazaki, M., Itakura, S. et al. Great apes’ understanding of biomechanics: eye-tracking experiments using three-dimensional computer-generated animations. Primates 62, 735–747 (2021). https://doi.org/10.1007/s10329-021-00932-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10329-021-00932-8