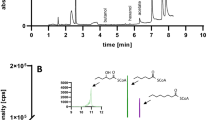
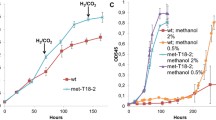
Abstract
The cell energy fraction that powered maintenance and expression of genes encoding pro-phage elements, pta-ack cluster, early sporulation, sugar ABC transporter periplasmic proteins, 6-phosphofructokinase, pyruvate kinase, and fructose-1,6-disphosphatase in acetogen Clostridium sp. MT871 was re-directed to power synthetic operon encoding isobutanol biosynthesis at the expense of these genes achieved via their elimination. Genome tailoring decreased cell duplication time by 7.0 ± 0.1 min (p < 0.05) compared to the parental strain, with intact genome and cell duplication time of 68 ± 1 min (p < 0.05). Clostridium sp. MT871 with tailored genome was UVC-mutated to withstand 6.1 % isobutanol in fermentation broth to prevent product inhibition in an engineered commercial biocatalyst producing 5 % (674.5 mM) isobutanol during two-step continuous fermentation of CO2/H2 gas blend. Biocatalyst Clostridium sp. MT871RG11IBR6 was engineered to express six copies of synthetic operon comprising optimized synthetic format dehydrogenase, pyruvate formate lyase, acetolactate synthase, acetohydroxyacid reductoisomerase, 2,3-dihydroxy-isovalerate dehydratase, branched-chain alpha-ketoacid decarboxylase gene, aldehyde dehydrogenase, and alcohol dehydrogenase, regaining cell duplication time of 68 ± 1 min (p < 0.05) for the parental strain. This is the first report on isobutanol production by an engineered acetogen biocatalyst suitable for commercial manufacturing of this chemical/fuel using continuous fermentation of CO2/H2 blend thus contributing to the reversal of global warming.



Similar content being viewed by others
References
Atsumi S, Li Z, Liao JC (2009) Acetolactate synthase from Bacillus subtilis serves as a 2-ketoisovalerate decarboxylase for isobutanol biosynthesis in Escherichia coli. Appl Environ Microbiol 75(19):6306–6311. doi:10.1128/AEM.01160-09
Berzin V, Kiriukhin M, Tyurin M (2012) Selective production of acetone during continuous synthesis gas fermentation by engineered biocatalyst Clostridium sp. MAceT113. Lett Appl Microbiol. doi:10.1111/j.1472-765X.2012.03272.x
Berzin V, Tyurin M (2012) Acetogen biocatalyst Clostridium sp. MTEtOH871 engineered with our proprietary electrotransformation technology and equipment: continuous synthesis gas fermentation for selective ethanol production. J Biotech Res 4:54–64
Berzin V, Kiriukhin M, Tyurin M (2012) Elimination of acetate production to improve ethanol yield during continuous synthesis gas fermentation by engineered biocatalyst Clostridium sp. MTEtOH550. Appl Biochem Biotechnol 167(2):338–347. doi:10.1007/s12010-012-9697-5
Berzin V, Kiriukhin M, Tyurin M (2013) Cre-lox66/lox71-based elimination of phosphotransacetylase or acetaldehyde dehydrogenase shifted carbon flux in acetogen rendering selective overproduction of ethanol or acetate. Appl Biochem Biotechnol 195(3):181–188. doi:10.1007/s12010-012-9864-8
Berzin V, Kiriukhin M, Tyurin M (2013) Selective n-butanol production by Clostridium sp. MTButOH1365 during continuous synthesis gas fermentation due to expression of synthetic thiolase, 3-hydroxy butyryl-CoA dehydrogenase, crotonase, butyryl-CoA dehydrogenase, butyraldehyde dehydrogenase and NAD-dependent butanol dehydrogenase. Appl Biochem Biotechnol 169(3):950–959. doi:10.1007/s12010-012-0060-7
Berzin V, Kiriukhin M, Tyurin M (2013) “Curing” of plasmid DNA in acetogen using microwave or applying an electric pulse improves cell growth and metabolite production as compared to the plasmid-harboring strain. Arch Microbiol 195(3):181–188. doi:10.1007/s00203-012-0862-6
