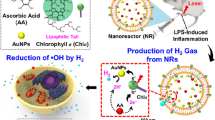
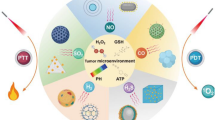
Abstract
Gas therapy appears promising in treating cancer, ischemia-reperfusion injury, heart attack, and acute situations of shock. Gas therapy is also becoming a good choice in preventive medicine against dementia, reducing cancer occurrence, cardiovascular diseases, and delaying aging. On-demand gas release allows precision therapy with few risks of gas poisoning. Here, we review targeted and controlled gas therapy, gas nanogenerators for cancer bioimaging, and gas-sensitized synergistic therapy. We focus on gases with anticancer effects such as hydrogen, carbon monoxide, carbon dioxide, nitric oxide, oxygen sulfur dioxide, hydrogen sulfide, and heavy gases. We discuss the efficiency of gas therapy combined with other therapies methods such as starvation therapy, ultrasound therapy, chemotherapy, radiotherapy, and photodynamic therapy, and applications in multimodal and ultrasound imaging.

Copyright 2017, Wiley-VCH

Copyright 2016 American Chemistry Society. b Formation of hematoporphyrin monomethyl ether/mesoporous calcium carbonate-hyaluronic acid and its mechanism by which the extraordinary effect of the destruction of cancer with multimechanisms, resulting in cell necrosis cell apoptosis and also blood vessel destruction. Adapted with permission from Feng et al. (2018a,b), Copyright 2018 Wiley-VCH

Copyright 2016 American Chemistry Society. b Design and application of photocatalytic nanomaterials (histidine-silver phosphate-doped carbon-dot-decorated carbon nitride nanoparticles) targeting cancer cells and converting carbon dioxide into carbon monoxide intratumorally. The formed carbon monoxide helps in increasing the intracellular oxidative stress in cancer cells and eventually increased the therapeutic effect of chemotherapy. Adapted with permission from Zheng et al. (2017), Copyright 2017 Wiley-VCH

Copyright 2015 Wiley–VCH

Copyright 2018 Wiley-VCH. b Multimodal imaging lead-oxygen-boosted photodynamic therapy nanoplatform. Adapted with permission from Liang et al. (2018), Copyright 2018 Elsevier

Copyright 2018 Nature

Copyright 2018 The Royal Society of Chemistry
Similar content being viewed by others
References
Aslund AKO, Snipstad S, Healey A, Kvale S, Torp SH, Sontum PC, Davies CdL, van Wamel A (2017) Efficient enhancement of blood-brain barrier permeability using acoustic cluster therapy (ACT). Theranostics 7(1):23–30. https://doi.org/10.7150/thno.16577
Bailey MR, Khokhlova VA, Sapozhnikov OA, Kargl SG, Crum LA (2003) Physical mechanisms of the therapeutic effect of ultrasound (a review). Acoust Phys 49:369–388. https://doi.org/10.1134/1.1591291
Bell CC, Gilan O (2020) Principles and mechanisms of non-genetic resistance in cancer. Br J Cancer 122(4):465–472. https://doi.org/10.1038/s41416-019-0648-6
Carpenter AW, Schoenfisch MH (2012) Nitric oxide release: part II therapeutic applications. Chem Soc Rev 41(10):3742–3752. https://doi.org/10.1039/c2cs15273h
Carpentier A, Canney M, Vignot A, Reina V, Beccaria K, Horodyckid C, Karachi C, Leclercq D, Lafon C, Chapelon JY, Capelle L, Cornu P, Sanson M, Hoang-Xuan K, Delattre JY, Idbaih A (2016) Clinical trial of blood-brain barrier disruption by pulsed ultrasound. Sci Transl Med 8(343):343re2. https://doi.org/10.1126/scitranslmed.aaf6086
