Abstract
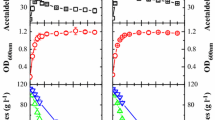
Acetaldehyde is relevant for wine aroma, wine color, and microbiological stability. Yeast are known to play a crucial role in production and utilization of acetaldehyde during fermentations but comparative quantitative data are scarce. This research evaluated the acetaldehyde metabolism of 26 yeast strains, including commercial Saccharomyces and non-Saccharomyces, in a reproducible resting cell model system. Acetaldehyde kinetics and peak values were highly genus, species, and strain dependent. Peak acetaldehyde values varied from 2.2 to 189.4 mg l−1 and correlated well (r 2 = 0.92) with the acetaldehyde production yield coefficients that ranged from 0.4 to 42 mg acetaldehyde per g of glucose in absence of SO2. S. pombe showed the highest acetaldehyde production yield coefficients and peak values. All other non-Saccharomyces species produced significantly less acetaldehyde than the S. cerevisiae strains and were less affected by SO2 additions. All yeast strains could degrade acetaldehyde as sole substrate, but the acetaldehyde degradation rates did not correlate with acetaldehyde peak values or acetaldehyde production yield coefficients in incubations with glucose as sole substrate.





Similar content being viewed by others
References
Anonymous (2009) The maximum sulphur dioxide content of wines. Off J Eur Union L193 Annex I B:26–29
Bely M, Stoeckle P, Masneuf-Pomarede I, Dubourdieu D (2008) Impact of mixed Torulaspora delbrueckii-Saccharomyces cerevisiae culture on high-sugar fermentation. Int J Food Microbiol 122:312–320
Bisson LF (2004) Biotechnology of wine yeast. Food Biotechnol 18:63–96
Cabranes C, Moreno J, Mangas JJ (1998) Cider production with immobilized Leuconostoc oenos. J Inst Brew 104:127–130
Cheraiti N, Guezenec S, Salmon JM (2010) Very early acetaldehyde production by industrial Saccharomyces cerevisiae strains: a new intrinsic character. Appl Microbiol Biotechnol 86:693–700
Cheraiti N, Sauvage FX, Salmon JM (2008) Acetaldehyde addition throughout the growth phase alleviates the phenotypic effect of zinc deficiency in Saccharomyces cerevisiae. Appl Microbiol Biotechnol 77:1093–1109
Clarke RJ, Bakker J (2004) Wine flavour chemistry. Blackwell, Oxford
Cortes MB, Moreno J, Zea L, Moyano L, Medina M (1998) Changes in aroma compounds of sherry wines during their biological aging carried out by Saccharomyces cerevisiae races bayanus and capensis. J Agric Food Chem 46:2389–2394
Delfini C, Formica JV (2001) Wine microbiology: science and technology. Marcel Dekker, New York
Dharmadhikari MR, Wilker KL (1998) Deacidification of high malate must with Schizosaccharomyces pombe. Am J Enol Vitic 49:408–412
Diaz-Montano DM, Delia ML, Estarron-Espinosa M, Strehaiano P (2008) Fermentative capability and aroma compound production isolated from Agave tequilana Weber juice. Enzyme Microb Technol 42:608–616
Ferreira ACD, Barbe JC, Bertrand A (2002) Heterocyclic acetals from glycerol and acetaldehyde in port wines: evolution with aging. J Agric Food Chem 50:2560–2564
Fleet GH (1993) Wine microbiology and biotechnology. Harwood, Chur
Fleet GH (2008) Wine yeasts for the future. FEMS Yeast Res 8:979–995
Herrero M, Garcia LA, Diaz M (2003) The effect of SO2 on the production of ethanol, acetaldehyde, organic acids, and flavor volatiles during industrial cider fermentation. J Agric Food Chem 51:3455–3459
Jussier D, Dubé Morneau A, Mira de Orduña R (2006) Effect of simultaneous inoculation of yeast and bacteria on fermentation kinetics and key wine parameters during white winemaking. Appl Environ Microbiol 72:221–227
Lachenmeier DW, Kanteres F, Rehm J (2009) Carcinogenicity of acetaldehyde in alcoholic beverages: risk assessment outside ethanol metabolism. Addiction 104:533–550
Lachenmeier DW, Sohnius EM (2008) The role of acetaldehyde outside ethanol metabolism in the carcinogenicity of alcoholic beverages: evidence from a large chemical survey. Food Chem Toxicol 46:2903–2911
