Abstract
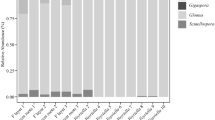
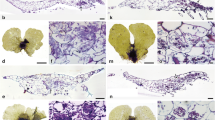
In general, Glomeromycotina was thought to be the earliest fungi forming mycorrhiza-like structure (MLS) in land plant evolution. In contrast, because the earliest divergent lineage of extant land plants, i.e. Haplomitriopsida liverworts, associates only with Mucoromycotina mycobionts, recent studies suggested that those fungi are novel candidates for the earliest mycobionts. Therefore, Mucoromycotina–Haplomitriopsida association currently attracts attention as an ancient mycorrhiza-like association. However, mycobionts were identified in only 7 of 16 Haplomitriopsida species and the mycobionts diversity of this lineage is largely unclarified. To clarify the taxonomic composition of mycobionts in Haplomitriopsida, we observed MLSs in the rhizome of Haplomitrium mnioides (Haplomitriopsida), the Asian representative Haplomitriopsida species, and conducted molecular identification of mycobionts. It was recorded for the first time that Glomeromycotina and Mucoromycotina co-occur in Haplomitriopsida as mycobionts. Significantly, the arbuscule-like branching (ALB) of Glomeromycotina was newly described. As the Mucoromycotina fungi forming MLSs in H. mnioides, Endogonaceae and Densosporaceae were detected, in which size differences of hyphal swelling (HS) were found between the fungal families. This study provides a novel evidence in the MLS of Haplomitriopsida, i.e. the existence of Glomeromycotina association as well as the dominant Mucoromycotina association. In addition, since hyphal characteristics of the HS-type MLS were quite similar to those of fine endophytes (FE) of Endogonales in other bryophytes and vascular plants previously described, this MLS is suggested to be included in FE. These results suggest that Glomeromycotina and Mucoromycotina were acquired concurrently as the mycobionts by the earliest land plants evolved into arbuscular mycorrhizae and FE. Therefore, dual association of Haplomitriopsida, with Endogonales and Glomeromycotina will provide us novel insight on how the earliest land plants adapted to terrestrial habitats with fungi.




Similar content being viewed by others
References
Bakalin V, Vilnet A (2017) How many species are in Apotreubia S. Hatt. & Mizut. (Marchantiophyta)? Nova Hedwig 104:473–482. https://doi.org/10.1127/nova_hedwigia/2016/0386
Bartholomew-Began SE (1991) A morphogenetic re-evaluation of Haplomitrium Nees (Hepatophyta). Bryophyt Bibl 41:1–297
Bidartondo MI, Read DJ, Trappe JM, Merckx V, Ligrone R, Duckett JG (2011) The dawn of symbiosis between plants and fungi. Biol Lett 7:574–577. https://doi.org/10.1098/rsbl.2010.1203
Błaszkowski J (2012) Glomeromycota. W Szafer Institute of Botany, Polish Academy of Sciences, Kraków
Carafa A, Duckett JG, Ligrone R (2003) Subterranean gametophytic axes in the primitive liverwort Haplomitrium harbour a unique type of endophytic association with aseptate fungi. New Phytol 160:185–197. https://doi.org/10.1046/j.1469-8137.2003.00849.x
Chang Y, Desirò A, Na H, Sandor L, Lipzen A, Clum A, Barry K, Grigoriev IV, Martin FM, Stajich JE, Smith ME, Bonito G, Spatafora JW (2019) Phylogenomics of Endogonaceae and evolution of mycorrhizas within Mucoromycota. New Phytol 222:511–525. https://doi.org/10.1111/nph.15613
Desirò A, Duckett JG, Pressel S, Villarreal JC, Bidartondo MI (2013) Fungal symbioses in hornworts: a chequered history. Biol Sci, Proc R Soc B. https://doi.org/10.1098/rspb.2013.0207
Desirò A, Rimington WR, Jacob A, Pol NV, Smith ME, Trappe JM, Bidartondo MI, Bonito G (2017) Multigene phylogeny of Endogonales, an early diverging lineage of fungi associated with plants. IMA Fungus 8:245–257. https://doi.org/10.5598/imafungus.2017.08.02.03
Duckett JG, Carafa A, Ligrone R (2006) A highly differentiated glomeromycotean association with the mucilage-secreting, primitive antipodean liverwort Treubia (Treubiaceae): clues to the origins of mycorrhizas. Am J Bot 93:797–813. https://doi.org/10.3732/ajb.93.6.797