Casas A, Di Venosa G, Hasan T, Al B (2011) Mechanisms of resistance to photodynamic therapy. Curr Med Chem 18(16):2486–2515. https://doi.org/10.2174/092986711795843272
Chen J, Luo H, Liu Y, Zhang W, Li H, Luo T, Zhang K, Zhao Y, Liu J (2017a) Oxygen-self-produced nanoplatform for relieving hypoxia and breaking resistance to sonodynamic treatment of pancreatic cancer. ACS Nano 11(12):12849–12862. https://doi.org/10.1021/acsnano.7b08225
Chen L, He Q, Lei M, Xiong L, Shi K, Tan L, Jin Z, Wang T, Qian Z (2017b) Facile coordination-precipitation route to insoluble metal roussin’s black salts for NIR-responsive release of NO for anti-metastasis. ACS Appl Mater Interfaces 9(42):36473–36477. https://doi.org/10.1021/acsami.7b11325
Chen L, Zhou SF, Su L, Song J (2019) Gas-mediated cancer bioimaging and therapy. ACS Nano 13(10):10887–10917. https://doi.org/10.1021/acsnano.9b04954
Chuang EY, Lin CC, Chen KJ, Wan DH, Lin KJ, Ho YC, Lin PY, Sung HW (2016) A FRET-guided, NIR-responsive bubble-generating liposomal system for in vivo targeted therapy with spatially and temporally precise controlled release. Biomaterials 93:48–59. https://doi.org/10.1016/j.biomaterials.2016.03.040
Chung MF, Liu HY, Lin KJ, Chia WT, Sung HW (2015) A pH-responsive carrier system that generates NO bubbles to trigger drug release and reverse P-glycoprotein-mediated multidrug resistance. Angew Chem Int Ed Engl 54(34):9890–9893. https://doi.org/10.1002/anie.201504444
de Luca A, Moroni N, Serafino A, Primavera A, Pastore A, Pedersen JZ, Petruzzelli R, Farrace MG, Pierimarchi P, Moroni G, Federici G, Sinibaldi Vallebona P, Lo Bello M (2011) Treatment of doxorubicin-resistant MCF7/Dx cells with nitric oxide causes histone glutathionylation and reversal of drug resistance. Biochem J 440(2):175–183. https://doi.org/10.1042/BJ20111333
De Ridder M, Verellen D, Verovski V, Storme G (2008) Hypoxic tumor cell radiosensitization through nitric oxide. Nitric Oxide 19(2):164–169. https://doi.org/10.1016/j.niox.2008.04.015
Deng Y, Jia F, Chen S, Shen Z, Jin Q, Fu G, Ji J (2018) Nitric oxide as an all-rounder for enhanced photodynamic therapy: hypoxia relief, glutathione depletion, and reactive nitrogen species generation. Biomaterials 187:55–65. https://doi.org/10.1016/j.biomaterials.2018.09.043
Dewhirst MW, Secomb TW (2017) Transport of drugs from blood vessels to tumour tissue. Nat Rev Cancer 17(12):738–750. https://doi.org/10.1038/nrc.2017.93
Dimcevski G, Kotopoulis S, Bjanes T, Hoem D, Schjott J, Gjertsen BT, Biermann M, Molven A, Sorbye H, McCormack E, Postema M, Gilja OH (2016) A human clinical trial using ultrasound and microbubbles to enhance gemcitabine treatment of inoperable pancreatic cancer. J Control Release 243:172–181. https://doi.org/10.1016/j.jconrel.2016.10.007
Fan W, Bu W, Zhang Z, Shen B, Zhang H, He Q, Ni D, Cui Z, Zhao K, Bu J, Du J, Liu J, Shi J (2015) X-ray radiation-controlled NO-release for on-demand depth-independent hypoxic radiosensitization. Angew Chem Int Ed Engl 54(47):14026–14030. https://doi.org/10.1002/anie.201504536
