Abstract
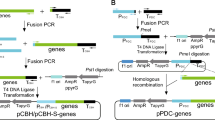
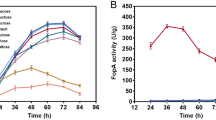
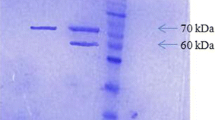
In an effort to produce phytases cost-effectively, and to determine the efficiency of a novel expression system, the genes for Aspergillus awamori (phyA) phytase and Aspergillus fumigatus (phyA) phytase (a putative thermostable enzyme) were cloned and overexpressed in A. awamori. Regulation of phytase expression was achieved by separately placing the genes under the transcriptional control of the glucoamylase A (glaA) promoter of A. awamori. A gene fusion strategy was employed that involved the insertion of a hexapeptide Kex-2 protease cleavage site between the native glucoamylase and heterologous proteins and allowed for the efficient secretion and processing of the resultant chimeric proteins produced in this system by an endogenous Kex-2 protease. The genes for both of the above-mentioned phytases have already been cloned; however, this is the first report of either of the two phytases being fused with the glucoamylase gene, placed under the transcriptional control of the glaA promoter and overexpressed in A. awamori. Following transformation of A. awamori with separate expression vectors (one for each phytase), induction of phytase expression in submerged culture was effected by utilisation of a starch-containing medium. Optimisation of heterologous protein production in small shake-flask cultures involved changes in medium constituents. Maximum phytase expression levels of 200 phytase units (PU) ml−1 and 62 PU ml–1 for recombinantly expressed phytases from A. awamori and A. fumigatus, respectively, were obtained in crude fermentation extracts. Subsequent process scale-up to 4 l batch fermentation yielded phytase production levels comparable to those obtained on small scale. The enzyme yields herein reported demonstrate that the expression system developed and the host strain utilised were capable of expressing phytase at levels comparable to, or exceeding, previously reported data.





Similar content being viewed by others
References
Berka RM, Rey MW, Brown KM, Byun T, Klotz AV (1998) Molecular characterisation and expression of a phytase gene from the thermophilic fungus Thermomyces lanuginosus. Appl Environ Microbiol 64:4423–4427
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilising the principle of protein-dye binding. Anal Biochem 72:248–254
Chelius MK, Wodzinski RJ (1994) Strain improvement of Aspergillus niger for phytase production. Appl Microbiol Biotechnol 41:79–83
Conneely OM (1992) From DNA to feed conversion: using biotechnology to improve enzyme yields and livestock performance. In: Lyons TP (ed) Biotechnology in the feed industry. Proceedings of Alltech's 8th annual symposium, Nicholasville, Ky., pp 57–66
Cromwell GL, Coffey RD (1991) Phosphorous, a key essential nutrient, yet a possible major pollutant—its central role in animal nutrition. In: Lyons TP (ed) Biotechnology in the feed industry. Proceedings of Alltech's 7th annual symposium, Nicholasville, Ky., pp 133–145
Dvorakova J (1998) Phytase: sources, preparation and exploitation. Folia Microbiol 43:323–338
Graf E (1986) Chemistry and applications of phytic acid: an overview. In: Graf E (ed) Phytic acid, chemistry and applications. Pilatus Press, Minneapolis, Minn., pp 1–21
Han YM, Lei XG (1999) Role of glycosylation in the functional expression of an Aspergillus niger phytase (phyA) in Pichia pastoris. Arch Biochem Biophys 364:83–90
Han YM, Wilson DB, Lei XG (1999) Expression of an Aspergillus niger phytase gene (phyA) in Saccharomyces cerevisiae. Appl Environ Microbiol 65:1915–1918
Heinonen JK, Lahti RJ (1981) A new and convenient colorimetric determination of inorganic orthophosphate and its application to the assay of inorganic pyrophosphatase. Anal Biochem 113:313–317
Kim DH, Oh BC, Choi WC, Lee JK, Oh TK (1999) Enzymatic evaluation of Bacillus amyloliquefaciens phytase as a feed additive. Biotechnol Lett 21:925–927
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Li J, Hegeman CE, Hanlon RW, Lacy GH, Denbow DM, Grabau EA (1997) Secretion of active recombinant phytase from soybean cell suspension cultures. Plant Physiol 114:1103–1111
Liu B-L, Rafiq A, Tzeng Y-M, Rob A (1998) The induction and characterisation of phytase and beyond. Enzyme Microb Technol 22:415–424