Edgar RC (2016) UCHIME2: improved chimera prediction for amplicon sequencing. bioRxiv. https://doi.org/10.1101/074252
Field KJ, Pressel S (2018) Unity in diversity: structural and functional insights into the ancient partnerships between plants and fungi. New Phytol 220:996–1011. https://doi.org/10.1111/nph.15158
Field KJ, Cameron DD, Leake JR, Tille S, Bidartondo MI, Beerling DJ (2012) Contrasting arbuscular mycorrhizal responses of vascular and non-vascular plants to a simulated Palaeozoic CO2 decline. Nat Commun 3:835. https://doi.org/10.1038/ncomms1831
Field KJ, Pressel S, Duckett JG, Rimington WR, Bidartondo MI (2015a) Symbiotic options for the conquest of land. Trends Ecol Evol 30:477–486. https://doi.org/10.1016/j.tree.2015.05.007
Field KJ, Rimington WR, Bidartondo MI, Allinson KE, Beerling DJ, Cameron DD, Duckett JG, Leake JR, Pressel S (2015b) First evidence of mutualism between ancient plant lineages (Haplomitriopsida liverworts) and Mucoromycotina fungi and its response to simulated Palaeozoic changes in atmospheric CO2. New Phytol 205:743–756. https://doi.org/10.1111/nph.13024
Field KJ, Rimington WR, Bidartondo MI, Allinson KE, Beerling DJ, Cameron DD, Duckett JG, Leake JR, Pressel S (2015c) Functional analysis of liverworts in dual symbiosis with Glomeromycota and Mucoromycotina fungi under a simulated Palaeozoic CO2 decline. ISME J 10:1514–1526. https://doi.org/10.1038/ismej.2015.204
Field KJ, Bidartondo MI, Rimington WR, Hoysted GA, Beerling D, Cameron DD, Duckett JG, Leake JR, Pressel S (2019) Functional complementarity of ancient plant-fungal mutualisms: contrasting nitrogen, phosphorus and carbon exchanges between Mucoromycotina and Glomeromycotina fungal symbionts of liverworts. New Phytol 223:908–921. https://doi.org/10.1111/nph.15819
Gianinazzi-Pearson V, Morandi D, Dexheimer J, Gianinazzi S (1981) Ultrastructural and ultracytochemical features of a Glomus tenuis mycorrhiza. New Phytol 88:633–639. https://doi.org/10.1111/j.1469-8137.1981.tb01739.x
Goebel K (1891) Morphologische und biologische studien. IV. Über javanische Lebermoose. 1 Treubia. Ann Jard Bot Buitenzorg 9:1–11
Grubb PJ (1970) Observations on the structure and biology of Haplomitrium and Takakia, hepatics with roots. New Phytol 69:303–326. https://doi.org/10.1111/j.1469-8137.1970.tb02430.x
Gucwa-Przepióra E, Błaszkowski J, Kurtyka R, Małkowski Ł, Małkowski E (2013) Arbuscular mycorrhiza of Deschampsia cespitosa (Poaceae) at different soil depths in highly metal-contaminated site in southern Poland. Acta Soc Bot Pol 82:251–258. https://doi.org/10.5586/asbp.2013.033
Hattori S, Mizutani M (1958) What is Takakia lepidozioides? J Hattori Bot Lab 38:115–121
Hirose D, Degawa Y, Yamamoto K, Yamada A (2014) Sphaerocreas pubescens is a member of the Mucoromycotina closely related to fungi associated with liverworts and hornworts. Mycoscience 55:221–226. https://doi.org/10.1016/j.myc.2013.09.002
Humphreys CP, Franks PJ, Rees M, Bidartondo MI, Leake JR, Beerling DJ (2010) Mutualistic mycorrhiza-like symbiosis in the most ancient group of land plants. Nat Commun 1:103. https://doi.org/10.1038/ncomms1105
Inoue H (1960) Studies in Treubia nana (Hepaticae) with special reference to the antheridial development. Bot Mag Tokyo 73:225–230. https://doi.org/10.15281/jplantres1887.73.225
Izumitsu K, Hatoh K, Sumita T, Kitade Y, Morita A, Gafur A, Ohta A, Kawai M, Yamanaka T, Neda H, Ota Y, Tanaka C (2012) Rapid and simple preparation of mushroom DNA directly from colonies and fruiting bodies for PCR. Mycoscience 53:396–401. https://doi.org/10.1007/S10267-012-0182-3
Koske RE, Gemma JN (1989) A modified procedure for staining roots to detect VA mycorrhizas. Mycol Res 92:486–488. https://doi.org/10.1016/S0953-7562(89)80195-9
Koske RE, Tessier B (1983) A convenient, permanent slide mounting medium. Mycol Soc Am Newslett 34:59
Lee J, Lee S, Young JP (2008) Improved PCR primers for the detection and identification of arbuscular mycorrhizal fungi. FEMS Microbiol Ecol 65:339–349. https://doi.org/10.1111/j.1574-6941.2008.00531.x