Fan CH, Cheng YH, Ting CY, Ho YJ, Hsu PH, Liu HL, Yeh CK (2016a) Ultrasound/magnetic targeting with SPIO-DOX-microbubble complex for image-guided drug delivery in brain tumors. Theranostics 6(10):1542–1556. https://doi.org/10.7150/thno.15297
Fan J, He Q, Liu Y, Zhang F, Yang X, Wang Z, Lu N, Fan W, Lin L, Niu G, He N, Song J, Chen X (2016b) Light-responsive biodegradable nanomedicine overcomes multidrug resistance via NO-enhanced chemosensitization. ACS Appl Mater Interfaces 8(22):13804–13811. https://doi.org/10.1021/acsami.6b03737
Fan W, Lu N, Huang P, Liu Y, Yang Z, Wang S, Yu G, Liu Y, Hu J, He Q, Qu J, Wang T, Chen X (2017) Glucose-responsive sequential generation of hydrogen peroxide and nitric oxide for synergistic cancer starving-like/gas therapy. Angew Chem Int Ed Engl 56(5):1229–1233. https://doi.org/10.1002/anie.201610682
Fan W, Lu N, Shen Z, Tang W, Shen B, Cui Z, Shan L, Yang Z, Wang Z, Jacobson O, Zhou Z, Liu Y, Hu P, Yang W, Song J, Zhang Y, Zhang L, Khashab NM, Aronova MA, Lu G, Chen X (2019) Generic synthesis of small-sized hollow mesoporous organosilica nanoparticles for oxygen-independent X-ray-activated synergistic therapy. Nat Commun 10(1):1241. https://doi.org/10.1038/s41467-019-09158-1
Feng Q, Li Y, Yang X, Zhang W, Hao Y, Zhang H, Hou L, Zhang Z (2018a) Hypoxia-specific therapeutic agents delivery nanotheranostics: a sequential strategy for ultrasound mediated on-demand tritherapies and imaging of cancer. J Controlled Release 275:192–200. https://doi.org/10.1016/j.jconrel.2018.02.011
Feng Q, Zhang W, Yang X, Li Y, Hao Y, Zhang H, Hou L, Zhang Z (2018) pH/Ultrasound dual-responsive gas generator for ultrasound imaging-guided therapeutic inertial cavitation and sonodynamic therapy. Adv Healthc Mater. https://doi.org/10.1002/adhm.201700957
Frederiksen LJ, Sullivan R, Maxwell LR, Macdonald-Goodfellow SK, Adams MA, Bennett BM, Siemens DR, Graham CH (2007) Chemosensitization of cancer in vitro and in vivo by nitric oxide signaling. Clin Cancer Res 13(7):2199–2206. https://doi.org/10.1158/1078-0432.CCR-06-1807
Gao W, Li S, Liu Z, Sun Y, Cao W, Tong L, Cui G, Tang B (2017) Targeting and destroying tumor vasculature with a near-infrared laser-activated “nanobomb” for efficient tumor ablation. Biomaterials 139:1–11. https://doi.org/10.1016/j.biomaterials.2017.05.037
Ge L, Yang M, Yang NN, Yin XX, Song WG (2017) Molecular hydrogen: a preventive and therapeutic medical gas for various diseases. Oncotarget 8(60):102653–102673. https://doi.org/10.18632/oncotarget.21130
He Q (2017) Precision gas therapy using intelligent nanomedicine. Biomater Sci 5(11):2226–2230. https://doi.org/10.1039/c7bm00699c
Helfield B (2019) A review of phospholipid encapsulated ultrasound contrast agent microbubble physics. Ultrasound Med Biol 45(2):282–300. https://doi.org/10.1016/j.ultrasmedbio.2018.09.020
Hirsch LR, Stafford RJ, Bankson JA, Sershen SR, Rivera B, Price RE, Hazle JD, Halas NJ, West JL (2003) Nanoshell-mediated near-infrared thermal therapy of tumors under magnetic resonance guidance. Proc Natl Acad Sci U S A 100(23):13549–13554. https://doi.org/10.1073/pnas.2232479100
