Abstract
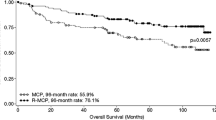
Maintenance treatment aims to sustain the response of induction chemotherapy, reduce the relapse rate, and provide a longer progression-free survival and time to the next course of chemotherapy. It must have an acceptable toxicity profile in order to obtain the trust of both patient and physician over the long term; it must maintain the patient in remission. An ideal maintenance treatment would be effective as monotherapy, with minimal side effects in the short and long term, and easy to administer, requiring minimal supervision. Different drugs are used for non-hodgkin lymphoma, but the results are sometimes controversial; the first was interferon alpha, which is not a standard treatment. More recently, rituximab has demonstrated a significant improvement in progression-free survival, but no improvement in overall survival in the treatment of follicular NHL. The PRIMA study confirms this data: progression-free survival at 3 years was 75% (against 58% in the control arm). So, rituximab can be used as standard maintenance therapy. Other molecules are also developed for this purpose, such as enzastaurin and lenalidomide, for which we do not yet have a definitive evaluation.
Résumé
Le traitement de maintenance a pour objectifs de pérenniser la réponse après une chimiothérapie d’induction, de diminuer le taux de rechute, d’allonger la progression-free survival (PFS) ou survie sans progression. Il doit avoir un profil de toxicité acceptable afin d’obtenir une adhésion du patient et de son praticien sur le long terme, et maintenir le malade en rémission. Dans les lymphomes malins non hodgkiniens, différentes molécules sont utilisées avec des résultats divers; la première d’entre elles fut l’interféron alpha, dont l’utilisation délicate n’a pas permis d’en faire un standard de traitement. Plus récemment, le rituximab en maintenance a démontré une nette amélioration de la PFS, sans amélioration de l’overall survival (OS) ou survie globale dans le traitement du LMNH folliculaire. L’essai PRIMA confirme ses données: la PFS à trois ans est de 75 % (contre 58 % dans le bras témoin). Il permet de définir un standard de traitement du traitement de maintenance par rituximab. D’autres molécules sont développées également dans cet objectif comme l’enzastaurin, le lénalidomide, pour lesquelles nous ne disposons pas encore d’évaluation définitive.
Similar content being viewed by others
Références
Cheson BD, Horning SJ, Coiffier B, et al. (1999) Report of an international workshop to standardize response criteria for non-Hodgkin’s lymphomas. NCI Sponsored International Working Group. J Clin Oncol 4: 1244
Cheson BD, Pfistner B, Juweid ME, et al (2007) Revised response criteria for malignant lymphoma. J Clin Oncol 5: 579–586
Forstpointner R, Unterhalt M, Dreyling M, et al. (2006) Maintenance therapy with rituximab leads to a significant prolongation of response duration after salvage therapy with a combination of rituximab, fludarabine, cyclophosphamide, and mitoxantrone (R-FCM) in patients with recurring and refractory follicular and mantle cell lymphomas: results of a prospective randomized study of the German Low Grade Lymphoma Study Group (GLSG). Blood 108: 4003–4008
Ghielmini M, Schmitz S-FH, Cogliatti SB, et al (2004) Prolonged treatment with rituximab in patients with follicular lymphoma significantly increases event-free survival and response duration compared with the standard weekly x 4 schedule. Blood 103: 4416–4423
Gordan LN, Grow WB, Pusateri A, et al. (2005) Phase II trial of individualized rituximab dosing for patients with CD20-positive lymphoproliferative disorders. J Clin Oncol 6: 1096–1102
Hainsworth JD, Litchy S, Shaffer DW, et al (2005) Maximizing therapeutic benefit of rituximab: maintenance therapy versus re-treatment at progression in patients with indolent non-Hodgkin’s lymphoma-a randomized phase II trial of the Minnie Pearl Cancer Research Network. J Clin Oncol 23: 1088–1095
Hochster H, Weller E, Gascoyne RD, et al. (2009) Maintenance rituximab after cyclophosphamide, vincristine, and prednisone prolongs progression-free survival in advanced indolent lymphoma: results of the randomized phase III ECOG1496 Study. J Clin Oncol 10: 1607–1614
Lynch JW, Hei DL, Braylan RC, et al (2002) Phase II study of fludarabine combined with interferon-alpha-2a followed by maintenance therapy with interferonalpha-2a in patients with low-grade nonhodgkin’s lymphoma. Am J Clin Oncol 4: 391–397
Rohatiner AZS, Gregory WM, Peterson B, et al. (2005) Meta-analysis to evaluate the role of interferon in follicular lymphoma. J Clin Oncol 10: 2215–2223
Salles G, Seymour JF, Offner F, et al (2011) Rituximab maintenance for 2 years in patients with high tumour burden follicular lymphoma responding to rituximab plus chemotherapy (PRIMA): a phase 3 randomised controlled trial. Lancet 377: 42–51
Solal-Celigny P, Lepage E, Brousse N, et al. (1993) Recombinant interferon alfa-2b combined with a regimen containing doxorubicin in patients with advanced follicular lymphoma. Groupe d’étude des lymphomes de l’adulte. N Engl J Med 22: 1608–1614
Solal-Celigny P, Lepage E, Brousse N, et al (1998) Doxorubicin-containing regimen with or without interferon alfa-2b for advanced follicular lymphomas: final analysis of survival and toxicity in the Groupe d’étude des lymphomes folliculaires 86 Trial. J Clin Oncol 7: 2332–2338
van Oers MHJ, Klasa R, Marcus RE, et al. (2006) Rituximab maintenance improves clinical outcome of relapsed/resistant follicular non-Hodgkin lymphoma in patients both with and without rituximab during induction: results of a prospective randomized phase 3 intergroup trial. Blood 10: 3295–3301
van Oers MHJ, Van Glabbeke M, Giurgea L, et al (2010) Rituximab maintenance treatment of relapsed/resistant follicular non-Hodgkin’s lymphoma: long-term outcome of the EORTC 20981 phase III randomized intergroup study. J Clin Oncol 17: 2853–2858
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Pascal, L., Terriou, L. & Morschhauser, F. Traitement de maintenance des lymphomes malins non hodgkiniens. Oncologie 13, 543–547 (2011). https://doi.org/10.1007/s10269-011-2062-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10269-011-2062-4