Abstract
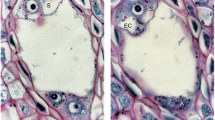
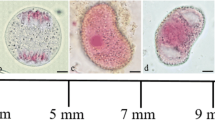
Pilobolus crystallinus has three putative glyceraldehyde-3-phosphate dehydrogenase (gapdh) genes (pcgapdh1, pcgapdh2 and pcgapdh3). The results of this study demonstrate that expression of pcgapdh2 was increased by irradiation and that this increased expression was correlated with the formation of asexual reproductive organs (trophocysts). Interestingly, expression of pcgapdh2 was restricted to trophocysts. The formation of trophocysts was likely promoted by light, and the expression of pcgapdh2 was increased as a result of trophocyst formation. This is the first report that shows the regulation of a gapdh gene in an organ-specific manner in fungi.



Similar content being viewed by others
References
Boer PH, Adra CN, Lau Y-F, McBurney MW (1987) The testis-specific phosphoglycerate kinase gene pgk-2 is a recruited retroposon. Mol Cell Biol 7:3107–3112
De Maeseneire SL, Dauvrin T, Jonniaux J-L, Van Bogaert INA, Soetaert WK, Vandamme EJMC (2008) Cloning and characterization of the glyceraldehyde-3-phosphate dehydrogenase gene and the use of its promoter for expression in Myrothecium gramineum, a novel expression host. FEMS Microbiol Lett 281:140–146
Delgado ML, O’Connor JE, Azorín I, Renau-Piqueras J, Gil ML, Gozalbo D (2001) The glyceraldehyde-3-phosphate dehydrogenase polypeptides encoded by the Saccharomyces cerevisiae TDH1, TDH2 and TDH3 genes are also cell wall proteins. Microbiology 147:411–417
Diez M, Rua J, Busto F, Soler J (1988) Analysis of the effect of light on carbohydrate metabolism in Phycomyces sporangiophores and mycelia. Int J Biochem 20:801–806
Eddy EM, Welch JE, O’Brien DA (1994) Role and regulation of spermatogenic cell-specific enzymes of glycolysis. In: Bartke A (ed) Function of somatic cells in the testis. Springer, New York, pp 362–372
Fernandes PA, Sena-Esteves M, Moradas-Ferreira P (1995) Characterization of the glyceraldehyde-3-phosphate dehydrogenase gene family from Kluyveromyces marxianus: polymerase chain reaction-single-strand conformation polymorphism as a tool for the study of multigenic families. Yeast 11:725–733
Gil ML, Delgado ML, Gozalbo D (2001) The Candida albicans cell wall-associated glyceraldehyde-3-phosphate dehydrogenase activity increases in response to starvation and temperature upshift. Med Mycol 39:387–394
Harmsen MC, Schuren FH, Moukha SM, van Zuilen CM, Punt PJ, Wessels JG (1992) Sequence analysis of the glyceraldehyde-3-phosphate dehydrogenase genes from the basidiomycetes Schizophyllum commune, Phanerochaete chrysosporium and Agaricus bisporus. Curr Genet 22:447–454
Hirano T, Sato T, Okawa K, Kanda K, Yaegashi K, Enei H (1999) Isolation and characterization of the glyceraldehydes-3-phosphate dehydrogenase gene of Lentinus edodes. Biosci Biotechnol Biochem 63:1223–1227
Ishitani R, Chuang D-M (1996) Glyceraldehyde-3-phosphate dehydrogenase antisense oligodeoxynucleotides protect against cytosine arabinonucleoside-induced apoptosis in cultured cerebellar neurons. Proc Natl Acad Sci USA 93:9937–9941
Krynetski EY, Krynetskaia NF, Gallo AE, Murti KG, Evans WE (2001) A novel protein complex distinct from mismatch repair binds thioguanylated DNA. Mol Pharmacol 59:367–374
Kubo H (2009) Isolation of madA homologs in Pilobolus crystallinus. Mycoscience 50:400–406
Kubo H (2011) Cloning and expression analysis of putative glyceraldehyde-3-phosphate dehydrogenase genes in Pilobolus crystallinus. Mycoscience 52:99–106
Kubo H, Mihara H (1986) Effects of light and temperature on sporangiophore initiation in Pilobolus crystallinus (Wiggers) Tode. Planta 168:337–339
Kubo H, Mihara H (1996) Effects of microbeam light on growth and phototropism of Pilobolus crystallinus sporangiophore. Mycoscience 37:31–34
McAlister L, Holland MJ (1985) Differential expression of the three yeast glyceraldehyde-3-phosphate dehydrogenase genes. J Biol Chem 260:15019–15027
Page RM (1956) Studies on the development of asexual reproductive structures in Pilobolus. Mycologia 48:206–224
Robbins AR, Ward RD, Oliver C (1995) A mutation in glyceraldehyde-3-phosphate dehydrogenase alters endocytosis in CHO cells. J Cell Biol 130:1093–1104
Sirover MA (2005) New nuclear functions of the glycolytic protein, glyceraldehyde-3-phosphate dehydrogenase, in mammalian cells. J Cell Biochem 95:45–52
Tisdale EJ (2001) Glyceraldehyde-3-phosphate dehydrogenase is required for vesicular transport in the early secretory pathway. J Biol Chem 276:2480–2486
Van Bogaert INA, De Maeseneire SL, Develter D, Soetaert W, Vandamme EJ (2008) Cloning and characterisation of the glyceraldehyde 3-phosphate dehydrogenase gene of Candida bombicola and use of its promoter. J Ind Microbiol Biotechnol 35:1085–1092
Waterham HR, Digan ME, Koutz PJ, Lair SV, Cregg JM (1997) Isolation of the Pichia pastoris glyceraldehyde-3-phosphate dehydrogenase gene and regulation and use of its promoter. Gene 186:37–44
Welch JE, Schatte EC, O’Brien DA, Eddy EM (1992) Expression of a glyceraldehyde 3-phosphate dehydrogenase gene specific to mouse spermatogenic cells. Biol Reprod 46:869–878
Wolff AM, Arnau J (2002) Cloning of glyceraldehyde-3-phosphate dehydrogenase-encoding genes in Mucor circinelloides (syn. racemosus) and use of the GAPDH1 promoter for recombinant protein production. Fungal Genet Biol 35:21–29
Zheng L, Roeder RG, Luo Y (2003) S phase activation of the histone H2B promoter by OCA-S, a coactivator complex that contains GAPDH as a key component. Cell 114:255–266
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kubo, H. Asexual reproductive organ-specific expression of the glyceraldehyde-3-phosphate dehydrogenase 2 gene of Pilobolus crystallinus . Mycoscience 53, 147–151 (2012). https://doi.org/10.1007/s10267-011-0143-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10267-011-0143-2