Huang HY, Hu SH, Hung SY, Chiang CS, Liu HL, Chiu TL, Lai HY, Chen YY, Chen SY (2013) SPIO nanoparticle-stabilized PAA-F127 thermosensitive nanobubbles with MR/US dual-modality imaging and HIFU-triggered drug release for magnetically guided in vivo tumor therapy. J Control Release 172(1):118–127. https://doi.org/10.1016/j.jconrel.2013.07.029
Huang CC, Chia WT, Chung MF, Lin KJ, Hsiao CW, Jin C, Lim WH, Chen CC, Sung HW (2016) An implantable depot that can generate oxygen in situ for overcoming hypoxia-induced resistance to anticancer drugs in chemotherapy. J Am Chem Soc 138(16):5222–5225. https://doi.org/10.1021/jacs.6b01784
Huerta S, Chilka S, Bonavida B (2008) Nitric oxide donors: novel cancer therapeutics (review). Int J Oncol 33(5):909–927
Huynh E, Leung BY, Helfield BL, Shakiba M, Gandier JA, Jin CS, Master ER, Wilson BC, Goertz DE, Zheng G (2015) In situ conversion of porphyrin microbubbles to nanoparticles for multimodality imaging. Nat Nanotechnol 10(4):325–332. https://doi.org/10.1038/nnano.2015.25
Ishibashi T (2013) Molecular hydrogen: new antioxidant and anti-inflammatory therapy for rheumatoid arthritis and related diseases. Curr Pharm Des 19(35):6375–6381. https://doi.org/10.2174/13816128113199990507
Jahanban-Esfahlan R, de la Guardia M, Ahmadi D, Yousefi B (2018) Modulating tumor hypoxia by nanomedicine for effective cancer therapy. J Cell Physiol 233(3):2019–2031. https://doi.org/10.1002/jcp.25859
Ji X, Wang B (2018) Strategies toward organic carbon monoxide prodrugs. Acc Chem Res 51(6):1377–1385. https://doi.org/10.1021/acs.accounts.8b00019
Jin Z, Wen Y, Hu Y, Chen W, Zheng X, Guo W, Wang T, Qian Z, Su BL, He Q (2017) MRI-guided and ultrasound-triggered release of NO by advanced nanomedicine. Nanoscale 9(10):3637–3645. https://doi.org/10.1039/c7nr00231a
Jordan BF, Sonveaux P, Feron O, Gregoire V, Beghein N, Dessy C, Gallez B (2004) Nitric oxide as a radiosensitizer: evidence for an intrinsic role in addition to its effect on oxygen delivery and consumption. Int J Cancer 109(5):768–773. https://doi.org/10.1002/ijc.20046
Ke H, Wang J, Tong S, Jin Y, Wang S, Qu E, Bao G, Dai Z (2013) Gold nanoshelled liquid perfluorocarbon magnetic nanocapsules: a nanotheranostic platform for bimodal ultrasound/magnetic resonance imaging guided photothermal tumor ablation. Theranostics 4(1):12–23. https://doi.org/10.7150/thno.7275
Kim J, Yung BC, Kim WJ, Chen X (2017) Combination of nitric oxide and drug delivery systems: tools for overcoming drug resistance in chemotherapy. J Control Release 263:223–230. https://doi.org/10.1016/j.jconrel.2016.12.026
Lammertink BH, Bos C, Deckers R, Storm G, Moonen CT, Escoffre JM (2015) Sonochemotherapy: from bench to bedside. Front Pharmacol 6:138. https://doi.org/10.3389/fphar.2015.00138
Lentacker I, De Cock I, Deckers R, De Smedt SC, Moonen CT (2014) Understanding ultrasound induced sonoporation: definitions and underlying mechanisms. Adv Drug Deliv Rev 72:49–64. https://doi.org/10.1016/j.addr.2013.11.008
Li WP, Su CH, Tsao LC, Chang CT, Hsu YP, Yeh CS (2016) Controllable CO release following near-infrared light-induced cleavage of iron carbonyl derivatized prussian blue nanoparticles for CO-assisted synergistic treatment. ACS Nano 10(12):11027–11036. https://doi.org/10.1021/acsnano.6b05858