Ligrone R, Carafa A, Lumini E, Bianciotto V, Bonfante P, Duckett JG (2007) Glomeromycotean associations in liverworts: a molecular, cellular, and taxonomic analysis. Am J Bot 94:1756–1777. https://doi.org/10.3732/ajb.94.11.1756
Lilienfeld F (1911) Beiträge zur kenntnis der art Haplomitrium hookeri Nees. Bull Acad Sci Crac Sér B 1911:315–339
Morton JB, Redecker D (2001) Two new families of Glomales, Archaeosporaceae and Paraglomaceae, with two new genera Archaeospora and Paraglomus, based on concordant molecular and morphological characters. Mycologia 93:181–195. https://doi.org/10.2307/3761615
Oguchi S, Sato K (2003) Springing out mechanisms to groundwater at the upper reaches of rivers of Mt. Higashiyama, Hachijo-jima Island. Proc Inst Nat Sci Nihon Univ 38:233–242
Öpik M, Vanatoa A, Vanatoa E, Moora M, Davison J, Kalwij JM, Reier U, Zobel M (2010) The online database MaarjAM reveals global and ecosystemic distribution patterns in arbuscular mycorrhizal fungi (Glomeromycota). New Phytol 188:223–241. https://doi.org/10.1111/j.1469-8137.2010.03334.x
Orchard S, Standish RJ, Nicol D, Gupta VVSR, Ryan MH (2016) The response of fine root endophyte (Glomus tenue) to waterlogging is dependent on host plant species and soil type. Plant Soil 403:305–315. https://doi.org/10.1007/s11104-016-2804-6
Orchard S, Hilton S, Bending GD, Dickie IA, Standish RJ, Gleeson DB, Jeffery RP, Powell JR, Walker C, Bass D, Monk J, Simonin A, Ryan MH (2017a) Fine endophytes (Glomus tenue) are related to Mucoromycotina, not Glomeromycota. New Phytol 213:481–486. https://doi.org/10.1111/nph.14268
Orchard S, Standish RJ, Dickie IA, Renton M, Walker C, Moot D, Ryan MH (2017b) Fine root endophytes under scrutiny: a review of the literature on arbuscule-producing fungi recently suggested to belong to the Mucoromycotina. Mycorrhiza 27:619–638. https://doi.org/10.1007/s00572-017-0782-z
Postma JWM, Olsson PA, Falkengren-Grerup U (2007) Root colonisation by arbuscular mycorrhizal, fine endophytic and dark septate fungi across a pH gradient in acid beech forests. Soil Biol Biochem 39:400–408. https://doi.org/10.1016/j.soilbio.2006.08.007
Redecker D, Kodner R, Graham LE (2000) Glomalean fungi from the Ordovician. Science 289:1920–1921. https://doi.org/10.1126/science.289.5486.1920
Rimington WR, Pressel S, Duckett JG, Field KJ, Read DJ, Bidartondo MI (2018) Ancient plants with ancient fungi: liverworts associate with early-diverging arbuscular mycorrhizal fungi. Biol Sci, Proc R Soc B. https://doi.org/10.1098/rspb.2018.1600
Ronquist F, Teslenko M, van der Mark P, Ayres DL, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. https://doi.org/10.1093/sysbio/sys029
Shi XQ, Zhu RL (2006) A range extension for Haplomitrium mnioides (Lindb.) R.M.Schust. Trop Bryol 27:87–90. https://doi.org/10.11646/bde.27.1.11
Silvestro D, Michalak I (2012) raxmlGUI: a graphical front-end for RAxML. Org Divers Evol 12:335–337. https://doi.org/10.1007/s13127-011-0056-0
Simon L, Bousquet J, Lévesque RC, Lalonde M (1993) Origin and diversification of endomycorrhizal fungi and coincidence with vascular land plants. Nature 363:67–69. https://doi.org/10.1038/363067a0
Smit E, Leeflang P, Glandorf B, Van Elsas JD, Wernars K (1999) Analysis of fungal diversity in the wheat rhizosphere by sequencing of cloned PCR-amplified genes encoding 18S rRNA and temperature gradient gel electrophoresis. Appl Environ Microbiol 65:2614–2621
Smith SE, Read DJ (2008) Mycorrhizal symbiosis, 3rd edn. Academic Press, Cambridge
Söderström L, Hagborg A, von Konrat M, Bartholomew-Began S, Bell D, Briscoe L, Brown E, Cargill DC, Costa DP, Crandall-Stotler BJ, Cooper ED, Dauphin G, Engel JJ, Feldberg K, Glenny D, Gradstein SR, He X, Heinrichs J, Hentschel J, Ilkiu-Borges AL, Katagiri T, Konstantinova NA, Larraín J, Long DG, Nebel M, Pócs T, Puche F, Reiner-Drehwald E, Renner MA, Sass-Gyarmati A, Schäfer-Verwimp A, Moragues JG, Stotler RE, Sukkharak P, Thiers BM, Uribe J, Váňa J, Villarreal JC, Wigginton M, Zhang L, Zhu RL (2016) World checklist of hornworts and liverworts. PhytoKeys 27:1–828. https://doi.org/10.3897/phytokeys.59.6261