Blattner FR, Plunkett G III, Bloch CA, Perna NT, Burland V, Riley M, Collado-Vides J, Glasner JD, Rode CK, Mayhew GF, Gregor J, Davis NW, Kirkpatrick HA, Goeden MA, Rose DJ, Mau B, Shao Y (1997) The complete genome sequence of Escherichia coli K-12. Science 277(5331):1453–1462. doi:10.1126/science.277.5331.1453
Brock TD (1989) Evolutionary relationships of the autotrophic bacteria. In: Schlegel HG, Bowien B (eds) Autotrophic bacteria. Science Tech, Madison, pp 499–512
Drake HL (1994) Acetogenesis. Chapman & Hall, London
Müller V (2003) Energy conservation in acetogenic bacteria. Applied and Environmental Microbiology 69(11):6345–6353. doi:10.1128/AEM.69.11.6345- 6353.2003
Gaddy JL, Arora DD, Ko C-W, Phillips JR, Basu R, Wikstrom CV, Clausen EC (2001) Methods for increasing the production of ethanol from microbial fermentation. US Patent 7285402
Grady MC, Jahic M, Patnalk R (2009) Method for producing butanol using two-phase extractive fermentation. Patent publication No.: US 2009/0305370A1
Ikehata H, Ono T (2011) The mechanisms of UV mutagenesis. J Radiat Res 52:115–125
Kiriukhin M, Tyurin M (2013) Mevalonate production by engineered acetogen biocatalyst during continuous fermentation of syngas or CO2/H2 blend. Bioprocess Biosyst Eng. doi:10.1007/s00449-013-0991-6
Li SY, Srivastava R, Suib SL, Li Y, Parnas RS (2011) Performance of batch, fed-batch, and continuous A-B-E fermentation with pH-control. Bioresour Technol 102(5):4241–4250
Minty JJ, Lesnefsky AA, Lin F, Chen Y, Zaroff TA, Veloso AB, Xie B, McConnell CA, Ward RJ, Schwartz DR, Rouillard J-M, Gao Y, Gulari E, Lin XN (2011) Evolution combined with genomic study elucidates genetic bases of isobutanol tolerance in Escherichia coli. Microb Cell Fact 10:18. doi:10.1186/1475-2859-10-18
Müller V (2003) Energy conservation in acetogenic bacteria. Appl Environ Microbiol 69(11):6345–6353. doi:10.1128/AEM.69.11.6345-6353.2003
Nakashima N, Goh S, Good L, Tamura T (2012) Multiple-gene silencing using antisense RNAs in Escherichia coli. Methods Mol Biol 815:307–319. doi:10.1007/978-1-61779-424-7_23
Nevin KP, Hensley SA, Franks AE, Summers ZM, Ou J, Woodard TL, Snoeyenbos-West OL, Lovley DR (2011) Electrosynthesis of organic compounds from carbon dioxide is catalyzed by a diversity of acetogenic microorganisms. Appl Environ Microbiol 77(9):2882–2886. doi:10.1128/AEM.02642-10
Nie H, Zhang T, Cui M, Lu H, Lovley DR, Russell TP (2013) Improved cathode for high efficient microbial-catalyzed reduction in microbial electrosynthesis cells. Phys Chem Chem Phys 15(34):14290–14294. doi:10.1039/c3cp52697f
Polz MF, Alm EJ, Hanage WP (2013) Horizontal gene transfer and the evolution of bacterial and archaeal population structure. Trends Genet 29(3):170–175. doi:10.1016/j.tig.2012.12.006
Ross SM (2000) Introduction to probability and statistics for engineers and scientists, 2nd edn. Academic, San Diego
Savage N (2011) Fuel options: the ideal biofuel. Nature 474(7352):S9–S11. doi:10.1038/474S09a
Savrasova EA, Kivero AD, Shakulov RS, Stoynova NV (2011) Use of the valine biosynthetic pathway to convert glucose into isobutanol. J Ind Microbiol Biotechnol 38(9):1287–1294. doi:10.1007/s10295-010-0907-2
Seaton SC, Silby MW, Levy SB (2013) Pleiotropic effects of GacA on Pseudomonas fluorescens Pf0-1 in vitro and in soil. Appl Environ Microbiol 79(17):5405–5410. doi:10.1128/AEM.00819-13
Simon SA, Gutknecht J (1980) Solubility of carbon dioxide in lipid bilayer membranes and organic solvents. Biochim Biophys Acta 596(3):352–358
Tanner RS, Miller LM, Yang D (1993) Clostridium ljungdahlii sp. nov., an acetogenic species in clostridial rRNA homology group I. Int J Syst Bacteriol 43(2):232–236