Li DS, Yoon SJ, Pelivanov I, Frenz M, O’Donnell M, Pozzo LD (2017) Polypyrrole-coated perfluorocarbon nanoemulsions as a sono-photoacoustic contrast agent. Nano Lett 17(10):6184–6194. https://doi.org/10.1021/acs.nanolett.7b02845
Li SY, Cheng H, Xie BR, Qiu WX, Zeng JY, Li CX, Wan SS, Zhang L, Liu WL, Zhang XZ (2017) Cancer cell membrane camouflaged cascade bioreactor for cancer targeted starvation and photodynamic therapy. ACS Nano 11(7):7006–7018. https://doi.org/10.1021/acsnano.7b02533
Li W, Hou W, Guo X, Luo L, Li Q, Zhu C, Yang J, Zhu J, Du Y, You J (2018) Temperature-controlled, phase-transition ultrasound imaging-guided photothermal-chemotherapy triggered by NIR light. Theranostics 8(11):3059–3073. https://doi.org/10.7150/thno.23885
Liang R, Liu L, He H, Chen Z, Han Z, Luo Z, Wu Z, Zheng M, Ma Y, Cai L (2018) Oxygen-boosted immunogenic photodynamic therapy with gold nanocages@manganese dioxide to inhibit tumor growth and metastases. Biomaterials 177:149–160. https://doi.org/10.1016/j.biomaterials.2018.05.051
Liao ZX, Chuang EY, Lin CC, Ho YC, Lin KJ, Cheng PY, Chen KJ, Wei HJ, Sung HW (2015) An AS1411 aptamer-conjugated liposomal system containing a bubble-generating agent for tumor-specific chemotherapy that overcomes multidrug resistance. J Control Release 208:42–51. https://doi.org/10.1016/j.jconrel.2015.01.032
Lin X, Qiu Y, Song L, Chen S, Chen X, Huang G, Song J, Chen X, Yang H (2019) Ultrasound activation of liposomes for enhanced ultrasound imaging and synergistic gas and sonodynamic cancer therapy. Nanoscale Horiz 4:747–756. https://doi.org/10.1039/C8NH00340H
Liu T, Zhang N, Wang Z, Wu M, Chen Y, Ma M, Chen H, Shi J (2017) Endogenous catalytic generation of o2 bubbles for in situ ultrasound-guided high intensity focused ultrasound ablation. ACS Nano 11(9):9093–9102. https://doi.org/10.1021/acsnano.7b03772
Liu Y, Yang F, Yuan C, Li M, Wang T, Chen B, Jin J, Zhao P, Tong J, Luo S, Gu N (2017) Magnetic nanoliposomes as in situ microbubble bombers for multimodality image-guided cancer theranostics. ACS Nano 11(2):1509–1519. https://doi.org/10.1021/acsnano.6b06815
Liu J, Xu F, Huang J, Xu J, Liu Y, Yao Y, Ao M, Li A, Hao L, Cao Y, Hu Z, Ran H, Wang Z, Li P (2018) Low-intensity focused ultrasound (LIFU)-activated nanodroplets as a theranostic agent for noninvasive cancer molecular imaging and drug delivery. Biomater Sci 6(11):2838–2849. https://doi.org/10.1039/c8bm00726h
Markman JL, Rekechenetskiy A, Holler E, Ljubimova JY (2013) Nanomedicine therapeutic approaches to overcome cancer drug resistance. Adv Drug Deliv Rev 65(13–14):1866–1879. https://doi.org/10.1016/j.addr.2013.09.019
McEwan C, Owen J, Stride E, Fowley C, Nesbitt H, Cochrane D, Coussios CC, Borden M, Nomikou N, McHale AP, Callan JF (2015) Oxygen carrying microbubbles for enhanced sonodynamic therapy of hypoxic tumours. J Control Release 203:51–56. https://doi.org/10.1016/j.jconrel.2015.02.004
Melamed JR, Edelstein RS, Day ES (2015) Elucidating the fundamental mechanisms of cell death triggered by photothermal therapy. ACS Nano 9(1):6–11. https://doi.org/10.1021/acsnano.5b00021
