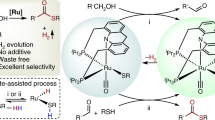
Abstract
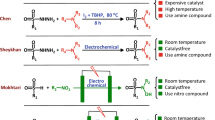
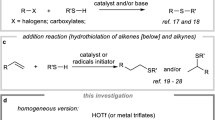
Catalytic dehydrogenative coupling of thiols with amines to produce sulfenamides and disulfides is an important reaction in the industry. Classical synthesis of sulfenamides involves the reaction between amine nucleophiles and sulfonyl chlorides, which are not widely available, toxic and unstable, and the reaction requires a combination of oxidizing and chlorinating reagents. Here we decribe an environmentally benign protocol for the efficient electrochemical dehydrogenative coupling of thiols with amines or thiols to form compounds possessing S–N or S–S bonds. We used potassium iodide as electrocatalyst in methanol for a greener reaction. The reaction operates under ambient conditions in the absence of oxidants, acids/bases or toxic/explosive electrolytes. Results show that most produced sulfenamides and disulfides are generated in yields up to 99%.


Similar content being viewed by others
References
Akhil D, Lakshmi D, Kartik A, Vo DVN, Arun J, Gopinath KP (2021) Production, characterization, activation and environmental applications of engineered biochar: a review. Environ Chem Lett 19:2261–2297
Anastas P, Eghbali N (2010) Green chemistry: principles and practice. Chem Soc Rev 39:301–312
Briggs EL, Tota A, Colella M, Degennaro L, Luisi R, Bull JA (2019) Synthesis of sulfonimidamides from sulfenamides via an alkoxy-amino-lambda (6)-sulfanenitrile Intermediate. Angew Chem Int Ed 58:14303–14310
Cao Y, Abdolmohammadi S, Ahmad R, Issakhov A, Ebadi AG, Vessally E (2021) Direct synthesis of sulfenamides, sulfinamides, and sulfonamides from thiols and amines. RSC Adv 11:32394–32407
Dou YC, Huang X, Wang H, Yang LT, Li H, Yuan B, Yang G (2017) Reusable cobalt-phthalocyanine in water: efficient catalytic aerobic oxidative coupling of thiols to construct S-N/S-S bonds. Green Chem 19:2491–2495
Guin A, Rathod T, Gaykar RN, Roy T, Biju AT (2020) Lewis acid catalyzed ring-opening 1,3-Aminothiolation of donor–acceptor cyclopropanes using sulfenamides. Org Lett 22:2276–2280
He MX, Cheng SY, Pan YZ, Tang HT, Pan YM (2021) Electrochemically mediated S—N bond formation: synthesis of sulfenamides. Chinese J Org Chem 41:2354–2360
Laudadio G, Barmpoutsis SC, Struik L, Govaerts S, Browne DL, Noël T (2019) Sulfonamide synthesis through electrochemical oxidative coupling of amines and thiols. J Am Chem Soc 141:5664–5668
Laudadio G, Straathof NJW, Lanting MD, Knoops B, Hessel W, Noël T (2017) An environmentally benign and selective electrochemical oxidation of sulfides and thiols in a continuous flow microreactor. Green Chem 19:4061–4066
Liu SP, Qi ZJ, Zhang Z, Qian B (2019) Iodine/Manganese dual catalysis for oxidative dehydrogenation coupling of amines with thiols. Org Lett 21:7722–7725
Milović E, Janković N, Vraneš M, Stefanović S, Petronijević J, Joksimović N, Muškinja J, Ratković Z (2021) Green one-pot synthesis of pyrido-dipyrimidine DNA-base hybrids in water. Environ Chem Lett 19:729–736
Mulina OM, Ilovaisky AI, Terent’ev AO (2018) Oxidative coupling with S–N bond formation. Eur J Org Chem 2018:4648–4672