Tremblay PL, Zhang T, Dar SA, Leang C, Lovley DR (2012) The Rnf complex of Clostridium ljungdahlii is a proton-translocating ferredoxin:NAD+ oxidoreductase essential for autotrophic growth. MBio 4(1):e00406–e00412. doi:10.1128/mBio.00406-12
Tyurin MV, Doroshenko VG, Oparina NYu (1997) Electrofusion of Escherichia coli cells. Membr Cell Biol 11(1):121–129
Tyurin M, Kiryukhin M, Berzin V (2012) Electrofusion of untreated cells of the newly isolated acetogen Clostridium sp. MT351 with integrated in the chromosome erm(B) or cat leading to the combined presence of these antibiotic resistance genes in the chromosome of the electrofusion products. J Biotech Res 4:1–12
Tyurin M, Kiriukhin M (2013) Ethanol overproduction during continuous syngas fermentation due to expression of amplified ethanol biosynthesis pathway integrated using Tn7-tool and powered at the expense of eliminated acetic acid production and spore formation. J Appl Microbiol. doi:10.1111/jam.12123
Tyurin M, Kiriukhin M (2013) 2,3-Butanediol production by engineered acetogen biocatalyst during continuous fermentation of syngas or CO2/H2 blend. Appl Biochem Biotechnol 170(6):1503–1524. doi:10.1007/s12010-013-0285-0
Tyurin M, Kiriukhin M (2013) Selective methanol or formate production during continuous CO2 fermentation by the acetogen biocatalysts engineered via integration of synthetic pathways using Tn7-tool. World J Microbiol Biotechnol 29(9):1611–1623. doi:10.1007/s11274-013-1324-2
Tyurin M (2013) Gene replacement and elimination using λRed- and FLP-based tool to re-direct carbon flux in acetogen biocatalyst during continuous CO2/H2 blend fermentation. J Ind Microbiol Biotechnol 40(7):749–758. doi:10.1007/s10295-013-1279-1
Tyurin M, Kiriukhin M (2013) Expression of amplified synthetic ethanol pathway integrated using Tn7-tool and powered at the expense of eliminated pta, ack, spo0A and spo0J during continuous syngas or CO2/H2 blend fermentation. J Appl Microbiol 114(4):1033–1045. doi:10.1111/jam.12123
Tyurin M (2013) Reversal of global warming using $3 trillion market force: chemicals and fuels produced directly and selectively in continuous fermentations of gas blends comprising CO and CO2. In: Carter B, Keyes and Olivia C, Lucero (eds) Environmental aspects of global warming. Nova Science, New Developments in Global Warming Research. https://www.novapublishers.com/catalog/product_info.php?products_id=42716
Gak E, Tyurin M, Kiriukhin M (2014) UV-induced mutagenesis in acetogens: resistance to methanol, ethanol, acetone, or n-butanol in recombinants with reduced genomes during continuous CO2/H2 gas blend fermentation. World J Microbiol Biotechnol. doi:10.1007/s11274-013-1579-7
Young KD (2006) The selective value of bacterial shape. Microbiol Mol Biol Rev 70(3):660–703. doi:10.1128/MMBR.00001-06
Zimmerman E @CNNMoneyTech June 25, 2013. http://money.cnn.com/2013/06/25/technology/enterprise/low-cost-genome-sequencing/
Acknowledgments
The research was supported by the angel funds of MT family friends. Biocatalyst Gurus, Inc. is the sole distributor of the electroporation and electrofusion equipment, custom multi-fermentor modules and MT254UVnator: www.syngasbiofuelsenergy.com.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors have made equal contribution to the reported project.
Rights and permissions
About this article
Cite this article
Gak, E., Tyurin, M. & Kiriukhin, M. Genome tailoring powered production of isobutanol in continuous CO2/H2 blend fermentation using engineered acetogen biocatalyst. J Ind Microbiol Biotechnol 41, 763–781 (2014). https://doi.org/10.1007/s10295-014-1416-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-014-1416-5