Min KH, Min HS, Lee HJ, Park DJ, Yhee JY, Kim K, Kwon IC, Jeong SY, Silvestre OF, Chen X, Hwang YS, Kim EC, Lee SC (2015) pH-controlled gas-generating mineralized nanoparticles: a theranostic agent for ultrasound imaging and therapy of cancers. ACS Nano 9(1):134–145. https://doi.org/10.1021/nn506210a
Motterlini R, Otterbein LE (2010) The therapeutic potential of carbon monoxide. Nat Rev Drug Discov 9(9):728–743. https://doi.org/10.1038/nrd3228
Ohsawa I, Ishikawa M, Takahashi K, Watanabe M, Nishimaki K, Yamagata K, Katsura K, Katayama Y, Asoh S, Ohta S (2007) Hydrogen acts as a therapeutic antioxidant by selectively reducing cytotoxic oxygen radicals. Nat Med 13(6):688–694. https://doi.org/10.1038/nm1577
Paproski RJ, Forbrich A, Huynh E, Chen J, Lewis JD, Zheng G, Zemp RJ (2016) Porphyrin nanodroplets: sub-micrometer ultrasound and photoacoustic contrast imaging agents. Small 12(3):371–380. https://doi.org/10.1002/smll.201502450
Powell CR, Dillon KM, Matson JB (2018) A review of hydrogen sulfide (H2S) donors: chemistry and potential therapeutic applications. Biochem Pharmacol 149:110–123. https://doi.org/10.1016/j.bcp.2017.11.014
Rapozzi V, Della Pietra E, Zorzet S, Zacchigna M, Bonavida B, Xodo LE (2013) Nitric oxide-mediated activity in anti-cancer photodynamic therapy. Nitric Oxide 30:26–35. https://doi.org/10.1016/j.niox.2013.01.002
Riganti C, Miraglia E, Viarisio D, Costamagna C, Pescarmona G, Ghigo D, Bosia A (2005) Nitric oxide reverts the resistance to doxorubicin in human colon cancer cells by inhibiting the drug efflux. Cancer Res 65(2):516–525
Rofstad EK, Sundfor K, Lyng H, Trope CG (2000) Hypoxia-induced treatment failure in advanced squamous cell carcinoma of the uterine cervix is primarily due to hypoxia-induced radiation resistance rather than hypoxia-induced metastasis. Br J Cancer 83(3):354–359. https://doi.org/10.1054/bjoc.2000.1266
Romao CC, Blattler WA, Seixas JD, Bernardes GJ (2012) Developing drug molecules for therapy with carbon monoxide. Chem Soc Rev 41(9):3571–3583. https://doi.org/10.1039/c2cs15317c
Shannon AM, Bouchier-Hayes DJ, Condron CM, Toomey D (2003) Tumour hypoxia, chemotherapeutic resistance and hypoxia-related therapies. Cancer Treat Rev 29(4):297–307. https://doi.org/10.1016/s0305-7372(03)00003-3
Shen W, Liu W, Yang H, Zhang P, Xiao C, Chen X (2018) A glutathione-responsive sulfur dioxide polymer prodrug as a nanocarrier for combating drug-resistance in cancer chemotherapy. Biomaterials 178:706–719. https://doi.org/10.1016/j.biomaterials.2018.02.011
Snipstad S, Sulheim E, de Lange DC, Moonen C, Storm G, Kiessling F, Schmid R, Lammers T (2018) Sonopermeation to improve drug delivery to tumors: from fundamental understanding to clinical translation. Expert Opin Drug Deliv 15(12):1249–1261. https://doi.org/10.1080/17425247.2018.1547279
Song G, Liang C, Yi X, Zhao Q, Cheng L, Yang K, Liu Z (2016) Perfluorocarbon-loaded hollow Bi2Se3 nanoparticles for timely supply of oxygen under near-infrared light to enhance the radiotherapy of cancer. Adv Mater 28(14):2716–2723. https://doi.org/10.1002/adma.201504617
Song X, Feng L, Liang C, Yang K, Liu Z (2016) Ultrasound triggered tumor oxygenation with oxygen-shuttle nanoperfluorocarbon to overcome hypoxia-associated resistance in cancer therapies. Nano Lett 16(10):6145–6153. https://doi.org/10.1021/acs.nanolett.6b02365