Stahl M (1949) Die Mycorrhiza der Lebermoose mit besonderer Berucksichtigung der thallosen formen. Planta 37:103–148. https://doi.org/10.1007/BF01929705
Stech M, Frey W (2004) Molecular circumscription and relationships of selected Gondwanan species of Haplomitrium (Calobryales, Haplomitriopsida, Hepaticophytina). Studies in austral temperate rain forest bryophytes 24. Nova Hedwig 78:57–70. https://doi.org/10.1127/0029-5035/2004/0078-0057
Stevens KJ, Wall CB, Janssen JA (2011) Effects of arbuscular mycorrhizal fungi on seedling growth and development of two wetland plants, Bidens frondosa L., and Eclipta prostrata (L.) L., grown under three levels of water availability. Mycorrhiza 21:279–288. https://doi.org/10.1007/s00572-010-0334-2
Strullu-Derrien C, Kenrick P, Pressel S, Duckett JG, Rioult J-P, Strullu D-G (2014) Fungal associations in Horneophyton ligneri from the Rhynie Chert (c. 407 million year old) closely resemble those in extant lower land plants: novel insights into ancestral plant–fungus symbioses. New Phytol 203:964–979. https://doi.org/10.1111/nph.12805
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729. https://doi.org/10.1093/molbev/mst197
Turnau K, Ronikier M, Unrug J (1999) Role of mycorrhizal links between plants in establishment of liverworts thalli in natural habitat. Acta Soc Bot Pol 68:63–68. https://doi.org/10.5586/asbp.1999.011
Walker C, Gollotte A, Redecker D (2018) A new genus, Planticonsortium (Mucoromycotina), and new combination (P. tenue), for the fine root endophyte, Glomus tenue (basionym Rhizophagus tenuis). Mycorrhiza 28:213–219. https://doi.org/10.1007/s00572-017-0815-7
Wang GM, Stribley DP, Tinker PB (1985) Soil pH and vesicular-arbuscular mycorrhizas. In: Fitter AH (ed) Ecological interactions in soil: plants, microbes and animals. Blackwell Scientific, Oxford, pp 219–224
Wang B, Yeun LH, Zue J-Y, Liu Y, Ané J-M, Qiu Y-L (2010) Presence of three mycorrhizal genes in the common ancestor of land plants suggests a key role of mycorrhizas in the colonization of land by plants. New Phytol 186:514–525. https://doi.org/10.1111/j.1469-8137.2009.03137.x
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, New York, pp 315–322
Wright ES, Yilmaz LS, Noguera DR (2012) DECIPHER, a search-based approach to chimera identification for 16S rRNA Sequences. Appl Environ Microbiol 78:717–725. https://doi.org/10.1128/AEM.06516-11
Yamamoto K, Degawa Y, Hirose D, Fukuda M, Yamada A (2015) Morphology and phylogeny of four Endogone species and Sphaerocreas pubescens collected in Japan. Mycol Prog 14:86. https://doi.org/10.1007/s11557-015-1111-6
Yamamoto K, Degawa Y, Takashima Y, Fukuda M, Yamada A (2017a) Endogone corticioides sp. nov. from subalpine conifer forests in Japan and China, and its multi-locus phylogeny. Mycoscience 58:23–29. https://doi.org/10.1016/j.myc.2016.08.001
Yamamoto K, Endo N, Degawa Y, Fukuda M, Yamada A (2017b) First detection of Endogone ectomycorrhizas in natural oak forests. Mycorrhiza 27:295–301. https://doi.org/10.1007/s00572-016-0740-1
Acknowledgements
We are grateful to Prof. Mitsuru Moriguchi, Dr. Takamichi Orihara, Ms. Yumiko Oba, Mr. Yoshiteru Kawabata, and Dr. Kanami Yoshino for supporting for our sampling. We are also grateful to Prof. Tomio Yamaguchi for depositing of H. mnioides specimens. We thank the technical staff in the Division of Instrumental Analysis, Research Center for Human and Environmental Sciences, Shinshu University for DNA sequencing. This study was supported in part by KAKENHI (No. JP14J09199, No. JP25291084 and JP15H01751) from the Japan Society for the Promotion of Science (JSPS).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yamamoto, K., Shimamura, M., Degawa, Y. et al. Dual colonization of Mucoromycotina and Glomeromycotina fungi in the basal liverwort, Haplomitrium mnioides (Haplomitriopsida). J Plant Res 132, 777–788 (2019). https://doi.org/10.1007/s10265-019-01145-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-019-01145-3