Abstract
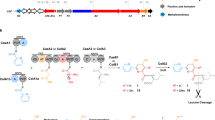
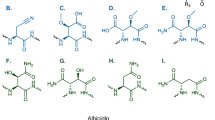
Precursor-directed biosynthesis has been shown to be a powerful tool for the production of polyketide analogues that would be difficult or cost prohibitive to produce from medicinal chemistry efforts alone. It has been most extensively demonstrated using a KS1 null mutation (KS10) to block the first round of condensation in the biosynthesis of the erythromycin polyketide synthase (DEBS) for the production of analogues of its aglycone, 6-deoxyerythronolide B (6-dEB). Here we show that removing the DEBS loading domain and first module (mod1Δ), rather than using the KS10 system, can lead to an increase in the utilization of some chemical precursors and production of 6-dEB analogues (R-6dEB) in both Streptomyces coelicolor and Saccharopolyspora erythraea. While the difference in utilization of the precursor was diketide specific, in strains fed (2R*, 3S*)-5-fluoro-3-hydroxy-2-methylpentanoate N-propionylcysteamine thioester, twofold increases in both utilization of the diketide and 15-fluoro-6dEB (15F-6dEB) production were observed in S. coelicolor, and S. erythraea exhibited a tenfold increase in production of 15-fluoro-erythromycin when utilizing the mod1Δ rather than the KS10 system.

Similar content being viewed by others
References
Burlingame MA, Medoza E, Ashley GW (2004) N-Acyl-2-benzoxalinones in titanium-mediated aldol reactions. Tetrahedron Lett 45:2961–2964
Cane DE, Yang CC (1987) Macrolide biosynthesis. 4. Intact incorporation of a chain-elongation intermediate into erythromycin. J Am Chem Soc 109:1255–1257
Chuck JA, McPherson M, Huang H, Jacobsen JR, Khosla C, Cane DE (1997) Molecular recognition of diketide substrates by a beta-ketoacyl-acyl carrier protein synthase domain within a bimodular polyketide synthase. Chem Biol 4:757–766
Desai R, Leaf T, Hu Z, Hutchinson CR, Hong A, Byng G, Galazzo J, Licari P (2004) Combining classical, genetic, and process strategies for improved precursor-directed production of 6-deoxyerythronolide B analogues. Biotechnol Prog 20:38–43
Desai R, Leaf T, Woo E, Licari P (2002) Enhanced production of heterologous macrolide aglycone by fed-batch cultivation of Streptomyces coelicolor. J Ind Microbiol Biotechnol 28:297–301
Flett F, Mersinias V, Smith CP (1997) High efficiency inter-generic conjugal transfer of plasmid DNA from Escherichia coli to methyl-restricting streptomycetes. FEMS Microbiol Lett 155:223–229
Frykman S, Leaf T, Carreras C, Licari P (2001) Precursor-directed production of erythromycin analogs by Saccharopolyspora erythraea. Biotechnol Bioeng 76:303–310
Gokhale RS, Tsuji SY, Cane DE, Khosla C (1999) Dissecting and exploiting intermodular communication in polyketide synthases. Science 284:482–485
Hu Z, Hopwood DA, Hutchinson CR (2003) Enhanced heterologous polyketide production in Streptomyces by exploiting plasmid co-integration. J Ind Microbiol Biotechnol 30:512–522
Hu Z, Pfeifer B, Chao E, Murli S, Kealey J, Carney J, Ashley G, Khosla C, Hutchinson CR (2003) A specific role of the Saccharopolyspora erythraea thioesterase II gene in the function of modular polyketide synthases. Microbiology 149:2213–2225
Jacobsen JR, Hutchinson CR, Cane DE, Khosla C (1997) Precursor-directed biosynthesis of erythromycin analogs by an engineered polyketide synthase. Science 277:367–369
Kao CM, Katz L, Khosla C (1994) Engineered biosynthesis of a complete macrolactone in a heterologous host. Science 265:509–512
Kao CM, Pieper R, Khosla C (1996) Evidence for two catalytically independent clusters of active sites in a functional modular polyketide synthase. Biochemistry 35:12363–12368
Katz L, McDaniel R (1999) Novel macrolides through genetic engineering. Med Res Rev 19:543–558
Kieser T, Bibb MJ, Buttner MJ, Chater KF, Hopwood DA (2000) Practical Streptomyces genetics. Crowesu, Norwich, England
Kinoshita K, Pfeifer BA, Khosla C, Cane DE (2003) Precursor-directed polyketide biosynthesis in Escherichia coli. Bioorg Med Chem Lett 13:3701–3704
Leaf T, Burlingame M, Desai R, Regentin R, Woo E, Ashley G, Licari P (2002) Employing racemic precursors in directed biosynthesis of 6-dEB anaolgs. J Chem Technol Biotechnol 77:1122–1126
Leaf T, Cadapan L, Carreras C, Regentin R, Ou S, Woo E, Ashley G, Licari P (2000) Precursor-directed biosynthesis of 6-deoxyerthronolide B analogs in Streptomyces coelicolor: understanding precursor effects. Biotechnol Prog 16:553–556
Moore BS, Hertweck C (2002) Biosynthesis and attachment of novel bacterial polyketide synthase starter units. Nat Prod Rep 19:70–99
O’Hagan D (1991) The polyketide metabolites. Ellis Horwood, New York
Pfeifer BA, Admiraal SJ, Gramajo H, Cane DE, Khosla C (2001) Biosynthesis of complex polyketides in a metabolically engineered strain of E. coli. Science 291:1683
Pfeifer BA, Khosla C (2001) Biosynthesis of polyketides in heterologous hosts. Microbiol Mol Biol Rev 65:106–118
Regentin R, Kennedy J, Wu N, Carney J, Licari P, Galazzo J, Desai R (2004) Precursor-directed biosynthesis of novel triketide lactones. Biotechnol Prog 20:122–127
Rodriguez E, Hu Z, Ou S, Volchegursky Y, Hutchinson CR, McDaniel R (2003) Rapid engineering of polyketide overproduction by gene transfer to industrially optimized strains. J Ind Microbiol Biotechnol 30:480–488
Staunton J, Weissman KJ (2001) Polyketide Biosynthesis: a millennium review. Nat Prod Rep 18:380–416
Walsh CT (2004) Polyketide and nonribosomal peptide antibiotics: modularity and versatility. Science 303:1805–1810
Weissman KJ, Bycroft M, Cutter AL, Hanefeld U, Frost EJ, Timoney MC, Harris R, Handa S, Roddis M, Staunton J, Leadlay PF (1998) Evaluating precursor-directed biosynthesis towards novel erythromycins through in vitro studies on a bimodular polyketide synthase. Chem Biol 5:743–754
Acknowledgments
We would like to thank John Carney and Chau Tran for assistance with compound analysis, Chaitan Khosla for the gift of DEBS1+TE polyclonal antibody, and Sumati Murli for her helpful comments during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ward, S.L., Desai, R.P., Hu, Z. et al. Precursor-directed biosynthesis of 6-deoxyerythronolide B analogues is improved by removal of the initial catalytic sites of the polyketide synthase. J Ind Microbiol Biotechnol 34, 9–15 (2007). https://doi.org/10.1007/s10295-006-0156-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-006-0156-6