Abstract
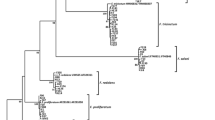
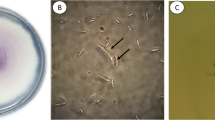
Lotus rhizome rot is one of the most important diseases affecting lotus production, leading to heavy economic losses in Japan. In this study, a causal agent of lotus rhizome rot in Yamaguchi prefecture, Japan, were identified as Fusarium commune based on their morphology and nucleotide sequences of EF-1α, HistonH3, and mtSSU. The inoculation tests showed that plants inoculated with F. commune isolated from lotus were more affected than those inoculated with strains from other hosts (tomato, turnip, and komatsuna) and soil in wheat fields. This result shows that particular strains of F. commune have virulence for lotus plants. Whole genome sequencing of the lotus-infecting F. commune revealed 20 effector candidates, including SIX4 and SIX9 homologs. Of these genes, 10 were expressed only in the roots of lotus but not in minimal medium. In particular, g3613 was specifically maintained in lotus-infecting F. commune, suggesting that this gene may be one of the candidate determinants of the host specificity of F. commune for lotus plants.


Similar content being viewed by others
References
Armstrong GM, Armstrong JK (1981) Formae speciales and race of Fusarium oxysporum causing wilt diseases. In: Nelson PE, Toussoun TA, Cook RJ (eds) Fusarium: diseases, biology, and taxonomy. Pennsylvania State University Press, University Park, pp 391–399
Deng S, Ma X, Chen Y, Feng H, Zhou D, Wang X, Zhang Y, Zhao M, Zhang J, Daly P, Wei L (2022) LAMP assay for distinguishing Fusarium oxysporum and Fusarium commune in lotus (Nelumbo nucifera) rhizomes. Plant Dis 106:231–246
Enya J, Togawa M, Takeuchi T, Yoshida S, Tsushima S, Arie T, Sakai T (2008) Biological and phylogenetic characterization of Fusarium oxysporum complex, which causes yellows on Brassica spp., and proposal of F. oxysporum f. sp. rapae, a novel forma specialis pathogenic on B. rapa in Japan. Phytopathology 98:475–483
FerraraM HM, Logrieco AF, Leslie JF, Mulè G (2019) A CRISPR-Cas9 system for genome editing of Fusarium proliferatum. Sci Rep 9:1–9
Gawehns F, Houterman PM, Ichou FA, Michielse CB, Hijdra M, Cornelissen BJC, Rep M, Takken FLW (2014) The Fusarium oxysporum effector Six6 contributes to virulence and suppresses I-2-mediated cell death. Mol Plant Microbe Interact 27:336–348
Glass NL, Donaldson GC (1995) Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Appl Environl Microbiol 61:1323–1330
Hamini-Kadar N, Edel-Hermann V, Gautheron N, Steinberg C (2010) First report of Fusarium commune and Fusarium redolens causing crown and root rot on tomato in Algeria. New Dis Rep 22:3
He Y, Chen J, Tang C, Deng Q, Guo L, Cheng Y, Li Z, Wang T, Xu J, Gao C (2022) Genetic diversity and population structure of Fusarium commune causing strawberry root rot in Southcentral China. Genes 13:899
Houterman PM, Cornelissen BJ, Rep M (2008) Suppression of plant resistance gene-based immunity by a fungal effector. PLoS Pathog 4:e1000061
Houterman P, Ma L, Van Ooijen G, De Vroomen MJ, Cornelissen BJ, Takken FL, Rep M (2009) The effector protein Avr2 of the xylem-colonizing fungus Fusarium oxysporum activates the tomato resistance protein I-2 intracellularly. Plant J 58:970–978
Huang Y, Busk PK, Lange L (2015) Cellulose and hemicellulose-degrading enzymes in Fusarium commune transcriptome and functional characterization of three identified xylanases. Enzyme Microb 73:9–19
Husna A, Zakaria L, Mohamed Nor NM (2021) Fusarium commune associated with wilt and root rot disease in rice. Plant Pathol 70:123–132
Ito S, Takahara H, Kawaguchi T, Tanaka S, Kameya-Iwaki M (2002) Post-transcriptional silencing of the tomatinase gene in Fusarium oxysporum f. sp lycopersici. J Phytopathol 150:74–480
Izuho M, Sumida Y, Ueki A, Ishioka G, Sasaki K, Mori S, Aoki H, Takehara T (2021) Mechanism of Rhizome rot pathogenesis in lotus root and its control by anaerobic soil disinfestation. Bull Yamaguchi Agri for Gen Tech Ctr 12:61–74 ((in Japanese))
Kashiwa T, Inami K, Fujinaga M, Ogiso H, Yoshida T, Teraoka T, Arie T (2013) An avirulence gene homologue in the tomato wilt fungus Fusarium oxysporum f. sp. lycopersici race 1 functions as a virulence gene in the cabbage yellows fungus F. oxysporum f. sp. conglutinans. J Gen Plant Pathol 79:412–421
Kershaw MJ, Talbot NJ (2009) Genome-wide functional analysis reveals that infection-associated fungal autophagy is necessary for rice blast disease. Proc Natl Acad Sci 106:15967–15972
Komada H (1975) Development of a selective medium for quantitative isolation of Fusarium oxysporum from natural soil. Rev Plant Prot Res 8:114–125