Li E, Mira de Orduña R (2010) A rapid method for the determination of microbial biomass by dry weight using a moisture analyzer with an infrared heating source and an analytical balance. Lett Appl Microbiol 50:283–288
Liu S-Q, Pilone GJ (2000) An overwiew of formation and roles of acetaldehyde in winemaking with emphasis on microbiological implications. Int J Food Sci Technol 35:49–61
Longo E, Velazquez JB, Sieiro C, Cansado J, Calo P, Villa TG (1992) Production of higher alcohols, ethyl-acetate, acetaldehyde and other compounds by 14 Saccharomyces cerevisiae wine strains isolated from the same region (Salnes, NW Spain). World J Microbiol Biotechnol 8:539–541
Mira de Orduña R, Liu S-Q, Patchett ML, Pilone GJ (2000) Kinetics of the arginine metabolism of malolactic wine lactic acid bacteria Lactobacillus buchneri CUC-3 and Oenococcus oeni Lo111. J Appl Microbiol 89:547–552
Miyake T, Shibamoto T (1993) Quantitative analysis of acetaldehyde in foods and beverages. J Agric Food Chem 41:1968–1970
Moreno-Arribas MV, Polo MC (2008) Wine chemistry and biochemistry. Springer, New York
Neuberg C, Hirsch J, Reinfurth E (1920) The three fermentation-forms of sugar, their coherences and balance. Biochem Zeitschr 105:307–336
Neuberg C, Reinfurth E (1918) The determination of the aldehyde stage in alcoholic fermentation an experimental argument for the acetaldehyde pyrunic acid theory. Biochem Zeitschr 89:365–414
Osborne JP, Dubé Morneau A, Mira de Orduña R (2006) Degradation of free and sulphur-dioxide-bound acetaldehyde by malolactic lactic acid bacteria in white wine. J Appl Microbiol 101:474–479
Osborne JP, Mira de Orduña R, Liu S-Q, Pilone GJ (2000) Acetaldehyde metabolism by wine lactic acid bacteria. FEMS Microbiol Lett 191:51–55
Pretorius IS (2000) Tailoring wine yeast for the new millennium: novel approaches to the ancient art of winemaking. Yeast 16:675–729
Romano P, Caruso M, Capece A, Lipani G, Paraggio M, Fiore C (2003) Metabolic diversity of Saccharomyces cerevisiae strains from spontaneously fermented grape musts. World J Microbiol Biotechnol 19:311–315
Romano P, Suzzi G, Turbanti L, Polsinelli M (1994) Acetaldehyde production in Saccharomyces cerevisiae wine yeasts. FEMS Microbiol Lett 118:213–218
Romero C, Bakker J (1999) Interactions between grape anthocyanins and pyruvic acid, with effect of pH and acid concentration on anthocyanin composition and color in model solutions. J Agric Food Chem 47:3130–3139
Saucier C, Little D, Glories Y (1997) First evidence of acetaldehyde-flavanol condensation products in red wine. Am J Enol Vitic 48:370–373
Schlegel HG (1992) Allgemeine Mikrobiologie. Thieme, Stuttgart
Somers TC, Wescombe LG (1982) Red wine quality: the critical role of SO2 during vinification and conservation. Austr Grapegrower Winemaker 19:68–74
Then R, Radler F (1970) Regulation of acetaldehyde concentration in culture medium during fermentation of glucose by Saccharomyces cerevisiae. Archiv fur Mikrobiologie 72:60–67
Timberlake CF, Bridle P (1977) Anthocyanins: colour augmentation with catechin and acetaldehyde. J Sci Food Agric 28:539–544
Valles BS, Bedrinana RP, Queipo AL, Alonso JJM (2008) Screening of cider yeasts for sparkling cider production (Champenoise method). Food Microbiol 25:690–697
Wildenradt HL, Singleton VL (1974) The production of aldehydes as a result of oxidation of polyphenolic compounds and its relation to wine aging. Am J Enol Vitic 25:119–126
Acknowledgments
The authors wish to thank the China Scholarship Council for supporting Erhu Li (State Scholarship Fund No. 2008630061). The authors also wish to thank Lallemand Inc. and Prof. Yanlin Liu for providing yeast strains.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, E., Mira de Orduña, R. Evaluation of the acetaldehyde production and degradation potential of 26 enological Saccharomyces and non-Saccharomyces yeast strains in a resting cell model system. J Ind Microbiol Biotechnol 38, 1391–1398 (2011). https://doi.org/10.1007/s10295-010-0924-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-010-0924-1