Maenz DD (2001) Enzymatic characteristics of phytases as they relate to their use in animal feeds. In: Bedford MR, Partridge GG (eds) Enzymes in farm animal nutrition. CABI Publishing, Oxon, UK, pp 61–84
Miettinen-Oinonen A, Torkkeli T, Paloheimo M, Nevalainen H (1997) Overexpression of the Aspergillus niger pH 2.5 acid phosphatase gene in a heterologous host Trichoderma reesei. J Biotechnol 58:13–20
Moore E, Helly VR, Conneely OM, Ward PP, Power RF, Headon DR (1995) Molecular cloning, expression and evaluation of phosphohydrolases for phytate-degrading activity. J Ind Microbiol Biotechnol 14:396–402
Nelson TS, Shieh TR, Wodzinski RJ, Ware JH (1968) The availability of phytate phosphorous in soybean meal before and after treatment with a mold phytase. Poult Sci 47:1842–1848
Pasamontes L, Haiker M, Wyss M, Tessier M, van Loon APGM (1997) Gene cloning, purification and characterisation of a heat-stable phytase from the fungus Aspergillus fumigatus. Appl Environ Microbiol 63:1696–1700
Pen J, Verwoerd TC, van Paridon PA, Beudeker RF, van den Elzen PJM, Geerse K, van der Klis JD, Versteegh HAJ, van Ooyen AJJ, Hoekema A (1993) Phytase-containing transgenic seeds as a novel feed additive for improved phosphorus utilization. Biol Technol 11:811–814
Piddington CS, Houston CS, Paloheimo M, Cantrell M, Miettinen-Oinonen A, Nevalainen H, Rambosek J (1993) The cloning and sequencing of the genes encoding phytase (phy) and pH 2.5-optimum acid phosphatase (aph) from Aspergillus niger var awamori. Gene 133:55–62
Rasmussen CD, Means RL, Lu KP, May GS, Means AR (1990) Characterisation and expression of the unique calmodulin gene of Aspergillus nidulans. J Biol Chem 265:13767–13775
Ravindran V, Bryden WL, Kornegay ET (1995) Phytates: occurrence, bioavailability and implications in poultry nutrition. Poult Avian Biol Rev 6:125–143
Rodriguez E, Mullaney EJ, Lei XG (2000) Expression of the Aspergillus fumigatus phytase gene in Pichia pastoris and characterisation of the recombinant enzyme. Biochem Biophys Res Commun 268:373–378
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
Tilburn J, Scazzocchio C, Taylor GG, Zabicky-Zissman JH, Lockington RA, Davies RW (1983) Transformation by integration in Aspergillus nidulans. Gene 26:205–221
Van Dijck PWM (1999) Chymosin and phytase: made by genetic engineering (no. 10 in a series of articles to promote a better understanding of the use of genetic engineering). J Biotechnol 67:77–80
Van Gorcom RFM, van Hartingsveldt W, van Paridon PA, Veenstra AE, Luiten RGM, Selten GCM (1991) Cloning and expression of microbial phytase. EPA 0 420 358, EPA, Washington, D.C.
Van Hartingsveldt W, van Zeijl CMJ, Harteveld GM, Gouka RJ, Suykerbuyk MEG, Luiten RGM, van Paridon PA, Selten GCM, Veenstra AE, van Gormon RFM, van den Hondel CAMJJ (1993) Cloning, characterisation and overexpression of the phytase-encoding gene (phyA) of Aspergillus niger. Gene 127:87–94
Verwoerd TC, van Paridon PA, van Ooyen AJJ, van Lent JWM, Hoekema A, Pen J (1995) Stable accumulation of Aspergillus niger phytase in transgenic tobacco leaves. Plant Physiol 109:1199–1205
Vohra A, Satyanarayana T (2003) Phytases: microbial sources, production, purification and potential biotechnological applications. Crit Rev Biotechnol 23:29–60
Ward PP, Piddington CS, Cunningham GA, Zhou X, Wyatt RD, Conneely OM (1995) A system for production of commercial quantities of human lactoferrin: a broad spectrum natural antibiotic. Biotechnology 13:498–503
Wodzinski RJ, Ullah AHJ (1996) Phytase. Adv Appl Microbiol 42:263–302
Wyss M, Pasamontes L, Remy R, Kohler J, Kusznir E, Gadient M, Muller F, van Loon APGM (1998) Comparison of the thermostability properties of three acid phosphatases from molds: Aspergillus fumigatus phytase, A. niger phytase and A. niger pH 2.5 acid phosphatase. Appl Environ Microbiol 64:4446–4451
Wyss M, Brugger R, Kronenberger A, Remy R, Fimbel R, Oesterhelt G, Lehmann M, van Loon APGM (1999) Biochemical characterisation of fungal phytases (myo-inositol hexakisphosphate phosphohydrolases): catalytic properties. Appl Environ Microbiol 65:367–373
Acknowledgement
The financial support of Alltech Inc. is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Martin, J.A., Murphy, R.A. & Power, R.F.G. Cloning and expression of fungal phytases in genetically modified strains of Aspergillus awamori . J IND MICROBIOL BIOTECHNOL 30, 568–576 (2003). https://doi.org/10.1007/s10295-003-0083-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10295-003-0083-8