Abstract
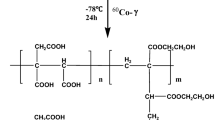
Polysaccharide natural seed coat from the tree Magonia pubescens, in the form of hydrogel was used to remove metals in aqueous solution. Swelling tests indicate that seed coat presents hydrogel behavior, with maximum water absorption of 292 g water/g. Adsorption experiments performed using Na+, Mg2+, K+, Ca2+, Cr3+, Fe3+ and Zn2+ demonstrated that the polysaccharide structure has a high capacity to extract these ions from the aqueous solution. Scanning electron microscopy revealed significant morphological changes of the material before and after water contact. Differential scanning calorimetry measurements indicate a signal shift of the water evaporation temperature in the material with adsorbed zinc. X-ray photoelectron spectroscopy analysis combined with theoretical studies by the density functional theory and on Hartree–Fock (HF) level evidence that the metallic ions were adsorbed through coordination with hydroxyl groups of polysaccharide. In the case of Zn2+ the lowest HF energy was observed for the tetracoordination mode, where Zn2+ is coordinated by two hydroxyl groups and two water molecules.






Similar content being viewed by others
References
Bagot D, Lebeau T, Jezequel K (2006) Microorganisms for remediation of cadmium-contaminated soils. Environ Chem Lett 4:207–211. doi:10.1007/s10311-006-0047-0
Bekiari V, Sotiropoulou M, Bokias G, Lianos P (2008) Use of poly(NN-dimethylacrylamide-co-sodium acrylate)hydrogel to extract cationic dyes and metals from water. Colloids Surf A Physicochem Eng Asp 312:214–218. doi:10.1016/j.colsurfa.2007.06.053
Choi SK, Kim DP, Kim SI, Chang EG (2001) Damage in etching of (BaSr)TiO3 thin films using inductively coupled plasma. J Vac Sci Technol A 19:1063–1067. doi:10.1116/1.1380227
Fiol N, Escudero C, Villaescusa I (2008) Chromium sorption and Cr(VI) reduction to Cr(III) by grape stalks and yohimbe bark. Bioresour Technol 99:5030–5036. doi:10.1016/j.biortech.2007.09.007
Frisch MJ et al (1998) Gaussian 98 Revision A.9 Gaussian Inc., Wallingford
Gorin PAJ, Teixeira AZA, Travassos LR, Laboriau MLS, Iacomini M (1996) Characterization of carbohydrate components of an unsual hydrogel formed by seed coats of Magonia Pubescens (Tingui). Carbohydr Res 282:325–333. doi:10.1016/0008-6215(95)00390-8
Grosse-Sommer A, Prud'Homme RK (1996) Degradable phosphazene-crosslinked hydrogels. J Control Release 40:261–267
Jolly G, Dupont L, Aplincourt M, Lambert J (2006) Improved Cu and Zn sorption on oxidized wheat lignocellulose. Environ Chem Lett 4:219–223. doi:10.1007/s10311-006-0051-4
Lima IS, Airoldi C (2004) A thermodynamic investigation on chitosan-divalent cation interactions. Thermochim Acta 421:133–139
Lin Y, Chen Q, Luo H (2007) Preparation and characterization of N-(2-carboxylbenzyl) chitosan as a potential pH-sensitive hydrogel for drug delivery. Carbohydr Res 342:87–95. doi:10.1016/j.carres.2006.11.002
Prado AGS, Airoldi C (2003) Humic acid-divalent cation interactions. Thermochim Acta 405:287–292. doi:10.1016/S0040-6031(03)00196-5
Silverstein RM, Webster FX (1998) Spectrometric identification of organic compounds, 6th edn. Wiley, New York
Sun S, Wang A (2006) Adsorption properties and mechanism of cross-linked carboxymethyl-chitosan resin with Zn(II) as template ion. React Funct Polym 66:819–826
Wagner CD, Naumkin AV, Kraut-Vass A, Allison Powell CJ, Rumble JR (2008) NIST Standard Reference Database 20, version 3.5, web version: http://srdata.nist.gov/xps/
Zhao Y, Su Haijia S, Fang L, Tan T (2005) Superabsorbent hydrogels from poly(aspartic acid) wiyh salt- temperature- and pH-responsiveness properties. Polymer (Guildf) 46:5368–5376. doi:10.1016/j.polymer.2005.04.015
Acknowledgments
The authors are grateful to The State of São Paulo Research Foundation (FAPESP), The National Council for Scientific and Technological Development (CNPq) and CAPQ-DQI-UFLA for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carvalho, H.W.P., Batista, A.P.L., Hammer, P. et al. Removal of metal ions from aqueous solution by chelating polymeric hydrogel. Environ Chem Lett 8, 343–348 (2010). https://doi.org/10.1007/s10311-009-0231-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10311-009-0231-0