Abstract
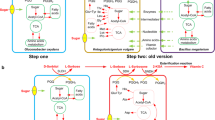
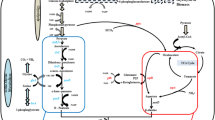
Defect in the amino acid biosynthetic pathways of Ketogulonicigenium vulgare, the producing strain for 2-keto-l-gulonic acid (2-KGA), is the key reason for its poor growth and low productivity. In this study, five different strains were firstly reconstructed by expressing absent genes in threonine, proline and histidine biosynthetic pathways for better 2-KGA productivity. When mono-cultured in the shake flasks, the strain SyBE_Kv02080002 expressing hsk from Gluconobacter oxydans in threonine biosynthetic pathway achieved the highest biomass and the titer increased by 25.13%. When co-cultured with Bacillus endophyticus, the fermentation cycle decreased by 28.57% than that of the original consortium in 5-L fermenter. Furthermore, reconstruction of threonine biosynthetic pathway resulted in up-regulation of genes encoding sorbosone dehydrogenase and idonate-dehydrogenase, which increased the 2-KGA productivity in SyBE_Kv02080002. This study shows that reconstruction of absent biosynthetic pathways in bacteria is an effective way to enhance the productivity of target products.





Similar content being viewed by others
Abbreviations
- 2-KGA:
-
2-Keto-l-gulonic
- SDH:
-
l-Sorbose dehydrogenase
- SNDH:
-
l-Sorbosone dehydrogenase
- IDH:
-
l-Idonate dehydrogenase
- HPP:
-
Histidinol-phosphate phosphatase
- HSK:
-
Homoserine kinase
- P5CR:
-
Pyrroline-5-carboxylate reductase
References
Claus GW, Batzing BL, Baker CA, Goebel EM (1975) Intracytoplasmic membrane formation and increased oxidation of glycerol growth of Gluconobacter oxydans. J Bacteriol 123(3):1169–1183
Chambers ST, Unin CM (1987) Isolation of glycine betaine and proline betaine from human urine. Assessment of their role as osmoprotective agents for bacteria and the kidney. J Clin Invest 79(3):731–737
Chen S, Jia N, Ding MZ, Yuan YJ (2016) Comparative analysis of l-sorbose dehydrogenase by docking strategy for 2-keto-l-gulonic acid production in Ketogulonicigenium vulgare and Bacillus endophyticus consortium. J Ind Microbiol Biotechnol 43(11):1507–1516
Dong X, Quinn PJ, Wang X (2012) Microbial metabolic engineering for l-threonine production. Sub-cell Biochem 64(1):283–302
Du J, Bai W, Song H, Yuan YJ (2013) Combinational expression of sorbose/sorbosone dehydrogenases and cofactor pyrroloquinoline quinone increases 2-keto-l-gulonic acid production in Ketogulonigenium vulgare–Bacillus cereus consortium. Metab Eng 19:50–56
Fox JT, Stover PJ (2008) Folate-mediated one-carbon metabolism. Vitam Horm 79:1–44
Fu S, Zhang W, Guo A, Wang J (2007) Identification of promoters of two dehydrogenase genes in Ketogulonicigenium vulgare DSM 4025 and their strength comparison in K. vulgare and Escherichia coli. Appl Microbiol Biotechnol 75(5):1127–1132
Jia N, Ding MZ, Du J et al (2016) Insights into mutualism mechanism and versatile metabolism of Ketogulonicigenium vulgare Hbe602 based on comparative genomics and metabolomics studies. Sci Rep 6:23068
Jin JH, Choi KK, Jung US et al (2004) Regulatory analysis of amino acid synthesis pathway in Escherichia coli: aspartate family. Enzym Microb Technol 35(35):694–706
Kovach ME, Elzer PH, Hill DS et al (1995) Four new derivatives of the broad-host-range cloning vector pBBR1MCS, carrying different antibiotic-resistance cassettes. Gene 166(1):175–176
Leduc S, de Troostembergh JC, Lebeault JM (2004) Folate requirements of the 2-keto-l-gulonic acid-producing strain Ketogulonigenium vulgare LMP P-20356 in l-sorbose/CSL medium. Appl Microbiol Biotechnol 65:163–167
Liang X, Dickman MB, Becker DF (2014) Proline biosynthesis is required for endoplasmic stress in Saccharomyces cerevisiae. J Biol Chem 289(40):27794–27806
Liu LM, Chen KJ, Zhang J, Liu J, Chen J (2011) Gelatin enhances 2-keto-l-gulonic acid production based on Ketogulonigenium vulgare genome annotation. J Biotechnol 156(3):182–187