Song G, Ji C, Liang C, Song X, Yi X, Dong Z, Yang K, Liu Z (2017) TaOx decorated perfluorocarbon nanodroplets as oxygen reservoirs to overcome tumor hypoxia and enhance cancer radiotherapy. Biomaterials 112:257–263. https://doi.org/10.1016/j.biomaterials.2016.10.020
Szabo C (2007) Hydrogen sulphide and its therapeutic potential. Nat Rev Drug Discov 6(11):917–935. https://doi.org/10.1038/nrd2425
Szabo C (2016) Gasotransmitters in cancer: from pathophysiology to experimental therapy. Nat Rev Drug Discov 15(3):185–203. https://doi.org/10.1038/nrd.2015.1
Tang W, Yang Z, Wang S, Wang Z, Song J, Yu G, Fan W, Dai Y, Wang J, Shan L, Niu G, Fan Q, Chen X (2018) Organic Semiconducting photoacoustic nanodroplets for laser-activatable ultrasound imaging and combinational cancer therapy. ACS Nano 12(3):2610–2622. https://doi.org/10.1021/acsnano.7b08628
Wan SS, Zeng JY, Cheng H, Zhang XZ (2018) ROS-induced NO generation for gas therapy and sensitizing photodynamic therapy of tumor. Biomaterials 185:51–62. https://doi.org/10.1016/j.biomaterials.2018.09.004
Wegiel B, Gallo D, Csizmadia E, Harris C, Belcher J, Vercellotti GM, Penacho N, Seth P, Sukhatme V, Ahmed A, Pandolfi PP, Helczynski L, Bjartell A, Persson JL, Otterbein LE (2013) Carbon monoxide expedites metabolic exhaustion to inhibit tumor growth. Cancer Res 73(23):7009–7021. https://doi.org/10.1158/0008-5472.CAN-13-1075
Wu L, Wen X, Wang X, Wang C, Sun X, Wang K, Zhang H, Williams T, Stacy AJ, Chen J, Schmieder AH, Lanza GM, Shen B (2018) Local intratracheal delivery of perfluorocarbon nanoparticles to lung cancer demonstrated with magnetic resonance multimodal imaging. Theranostics 8(2):563–574. https://doi.org/10.7150/thno.21466
Wu LH, Cai XJ, Zhu HF, Li JH, Shi DX, Su DF, Yue D, Gu ZW (2018) PDT-driven highly efficient intracellular delivery and controlled release of CO in combination with sufficient singlet oxygen production for synergistic anticancer therapy. Adv Funct Mater 28(41):1804324. https://doi.org/10.1002/adfm.201804324
Xiang HJ, Deng Q, An L, Guo M, Yang SP, Liu JG (2016) Tumor cell specific and lysosome-targeted delivery of nitric oxide for enhanced photodynamic therapy triggered by 808 nm near-infrared light. Chem Commun (Camb) 52(1):148–151. https://doi.org/10.1039/c5cc07006f
Yang G, Xu L, Chao Y, Xu J, Sun X, Wu Y, Peng R, Liu Z (2017) Hollow MnO2 as a tumor-microenvironment-responsive biodegradable nano-platform for combination therapy favoring antitumor immune responses. Nat Commun 8(1):902. https://doi.org/10.1038/s41467-017-01050-0
Yang T, Jin ZK, Wang ZH, Zhao PH, Zhao B, Fan MJ, Chen LH, Wang TF, Su BL, He QJ (2018) Intratumoral high-payload delivery and acid-responsive release of H2 for efficient cancer therapy using the ammonia borane-loaded mesoporous silica nanomedicine. Appl Mater Today 11:136–143. https://doi.org/10.1016/j.apmt.2018.01.008
Yu G, Yang J, Fu X, Wang Z, Shao L, Mao Z, Liu Y, Yang Z, Zhang F, Fan W, Song J, Zhou Z, Gao C, Huang F, Chen X (2018) Supramolecular hybrid material constructed from graphene oxide and pillar[6]arene-based host-guest complex as a ultrasound and photoacoustic signals nanoamplifier. Mater Horiz 5(3):429–435. https://doi.org/10.1039/C8MH00128F
