Abstract
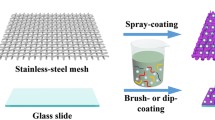
Copper surfaces decorated with micro- and nanostructures can be applied to self-cleaning superhydrophobic surfaces, energy devices and pollution remediation. Wet chemical etching is the classical strategy to grow micro- and nanostructures on copper surfaces. However, such processes usually produce hazardous chemical waste. Here, we present a one-step, eco-friendly and solvent-less alternative to conventional wet chemical methods. Our method involves the use of volatile compounds released from yolk and egg white of boiled chicken eggs as green oxidants for the growth of micro- and nanostructures on the copper surface. After treatment with egg components, copper substrates were modified with stearic acid to make them superhydrophobic. Stearic acid-coated substrates had a contact angle of about 150°, thus confirming their superhydrophobicity. Similar to copper sheets, superhydrophobic copper meshes were also prepared and used for the selective removal of oil from oil/water mixture. Results show that our method produces a wide range of shapes including hollow and solid spheres, nanoflakes, nanoneedles and dome shapes. We found that H2S, the major volatile compound emitted by eggs, is mainly responsible for the growth of microstructures. Therefore, our method can be used both to capture H2S and to design specific surfaces. The mesh exhibited excellent separation efficiency, more than 95% for the organic solvents. A key advantage of our method is that it can be performed also using waste and expired eggs.




Similar content being viewed by others
References
Crick CR, Parkin IP (2011) CVD of copper and copper oxide thin films via the in situ reduction of copper(ii) nitrate-a route to conformal superhydrophobic coatings. J Mater Chem 21:14712–14716. https://doi.org/10.1039/C1JM11955A
Crini G, Lichtfouse E (2019) Advantages and disadvantages of techniques used for wastewater treatment. Environ Chem Lett 17:145–155. https://doi.org/10.1007/s10311-018-0785-9
Elkhatib EA, Mahdy AM, Salama KA (2015) Green synthesis of nanoparticles by milling residues of water treatment. Environ Chem Lett 13:333–339. https://doi.org/10.1007/s10311-015-0506-6
Jo S-H, Kim K-H, Kim Y-H et al (2013) Study of odor from boiled eggs over time using gas chromatography. Microchem J 110:517–529. https://doi.org/10.1016/j.microc.2013.05.011
Lichtfouse E, Morin-Crini N, Fourmentin M et al (2019) Chitosan for direct bioflocculation of wastewater. Environ Chem Lett. https://doi.org/10.1007/s10311-019-00900-1
Lim WP, Wong CT, Ang SL et al (2006) Phase-selective synthesis of copper sulfide nanocrystals. Chem Mater 18:6170–6177. https://doi.org/10.1021/cm061686i
Liu J, Huang X, Li Y et al (2006) Hierarchical nanostructures of cupric oxide on a copper substrate: controllable morphology and wettability. J Mater Chem 16:4427–4434. https://doi.org/10.1039/B611691D
Liu XQ, Zhang G, Xing HQ et al (2011) Preparation of amphiphilic composite and removal of oil and hexavalent chromium from wastewater. Environ Chem Lett 9:127–132. https://doi.org/10.1007/s10311-009-0256-4
Liu L, Xu F, Ma L (2012) Facile fabrication of a superhydrophobic Cu surface via a selective etching of high-energy facets. J Phys Chem C 116:18722–18727. https://doi.org/10.1021/jp302794p
Liu D, Li B, Wu J, Liu Y (2019) Sorbents for hydrogen sulfide capture from biogas at low temperature: a review. Environ Chem Lett. https://doi.org/10.1007/s10311-019-00925-6
Mishra RK, Mishra P, Verma K et al (2019) Electrospinning production of nanofibrous membranes. Environ Chem Lett 17:767–800. https://doi.org/10.1007/s10311-018-00838-w
Mumm F, van Helvoort ATJ, Sikorski P (2009) Easy route to superhydrophobic copper-based wire-guided droplet microfluidic systems. ACS Nano 3:2647–2652. https://doi.org/10.1021/nn900607p
Rezakazemi M, Khajeh A, Mesbah M (2018) Membrane filtration of wastewater from gas and oil production. Environ Chem Lett 16:367–388. https://doi.org/10.1007/s10311-017-0693-4
Shi Y, Yang W, Feng X et al (2015) Bio-inspired fabrication of copper oxide nanowire films with switchable wettability via a facile thermal oxidation method. RSC Adv 5:26107–26113. https://doi.org/10.1039/C5RA00121H
Upadhyay RK, Dubey A, Waghmare PR et al (2016) Multifunctional reduced graphene oxide coated cloths for oil/water separation and antibacterial application. RSC Adv 6:62760–62767
Villaseñor MJ, Ríos Á (2018) Nanomaterials for water cleaning and desalination, energy production, disinfection, agriculture and green chemistry. Environ Chem Lett 16:11–34. https://doi.org/10.1007/s10311-017-0656-9
Zhao Y, Burda C (2012) Development of plasmonic semiconductor nanomaterials with copper chalcogenides for a future with sustainable energy materials. Energy Environ Sci 5:5564–5576. https://doi.org/10.1039/C1EE02734D
Zhao Y, Pan H, Lou Y et al (2009) Plasmonic Cu2−xS nanocrystals: optical and structural properties of copper-deficient copper(I) sulfides. J Am Chem Soc 131:4253–4261. https://doi.org/10.1021/ja805655b
Zhu H, Yang F, Li J, Guo Z (2016) High-efficiency water collection on biomimetic material with superwettable patterns. Chem Commun 52:12415–12417. https://doi.org/10.1039/c6cc05857d
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Upadhyay, R.K., Waghmare, P.R. Eco-friendly preparation of superhydrophobic copper surfaces for oil/water separation. Environ Chem Lett 18, 505–510 (2020). https://doi.org/10.1007/s10311-019-00952-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-019-00952-3