Liu LM, Li Y, Zhang J (2011) Complete genome sequence of the industrial strain Ketogulonicigenium vulgare WSH-001. J Bacteriol 193(21):6108–6109
Ma Q, Zhang WW, Zhang L et al (2012) Proteomic analysis of Ketogulonicigenium vulgare under glutathione reveals high demand for thiamin transport and antioxidant protection. PLoS One 7:e32156
Meng ZH, Lou ZY, Liu Z et al (2006) Crystal structure of human pyrroline-5-carboxylate reductase. J Mol Biol 359(5):1364–1377
Nikiforov MA, Chandriani S, O’Conne B et al (2002) A functional screen for Myc-responsive genes reveals serine hydroxymethyl transferase, a major source of the one-carbon unit for cell metabolism. Mol Cell Biol 22:5793–5800
Oppenheim J, Marcus L (1970) Correlation of ultrastructure in Azotobacter vinelandii with nitrogen source for growth. J Bacteriol 101(1):286–291
Orth JD, Conrad TM, Na J (2011) A comprehensive genome-scale reconstruction of Escherichia coli metabolism—2011. Mol Syst Biol 7(1):535. doi:10.1038/msb.2011.65
Petersen LN, Marineo S, Mandalà S et al (2010) The missing link in plant histidine biosynthesis: Arabidopsis myoinositol monophosphatase-like2 encodes a functional histidinol-phosphate phosphatase. Plant Physiol 152(3):1186–1196
Ridgway HF, Olson BH (1981) Scanning electron microscope evidence for bacterial colonization of a drinking-water distribution system. Appl Environ Microbiol 41:274–287
Rodriguez A, Martínez JA, Flores N et al (2014) Engineering Escherichia coli to overproduce aromatic amino acids and derived compounds. Microb Cell Fact 13(1):1–15
Ruszkowski M, Dauter Z (2016) Structural studies of Medicago truncatula histidinol-phosphate phosphatase from inositol monophosphatase superfamily reveal details of penultimate step of histidine biosynthesis in plants. J Biol Chem 291(19):9960–9973
Urbance JW, Bratina BJ, Stoddard SF et al (2001) Taxonomic characterization of Ketogulonigenium vulgare gen. nov. sp. nov. and Ketogulonigenium robustum sp. nov. which oxidize l-sorbose to 2-keto-l-gulonic acid. Int J Syst Evol Microbiol 51(3):1059–1070
Wang J, Alexander P, Wu L, Hammer R, Cleaver O, Mcknight SL (2009) Dependence of mouse embryonic stem cells on threonine catabolism. Science 325(5939):435–439
Wang EX, Ding MZ, Ma Q, Dong XT, Yuan YJ (2016) Reorganization of a synthetic microbial consortium for one-step vitamin c fermentation. Microb Cell Fact 15(1):1–11
Xiong XH, Han S, Wang JH et al (2011) Complete genome sequence of the bacterium Ketogulonicigenium vulgare Y25. J Bacteriol 193(1):315–316
Yang J, Moyana T, MacKenzie S, Xia Q, Xiang J (1998) One hundred seventy-fold increase in excretion of an FV fragment-tumor necrosis factor alpha fusion protein (sFV/TNF-alpha) from Escherichia coli caused by the synergistic effects of glycine and triton X-100. Appl Environ Microbiol 64:2869–2874
Yin GL, Tao ZX, Yan ZZ et al (1990) Fermentation process. US Pat 4935359
Zhou T, Daugherty M, Grishin NV et al (2000) Structure and mechanism of homoserine kinase: prototype for the GHMP kinase superfamily. Structure 8(12):1247–1257
Zou Y, Hu ML, Yuan JL et al (2012) Enhancement of 2-keto-gulonic acid yield by serial subcultivation of co-cultures of Bacillus cereus and Ketogulonicigenium vulgare. Bioresour Technol 132(2):370–373
Acknowledgements
This work was funded by the Ministry of Science and Technology of China (“973” Program: 2014CB745102), the National Natural Science Foundation of China (21390203, 21676190), and the International S&T Cooperation Program of China (2015DFA00960). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pan, CH., Wang, EX., Jia, N. et al. Reconstruction of amino acid biosynthetic pathways increases the productivity of 2-keto-l-gulonic acid in Ketogulonicigenium vulgare-Bacillus endophyticus consortium via genes screening. J Ind Microbiol Biotechnol 44, 1031–1040 (2017). https://doi.org/10.1007/s10295-017-1928-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-017-1928-x