Abstract
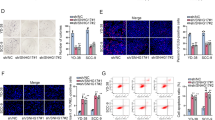
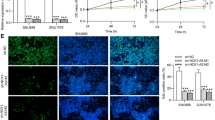
SLC16A1-AS1 is a newly identified lncRNA with different roles in different cancers. MiR-5088-5p is an oncogenic miRNA in breast cancer. However, their participation in oral squamous cell carcinoma (OSCC) is unknown. We predicted the interaction between SLC16A1-AS1 and miR-5088-5p, and this study was carried out to explore the crosstalk between them in OSCC. A total of 56 OSCC patients donated OSCC and paired non-tumor tissues, which were used to detect the differential expression of SLC16A1-AS1 and miR-5088-5p (mature and premature). Analysis of the subcellular location of SLC16A1-AS1 in OSCC cells and its direct interaction with premature miR-5088-5p was performed with cellular fractionation assay and RNA pull-down assay, respectively. The involvement of SLC16A1-AS1 in miR-5088-5p maturation was studied with overexpression assay. BrdU assay was performed to detect cell proliferation after transfection. OSCC tissue samples exhibited decreased expression levels of SLC16A1-AS1 and premature miR-5088-5p, but increased the expression levels of mature miR-5088-5p. SLC16A1-AS1 was detected in both nucleus and cytoplasm samples of OSCC cells and its direct interaction with premature miR-5088-5p was confirmed. Overexpression of SLC16A1-AS1 in OSCC cells resulted in inhibited maturation of miR-5088-5p. SLC16A1-AS1 suppressed the enhancing effects of miR-5088-5p on cell proliferation. SLC16A1-AS1 was downregulated in OSCC and it may inhibit cell proliferation by suppressing maturation of miR-5088-5p.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to their containing information that could compromise the privacy of research participants.
References
Ghantous Y, Abu EI. Global incidence and risk factors of oral cancer. Harefuah. 2017;156(10):645–9.
Ali J, Sabiha B, Jan HU, Haider SA, Khan AA, Ali SS. Genetic etiology of oral cancer. Oral Oncol. 2017;70:23–8. https://doi.org/10.1016/j.oraloncology.2017.05.004.
D’Souza S, Addepalli V. Preventive measures in oral cancer: an overview. Biomed Pharmacother. 2018;107:72–80. https://doi.org/10.1016/j.biopha.2018.07.114.
Awan KH, Hegde R, Cheever VJ, Carroll W, Khan S, Patil S, et al. Oral and pharyngeal cancer risk associated with occupational carcinogenic substances: Systematic review. Head Neck. 2018;40(12):2724–32. https://doi.org/10.1002/hed.25486.
Chen F, Yan L, Lin L, Liu F, Qiu Y, Wang J, et al. Dietary score and the risk of oral cancer: a case–control study in southeast China. Oncotarget. 2017;8(21):34610–6. https://doi.org/10.18632/oncotarget.16659.
Yang Y, Zhou M, Zeng X, Wang C. The burden of oral cancer in China, 1990–2017: an analysis for the global burden of disease, injuries, and risk factors study 2017. BMC Oral Health. 2021;21(1):44. https://doi.org/10.1186/s12903-020-01386-y.
Jiang X, Wu J, Wang J, Huang R. Tobacco and oral squamous cell carcinoma: a review of carcinogenic pathways. Tob Induc Dis. 2019;17:29. https://doi.org/10.18332/tid/105844.
Cheraghlou S, Schettino A, Zogg CK, Judson BL. Changing prognosis of oral cancer: an analysis of survival and treatment between 1973 and 2014. Laryngoscope. 2018;128(12):2762–9. https://doi.org/10.1002/lary.27315.
Yete S, D’Souza W, Saranath D. High-risk human papillomavirus in oral cancer: clinical implications. Oncology. 2018;94(3):133–41. https://doi.org/10.1159/000485322.
Macha MA, Matta A, Kaur J, Chauhan SS, Thakar A, Shukla NK, et al. Prognostic significance of nuclear pSTAT3 in oral cancer. Head Neck. 2011;33(4):482–9. https://doi.org/10.1002/hed.21468.
Wang C, Liu XQ, Hou JS, Wang JN, Huang HZ. Molecular mechanisms of chemoresistance in oral cancer. Chin J Dent Res. 2016;19(1):25–33. https://doi.org/10.3290/j.cjdr.a35694.
Rivera C. Essentials of oral cancer. Int J Clin Exp Pathol. 2015;8(9):11884–94.
Momen-Heravi F, Bala S. Emerging role of non-coding RNA in oral cancer. Cell Signal. 2018;42:134–43. https://doi.org/10.1016/j.cellsig.2017.10.009.
Tian J, Hu D. LncRNA SLC16A1-AS1 is upregulated in hepatocellular carcinoma and predicts poor survival. Clin Res Hepatol Gastroenterol. 2021;45(2): 101490. https://doi.org/10.1016/j.clinre.2020.07.001.
Long Y, Li H, Jin Z, Zhang X. LncRNA SLC16A1-AS1 is upregulated in glioblastoma and promotes cancer cell proliferation by regulating miR-149 methylation. Cancer Manag Res. 2021;13:1215–23. https://doi.org/10.2147/cmar.S264613.
Zhang H, Jin S, Ji A, Ma Y, Zhang C, Wang A, et al. LncRNA SLC16A1-AS1 suppresses cell proliferation in cervical squamous cell carcinoma (CSCC) through the miR-194/SOCS2 axis. Cancer Manag Res. 2021;13:1299–306. https://doi.org/10.2147/cmar.S276629.
Seok HJ, Choi YE, Choi JY, Yi JM, Kim EJ, Choi MY, et al. Novel miR-5088-5p promotes malignancy of breast cancer by inhibiting DBC2. Mol Ther Nucleic Acids. 2021. https://doi.org/10.1016/j.omtn.2021.05.004.
Feng H, Zhang X, Lai W, et al. Long non-coding RNA SLC16A1-AS1: its multiple tumorigenesis features and regulatory role in cell cycle in oral squamous cell carcinoma. Cell Cycle. 2020;9(13):1641–53. https://doi.org/10.1080/15384101.2020.1762048.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
SY: study concepts, literature research, clinical studies, data analysis, experimental studies, manuscript writing and review; TL, DW: study design, literature research, experimental studies and manuscript editing; TL: definition of intellectual content, clinical studies, data acquisition and statistical analysis; DW: data acquisition, manuscript preparation and data analysis. All authors have read and approve the submission of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All other authors have no conflicts of interest. We declare that we do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
Ethical approval and consent to participate
All patients signed the written informed consent. All producers were approved by Daqing Oilfiled General Hospital Ethics Committee. Procedures operated in this research were completed in keeping with the standards set out in the Announcement of Helsinki and laboratory guidelines of research in China.
Consent to publish
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
10266_2022_712_MOESM1_ESM.tif
Supplemental Figure 1 In situ hybridization analysis of the expression of SLC16A1-AS1 and premature miR-5088-5p in paired OSCC samples (TIF 23430 KB)
Rights and permissions
About this article
Cite this article
Li, T., Wang, D. & Yang, S. Analysis of the subcellular location of lncRNA SLC16A1-AS1 and its interaction with premature miR-5088-5p in oral squamous cell carcinoma. Odontology 111, 41–48 (2023). https://doi.org/10.1007/s10266-022-00712-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10266-022-00712-w