Abstract
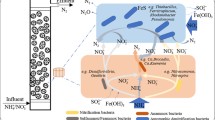
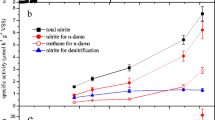
Heterotrophic denitrifying enriched culture (DEC) from a lab-scale high-rate denitrifying reactor was discovered to perform nitrate-dependent anaerobic ferrous oxidation (NAFO). The DEC was systematically investigated to reveal their denitrification activity, their NAFO activity, and the predominant microbial population. The DEC was capable of heterotrophic denitrification with methanol as the electron donor, and autotrophic denitrification with ferrous salt as the electron donor named NAFO. The conversion ratios of ferrous-Fe and nitrate-N were 87.41 and 98.74 %, and the consumption Fe/N ratio was 2.3:1 (mol/mol). The maximum reaction velocity and half saturation constant of Fe were 412.54 mg/(l h) and 8,276.44 mg/l, and the counterparts of N were 20.87 mg/(l h) and 322.58 mg/l, respectively. The predominant bacteria were Hyphomicrobium, Thauera, and Flavobacterium, and the predominant archaea were Methanomethylovorans, Methanohalophilus, and Methanolobus. The discovery of NAFO by heterotrophic DEC is significant for the development of wastewater treatment and the biogeochemical iron cycle and nitrogen cycle.




Similar content being viewed by others
References
An S, Stone H, Nemati M (2011) Biological removal of nitrate by an oil reservoir culture capable of autotrophic and heterotrophic activities: kinetic evaluation and modeling of heterotrophic process. J Hazard Mater 190(1):686–693
Association APH, Federation WPC, Federation WE (1915) Standard methods for the examination of water and wastewater, vol. 2. American Public Health Association, Washington, D.C.
Bae H-S, Im W-T, Suwa Y, Lee JM, Lee S-T, Chang Y-K (2009) Characterization of diverse heterocyclic amine-degrading denitrifying bacteria from various environments. Arch Microbiol 191(4):329–340
Bernardet J-F, Segers P, Vancanneyt M, Berthe F, Kersters K, Vandamme P (1996) Cutting a Gordian knot: emended classification and description of the genus Flavobacterium, emended description of the family Flavobacteriaceae, and proposal of Flavobacterium hydatis nom. nov. (basonym, Cytophaga aquatilis Strohl and Tait 1978). Int J Syst Bacteriol 46(1):128–148
Blöthe M, Roden EE (2009) Composition and activity of an autotrophic Fe(II)-oxidizing, nitrate-reducing enrichment culture. Appl Environ Microbiol 75(21):6937–6940
Chakraborty A, Picardal F (2013) Induction of nitrate-dependent Fe(II) oxidation by Fe(II) in Dechloromonas sp. strain UWNR4 and Acidovorax sp. strain 2AN. Appl Environ Microbiol 79(2):748–752
Chakraborty A, Roden EE, Schieber J, Picardal F (2011) Enhanced growth of Acidovorax sp. strain 2AN during nitrate-dependent Fe(II) oxidation in batch and continuous-flow systems. Appl Environ Microbiol 77(24):8548–8556
Chavarie C, Karamanev D, Godard F, Garnier A, Andre G (1993) Comparison of the kinetics of ferrous iron oxidation by three different strains of Thiobacillus ferrooxidans. Geomicrobiol J 11(1):57–63
Emmerich M, Bhansali A, Lösekann-Behrens T, Schröder C, Kappler A, Behrens S (2012) Abundance, distribution, and activity of Fe(II)-oxidizing and Fe(III)-reducing microorganisms in hypersaline sediments of Lake Kasin, southern Russia. Appl Environ Microbiol 78(12):4386–4399
Feinberg LF, Srikanth R, Vachet RW, Holden JF (2008) Constraints on anaerobic respiration in the hyperthermophilic archaea Pyrobaculum islandicum and Pyrobaculum aerophilum. Appl Environ Microbiol 74(2):396–402
Fesefeldt A, Kloos K, Bothe H, Lemmer H, Gliesche C (1998) Distribution of denitrification and nitrogen fixation genes in Hyphomicrobium spp. and other budding bacteria. Can J Microbiol 44(2):181–186
Fredricks DN, Schubert MM, Myerson D (2005) Molecular identification of an invasive gingival bacterial community. Clin Infect Dis 41(1):e1–e4
Hafenbradl D, Keller M, Dirmeier R, Rachel R, Roßnagel P, Burggraf S, Huber H, Stetter KO (1996) Ferroglobus placidus gen. nov., sp. nov., a novel hyperthermophilic archaeum that oxidizes Fe2+ at neutral pH under anoxic conditions. Arch Microbiol 166(5):308–314
Hedrich S, Schlomann M, Johnson DB (2011) The iron-oxidizing Proteobacteria. Microbiology 157(Pt 6):1551–1564. doi:10.1099/mic.0.045344-0