Rao RN, Jean S, Mukherjee M, Maiti B, Chanda K (2021) Green synthesis of biologically active heterocycles of medicinal importance: a review. Environ Chem Lett 19:3315
Ruslan NAAA, Kan SY, Hamzah AS, Chia PW (2021) Natural food additives as green catalysts in organic synthesis: a review. Environ Chem Lett 19:3359–3380
Ryu SH, Ra J, Ko HM (2020) Efficient synthesis of sulfenamides through mitsunobu-type coupling reaction of thiols with amines using dibenzyl azodicarboxylate. Asian J Org Chem 9:933–938
Sengul AB, Asmatulu E (2020) Toxicity of metal and metal oxide nanoparticles: a review. Environ Chem Lett 18:1659–1683
Shinkar EV, Smolyaninov IV, Kuzmin VV, Berberova NT (2019) Redox activation of hydrogen sulfide, thiols, and sulfur in electrosynthesis of organic di- and polysulfides. Russ J Gen Chem 89:689–696
Sun JF, Wu JT, Xu QQ, Zhou D, Yin JZ (2020) CO2 electrochemical reduction using single-atom catalysts. Preparation, characterization and anchoring strategies: a review. Environ Chem Lett 18:1593–1623
Tang SY, Liu Y, Li LJ, Ren XH, Li J, Yang G, Li H, Yuan B (2019) Scalable electrochemical oxidant-and metal-free dehydrogenative coupling of S-H/N-H. Org Biomol Chem 17:1370–1374
Wei ZX, Wang RJ, Zhang YH, Wang B, Xia Y, Abdukader A, Xue F, Jin WW, Liu CJ (2021) Electrochemical direct thiolation of lactams with mercaptans: an efficient access to N-Acylsulfenamides. Eur J Org Chem 21:4728–4732
Wiebe A, Gieshoff T, Möhle S, Rodrigo E, Zirbes M, Waldvogel SR (2018) Electrifying organic synthesis. Angew Chem Int Ed 57:5594–5619
Wu Y, Peng K, Hu Z, Fan Y, Shi Z, Hao E, Dong Z (2021) Iodine-Mediated cross-dehydrogenative coupling of heterocyclic thiols with amines: an easy and practical formation of S-N bond. Eur J Org Chem 43:5889–5894
Yang LT, Li SD, Dou YC, Zhen S, Li H, Zhang P, Yuan B, Yang G (2017) TEMPO-Catalyzed aerobic oxidative coupling of thiols for metal-free formation of S-N/S-S bonds. Asian J Org Chem 6:265–268
Yang ZZ, Shi YS, Zhan Z, Zhang H, Xing H, Lu R, Zhang Y, Guan M, Wu Y (2018) Sustainable electrocatalytic oxidant-free syntheses of thiosulfonates from thiols. ChemElectroChem 5:3619–3623
Yu LY, Lin CY, Liao CS, Zeng XH, Chen XW, Zhu ZZ, Huang YB, Li YB, Chen L (2020) Green chemistry: efficient acetalization of aldehydes with alcohols using the acid red 52 photocatalyst. Environ Chem Lett 18:1353–1359
Zhang H, Wang H, Jiang Y, Cao F, Gao W, Zhu L, Yang Y, Wang X, Wang Y, Chen J, Feng Y, Deng X, Lu Y, Hu X, Li X, Zhang J, Shi T, Wang Z (2020) Recent advances in Iodine-Promoted C−S/N−S bonds formation. Eur J Org Chem 26:17289–17317
Acknowledgements
The research leading to these results received funding support from the National Natural Science Foundation of China with Grant No. 21868011 and the National Key R&D Program of China with Grant No. 2017YFC1103800. We are also appreciated for financial support from the talent youth program of Shandong University of Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, T., Wang, R., Ma, L. et al. High-efficiency synthesis of sulfenamides and disulfides by electrochemical dehydrogenative coupling. Environ Chem Lett 20, 2765–2771 (2022). https://doi.org/10.1007/s10311-022-01459-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-022-01459-0