Abstract
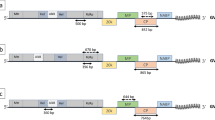
In DAS-ELISAs of 86 grapevine samples from northwestern Iran, Grapevine fanleaf virus (GFLV) was detected in 18 samples. RT-PCR with two primer pairs (M2/M4 or M0/M4) corresponding to GFLV movement protein (MP) amplified the expected 854- and/or 1,489-bp fragment(s), respectively, from all ELISA-positive samples. Four smaller and three larger PCR products were cloned and sequenced, which revealed that the MP region of the isolates was 1,044 nucleotides (nt) long, corresponding to the GFLV MP. There were 83–86% nucleotide and 93–94% amino acid identities deduced between the MPs of the sequenced isolates. Nucleotide sequence identities of 81–87 and 75–79% were found between the MP regions of these isolates and that of previously published GFLV and Arabis mosaic virus (ArMV) strains/isolates, respectively. On a consensus parsimony tree based on the nucleotide sequences, isolates La208 and X300 remained distinct from previously reported GFLVs. This is the first molecular characterization of GFLV MP isolates from Iran.


Similar content being viewed by others
References
Andret-Link C, Laporte C, Valet L, Ritzenthaler C, Demangeat G, Vigne E, Laval V, Pfeiffer P, Stussi-Garaud C, Fuchs M (2004) Grapevine fanleaf virus: still a major threat to the grapevine industry. J Plant Pathol 86:183–195
Anfoka GH, Shahrour W, Nakhla MK (2004) Detection and molecular characterization of Grapevine fanleaf virus and Grapevine leafroll-associated virus 3 in Jordan. J Plant Pathol 86:203–207
Bashir NS, Kalhor MR, Zarghani SN (2006) Detection, differentiation and phylogenetic analysis of Cucumber mosaic virus isolates from cucurbits in the northwest region of Iran. Virus Gene 32:277–288
Beachy RN (1995) Movement protein-mediated resistance. In: Proceedings of USDA-APHIS workshop on transgenic virus-resistant plants and new plant viruses, Maryland, pp 21–23
Clark MF, Adams AN (1977) Characteristic of the microplate method of enzyme-linked immunosorbent assay for the detection of plant viruses. J Gen Virol 34:475–483
Felsenstein J (2004) PHYLIP (Phylogeny Inference Package) version 3.6. Distributed by the author. Department of Genome Sciences, University of Washington, Seattle http://www.evolution.gs.washington.edu/phylip.html
ICTVdB—The Universal Virus Database, version 4, April 2006 [online]. http://www.ncbi.nlm.nih.gov/ICTVdb/ICTVdB/
Izadpanah K, Zaki-Aghl M, Zhang YP, Daubert SD, Rowhani A (2003) Bermuda grass as a potential reservoir host for Grapevine fanleaf virus. Plant Dis 87:1179–1182
Little A, Rezaian MA (2003) Grapevine viroids. In: Hadidi A, Flores R, Randles JW, Semancik JS (eds) Viroids. CSIRO, Victoria, Australia, pp 195–206
Martelli GP, Savino V (1990) Fanleaf degeneration. In: Pearson RC, Goheen A (eds) Compendium of grape diseases. APS, St Paul, MN, pp 48–89
Melcher U (2000) The ‘30K’ superfamily of viral movement proteins. J Gen Virol 81:257–266
Naraghi-Arani P, Daubert S, Rowhani A (2001) Quasispecies nature of the genome of Grapevine fanleaf virus. J Gen Virol 82:1791–1795
Nicholas KB, Nicholas HB Jr (1997) GeneDoc: a tool for editing and annotating multiple sequence alignments. Distributed by the authors
Raski DJ, Goheen AC, Lider LA, Meredith CP (1983) Strategies against grapevine fanleaf virus and its nematode vector. Plant Dis 67:335–337
Rowhani A, Chay C, Golino DA, Falk W (1993) Development of a polymerase chain reaction technique for the detection of Grapevine fanleaf virus in grapevine tissue. Phytopathology 83:749–753
Tanahashi T, Kita M, Kodama T, Sawai N, Yamaoka Y, Mitsufuji S, Katoh F, Imanishi J (2000) Comparison of PCR-restriction fragment length polymorphism analysis and PCR-direct sequencing methods for differentiating Helicobacter pylori ureB gene variants. J Clin Mirobiol 38:165–169
Vigne E, Bergdoll M, Guyader S, Fuchs M (2004a) Population structure and genetic variability within isolates of Grapevine fanleaf virus from a naturally infected vineyard in France: evidence for mixed infection and recombination. J Gen Virol 85:2435–2445
Vigne E, Komar V, Fuchs M (2004b) Field safety assessment of recombination in transgenic grapevines expressing the coat protein gene of Grapevine fanleaf virus. Transgenic Res 13:165–179
Vigne E, Demangeat G, Komar V, Fuchs M (2005) Characterization of a naturally occurring recombinant isolate of Grapevine fanleaf virus. Arch Virol 150:2241–2255
Wetzel T, Meunier L, Jaeger U, Reustle GM, Krczal G (2001) Complete nucleotide sequences of the RNAs 2 of German isolates of grapevine fanleaf and Arabis mosaic nepoviruses. Virus Res 75:139–145
Wetzel T, Jardak R, Meunier L, Ghorbel A, Reustle GM, Krczal G (2002) Simultaneous RT/PCR detection and differentiation of arabis mosaic and grapevine fanleaf nepoviruses in grapevines with a single pair of primers. J Virol Methods 101:63–69
Acknowledgments
Thanks to Vice Chancellor for Research Affairs of University of Tabriz for funding, to Dr Thierry Wetzel for the primers, to Mr Ismael Torabi for technical assistance and to Dr Sassan Asgari for reviewing this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
The sequence data reported in this paper have been submitted to the DDBJ/EMBL/GenBank databases and have been assigned accession numbers DQ286901 to DQ286916.
Rights and permissions
About this article
Cite this article
Bashir, N.S., Nikkhah, S. & Hajizadeh, M. Distinct phylogenetic positions of Grapevine fanleaf virus isolates from Iran based on the movement protein gene. J Gen Plant Pathol 73, 209–215 (2007). https://doi.org/10.1007/s10327-007-0012-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10327-007-0012-1