Yu M, Xu X, Cai Y, Zou L, Shuai X (2018) Perfluorohexane-cored nanodroplets for stimulations-responsive ultrasonography and O2-potentiated photodynamic therapy. Biomaterials 175:61–71. https://doi.org/10.1016/j.biomaterials.2018.05.019
Zhang K, Xu H, Chen H, Jia X, Zheng S, Cai X, Wang R, Mou J, Zheng Y, Shi J (2015) CO2 bubbling-based “nanobomb” system for targetedly suppressing Panc-1 pancreatic tumor via low intensity ultrasound-activated inertial cavitation. Theranostics 5(11):1291–1302. https://doi.org/10.7150/thno.12691
Zhang K, Xu H, Jia X, Chen Y, Ma M, Sun L, Chen H (2016) Ultrasound-triggered nitric oxide release platform based on energy transformation for targeted inhibition of pancreatic tumor. ACS Nano 10(12):10816–10828. https://doi.org/10.1021/acsnano.6b04921
Zhang C, Ni D, Liu Y, Yao H, Bu W, Shi J (2017) Magnesium silicide nanoparticles as a deoxygenation agent for cancer starvation therapy. Nat Nanotechnol 12(4):378–386. https://doi.org/10.1038/nnano.2016.280
Zhang C, Moonshi SS, Wang W, Ta HT, Han Y, Han FY, Peng H, Kral P, Rolfe BE, Gooding JJ, Gaus K, Whittaker AK (2018) High F-content perfluoropolyether-based nanoparticles for targeted detection of breast cancer by (19)F magnetic resonance and optical imaging. ACS Nano 12(9):9162–9176. https://doi.org/10.1021/acsnano.8b03726
Zhang Y, Zhan X, Xiong J, Peng S, Huang W, Joshi R, Cai Y, Liu Y, Li R, Yuan K, Zhou N, Min W (2018) Temperature-dependent cell death patterns induced by functionalized gold nanoparticle photothermal therapy in melanoma cells. Sci Rep 8:8720. https://doi.org/10.1038/s41598-018-26978-1
Zhang H, Xie M, Chen H, Bavi S, Sohail M, Bavi R (2021) Gas-mediated cancer therapy. Environ Chem Lett 19:149–166. https://doi.org/10.1007/s10311-020-01062-1
Zhao P, Jin Z, Chen Q, Yang T, Chen D, Meng J, Lu X, Gu Z, He Q (2018) Local generation of hydrogen for enhanced photothermal therapy. Nat Commun 9(1):4241. https://doi.org/10.1038/s41467-018-06630-2
Zheng DW, Li B, Li CX, Fan JX, Lei Q, Li C, Xu Z, Zhang XZ (2016) Carbon-dot-decorated carbon nitride nanoparticles for enhanced photodynamic therapy against hypoxic tumor via water splitting. ACS Nano 10(9):8715–8722. https://doi.org/10.1021/acsnano.6b04156
Zheng DW, Li B, Li CX, Xu L, Fan JX, Lei Q, Zhang XZ (2017) Photocatalyzing CO2 to CO for enhanced cancer therapy. Adv Mater. https://doi.org/10.1002/adma.201703822
Zhou Z, Zhang B, Wang H, Yuan A, Hu Y, Wu J (2018) Two-stage oxygen delivery for enhanced radiotherapy by perfluorocarbon nanoparticles. Theranostics 8(18):4898–4911. https://doi.org/10.7150/thno.27598
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. NRF-2018R1A5A1025077). This research was supported by the Chung-Ang University Research Scholarship Grants in 2020.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest:
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Thakar, S.B., Ghorpade, P.N., Shaker, B. et al. Gas-mediated cancer therapy combined with starvation therapy, ultrasound therapy, chemotherapy, radiotherapy, and photodynamic therapy: a review. Environ Chem Lett 19, 2981–2993 (2021). https://doi.org/10.1007/s10311-021-01218-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-021-01218-7