Kuang W, Zhang L, Ye L, Ma J, Shi X, Lin Y, Sun X, Cui R (2022) Genome and transcriptome sequencing analysis of Fusarium commune provides insights into the pathogenic mechanisms of the lotus rhizome rot. Microbiol Spectr 10:e00175-e222
Leslie JF, Summerell BA (2006) The Fusarium laboratory manual. Blackwell Publishing, Ames
Li J, Li H, Zheng L, Yan SL, Wang QZ (2016) First report of lotus root disease caused by Fusarium tricinctum in China. Plant Dis 100:1784–1784
Ma L, Houterman PM, Gawehns F, Cao L, Sillo F, Richter H, Clavijo-Ortiz MJ, Schmidt SM, Boeren S, Vervoort J, Cornelissen BJ, Rep M, Takken FL (2015) The AVR 2–SIX 5 gene pair is required to activate I-2-mediated immunity in tomato. New Phytol 208:507–518
Meisel L, Fonseca B, González S, Baeza-Yates R, Cambiazo V, Campos R, Gonzalez M, Orellana A, Retamales J, Silva H (2005) A rapid and efficient method for purifying high quality total RNA from peaches (Prunus persica) for functional genomics analyses. Biol Res 38:83–88
Ninomiya Y, Suzuki K, Ishii C, Inoue H (2004) Highly efficient gene replacements in Neurospora strains deficient for nonhomologous end-joining. Proc Natl Acad Sci 101:12248–12253
Nishikado Y, Watanabe K (1953) On the rhizome rot of lotus, Nelumbo nucifera Gaertn., caused by a new Fusarium, F. bulbigenum WR. Nelumbicolum Nis. et Wat. Berichte Des Ohara Instituts Fur Landwirschaftliche Forschungen 10:1–8
O’Donnell K, Kistler HC, Cigelnik E, Ploetz RC (1998) Multiple evolutionary origins of the fungus causing Panama disease of banana: concordant evidence from nuclear and mitochondrial gene genealogies. Proc Natl Acad Sci 95:2044–2049
Petersen TN, Brunak S, Von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786
Rep M, Van Der Does HC, Meijer M, Van Wijk R, Houterman PM, Dekker HL, De Koster CG, Cornelissen BJ (2004) A small, cysteine-rich protein secreted by Fusarium oxysporum during colonization of xylem vessels is required for I-3-mediated resistance in tomato. Mol Microbiol 53:1373–1383
Sasaki K, Nakahara K, Tanaka S, Shigyo M, Ito S (2015) Genetic and pathogenic variability of Fusarium oxysporum f. sp. cepae isolated from onion and Welsh onion in Japan. Phytopathology 105:525–532
Shen W, Le S, Li Y, Hu F (2016) SeqKit: a cross-platform and ultrafast toolkit for FASTA/Q file manipulation. PLoS ONE 11:e0163962
Skovgaard K, Rosendahl S, O’Donnell K, Nirenberg HI (2003) Fusarium commune is a new species identified by morphological and molecular phylogenetic data. Mycologia 95:630–636
Stewart JE, Kim MS, James RL, Dumroese RK, Klopfenstein NB (2006) Molecular characterization of Fusarium oxysporum and Fusarium commune isolates from a conifer nursery. Phytopathology 96:1124–1133
Thatcher LF, Gardiner DM, Kazan K, Manners JM (2012) A highly conserved effector in Fusarium oxysporum is required for full virulence on Arabidopsis. Mol Plant Microbe Interact 25:180–190
Ushimaru T, Terada H, Tsuboi K, Kogou Y, Sakaguchi A, Tsuji G, Kubo Y (2010) Development of an efficient gene targeting system in Colletotrichum higginsianum using a non-homologous end-joining mutant and Agrobacterium tumefaciens-mediated gene transfer. Mol Genet Genom 284:357–371
Yin X, Li XZ, Yin JJ, Wu X (2016) First report of Phytopythium helicoides causing rhizome rot of Asian lotus in China. Plant Dis 100:532
You JS, Lee YJ, Kim KS, Kim SH, Chang KJ (2014) Ethanol extract of lotus (Nelumbo nucifera) root exhibits an anti-adipogenic effect in human pre-adipocytes and anti-obesity and anti-oxidant effects in rats fed a high-fat diet. Nutr Res 34:258–267
Yu JM, Babadoost M (2013) Occurrence of Fusarium commune and F. oxysporum in horseradish roots. Plant Dis 97:453–460
Zaccardelli M, Vitale S, Luongo L, Merighi M, Corazza L (2008) Morphological and molecular characterization of Fusarium solani isolates. J Phytopathol 156:534–541
Zeng LS, Zheng ZB, Lu S, Xia L, Deng ZP, Hu GY, Du CX, Mai JP, Zhou JK (2017) Identification of pathogen causing rhizome rot of lotus (Nelumbo nucifera)based on morphology and multi-gene sequence analysis. Acta Phytopathologia Sin 47:163–173
Zhang M, Liu Q, Yang X, Xu J, Liu G, Yao X, Ren R, Xu J, Lou L (2020) CRISPR/Cas9-mediated mutagenesis of Clpsk1 in watermelon to confer resistance to Fusarium oxysporum f. sp. niveum. Plant Cell Rep 39:589–595
Acknowledgements
We thank Ms. Keiko Shinohara, Tokushima Agriculture, Forestry and Fisheries Technology Support Center, for kindly providing F. commune NR-200710001 strain.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Watanabe, A., Miura, Y., Sakane, K. et al. Identification and characterization of Fusarium commune, a causal agent of lotus rhizome rot. J Gen Plant Pathol 89, 170–178 (2023). https://doi.org/10.1007/s10327-023-01119-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10327-023-01119-3