Horn MA, Ihssen J, Matthies C, Schramm A, Acker G, Drake HL (2005) Dechloromonas denitrificans sp. nov., Flavobacterium denitrificans sp. nov., Paenibacillus anaericanus sp. nov. and Paenibacillus terrae strain MH72, N2O-producing bacteria isolated from the gut of the earthworm Aporrectodea caliginosa. Int J Syst Evol Microbiol 55(3):1255–1265
Isaka K, Kimura Y, Osaka T, Tsuneda S (2012) High-rate denitrification using polyethylene glycol gel carriers entrapping heterotrophic denitrifying bacteria. Water Res 46(16):4941–4948
Jiang B, Parshina S, Van Doesburg W, Lomans B, Stams A (2005) Methanomethylovorans thermophila sp. nov., a thermophilic, methylotrophic methanogen from an anaerobic reactor fed with methanol. Int J Syst Evol Microbiol 55(6):2465–2470
Karpuzcu ME, Stringfellow WT (2012) Kinetics of nitrate removal in wetlands receiving agricultural drainage. Ecol Eng 42:295–303
Keller M, Braun F-J, Dirmeier R, Hafenbradl D, Burggraf S, Rachel R, Stetter KO (1995) Thermococcus alcaliphilus sp. nov., a new hyperthermophilic archaeum growing on polysulfide at alkaline pH. Arch Microbiol 164(6):390–395
Kim B-Y, Weon H-Y, Cousin S, Yoo S-H, Kwon S-W, Go S-J, Stackebrandt E (2006) Flavobacterium daejeonense sp. nov. and Flavobacterium suncheonense sp. nov., isolated from greenhouse soils in Korea. Int J Syst Evol Microbiol 56(7):1645–1649
Li W, Zheng P, Wang L, Zhang M, Lu H, Xing Y, Zhang J, Wang R, Song J, Ghulam A (2013) Physical characteristics and formation mechanism of denitrifying granular sludge in high-load reactor. Bioresource Technol 142:683–687
Lins P, Schwarzenauer T, Reitschuler C, Wagner AO, Illmer P (2012) Methanogenic potential of formate in thermophilic anaerobic digestion. Waste Manag Res 30(10):1031–1040
McCarty PL, Smith DP (1986) Anaerobic wastewater treatment. Environ Sci Technol 20(12):1200–1206
Monod J (1949) The growth of bacterial cultures. Annu Rev Microbiol 3(1):371–394
Neilands JB (1981) Iron absorption and transport in microorganisms. Annu Rev Nutr 1:27–46
Neilands JB (1995) Siderophores: structure and function of microbial iron transport compounds. J Biol Chem 270:26723–26726
O’Reilly J, Lee C, Collins G, Chinalia F, Mahony T, O’Flaherty V (2009) Quantitative and qualitative analysis of methanogenic communities in mesophilically and psychrophilically cultivated anaerobic granular biofilms. Water Res 43(14):3365–3374
Özkaya B, Sahinkaya E, Nurmi P, Kaksonen AH, Puhakka JA (2007) Kinetics of iron oxidation by Leptospirillum ferriphilum dominated culture at pH below one. Biotechnol Bioeng 97(5):1121–1127
Pandey R, Malhotra S, Rajvaidya A, Sharma S, Peshwe S, Raman V, Bal A (2004) Chemo-biochemical desulphurization of various gaseous streams on bench scale. Water Air Soil Pollut 154(1–4):295–311
Paterek JR, Smith PH (1988) Methanohalophilus mahii gen. nov., sp. nov., a methylotrophic halophilic methanogen†. Int J Syst Bacteriol 38(1):122–123
Shen Z, Zhou Y, Wang J (2013) Comparison of denitrification performance and microbial diversity using starch/polylactic acid blends and ethanol as electron donor for nitrate removal. Bioresource Technol 131:33–39
Straub KL, Benz M, Schink B, Widdel F (1996) Anaerobic, nitrate-dependent microbial oxidation of ferrous iron. Appl Environ Microbiol 62(4):1458–1460
Völkl P, Huber R, Drobner E, Rachel R, Burggraf S, Trincone A, Stetter KO (1993) Pyrobaculum aerophilum sp. nov., a novel nitrate-reducing hyperthermophilic archaeum. Appl Environ Microbiol 59(9):2918–2926
Weber KA, Pollock J, Cole KA, O’Connor SM, Achenbach LA, Coates JD (2006) Anaerobic nitrate-dependent iron (II) bio-oxidation by a novel lithoautotrophic betaproteobacterium, strain 2002. Appl Environ Microbiol 72(1):686–694
Whitman WB, Goodfellow M, Kämpfer P, Busse H-J, Trujillo ME, Ludwig W, Suzuki K-i, Parte A (2012) Bergey’s manual® of systematic bacteriology, vol. 5. Springer, Berlin Heidelberg New York
Wu S-Y, Lai M-C (2011) Methanogenic archaea isolated from Taiwan’s Chelungpu fault. Appl Environ Microbiol 77(3):830–838
Acknowledgments
Financial support for this work by the National Natural Science Foundation of China (51278457) and Zhejiang Provincial National Science Foundation (Z5110094) are greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, R., Zheng, P., Xing, YJ. et al. Anaerobic ferrous oxidation by heterotrophic denitrifying enriched culture. J Ind Microbiol Biotechnol 41, 803–809 (2014). https://doi.org/10.1007/s10295-014-1424-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-014-1424-5