Abstract
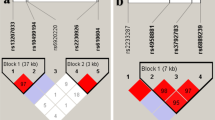
Objective To investigate the role of eight locus polymorphisms of tumor necrosis factor receptor-associated factor 2 (TRAF2) and TRAF5 gene and their interaction in the susceptibility to ankylosing spondylitis (AS) in Chinese Han population. Methods Eight single nucleotide polymorphisms (SNPs) of TRAF2 (rs3750511, rs10781522, rs17250673, rs59471504) and TRAF5 (rs6540679, rs12569232, rs4951523, rs7514863) gene were genotyped in 673 AS patients and 687 controls. Results The SNPs of TRAF2 and TRAF5 do not indicate a correlation with the susceptibility of AS in Chinese Han population. Genotype frequencies of rs3750511 were statistically significant in females between patients and controls. The allele frequencies of rs10781522 and genotype frequencies of rs3750511 were statistically significant between groups of different diseases activity. One three-locus model, TRAF2 (rs10781522, rs17250673) and TRAF5 (rs12569232), had a maximum testing accuracy of 52.67% and a maximum cross-validation consistency (10/10) that was significant at the level of P = 0.0001, after determined empirically by permutation testing. As to environmental variables, only marginal association was found between sleep quality and AS susceptibility. Conclusion TRAF2 rs3750511 polymorphism may be associated with the susceptibility and severity of AS. Besides, the interaction of TRAF2 and TRAF5 genes may be associated with AS susceptibility, but many open questions remain.

Similar content being viewed by others
References
Baeten D, Baraliakos X, Braun J, et al. Anti-interleukin-17A monoclonal antibody secukinumab in treatment of ankylosing spondylitis: a randomised, double-blind, placebo-controlled trial. Lancet. 2013;382:1705–13. https://doi.org/10.1016/s0140-6736(13)61134-4.
Ng SC, Liao Z, Yu DT, Chan ES, Zhao L, Gu J. Epidemiology of spondyloarthritis in the People’s Republic of China: review of the literature and commentary. Semin Arthritis Rheum. 2007;37:39–47. https://doi.org/10.1016/j.semarthrit.2007.01.003.
Zhai J, Rong J, Li Q, Gu J. Immunogenetic study in Chinese population with ankylosing spondylitis: are there specific genes recently disclosed? Clin Dev Immunol. 2013. https://doi.org/10.1155/2013/419357.
Boonen A. A review of work-participation, cost-of-illness and cost-effectiveness studies in ankylosing spondylitis. Nat Clin Pract Rheumatol. 2006;2:546–53. https://doi.org/10.1038/ncprheum0297.
Miao J, Chen Y, Zhang B, et al. Surgical treatment for odontoid fractures in patients with long-standing ankylosing spondylitis: a report of 3 cases and review of the literature. World Neurosurg. 2018;116:88–93. https://doi.org/10.1016/j.wneu.2018.05.055.
Coksevim NH, Durmus D, Kuru O. Effects of global postural reeducation exercise and anti-TNF treatments on disease activity, function, fatigue, mobility, sleep quality and depression in patients with active Ankylosing spondylitis: a prospective follow-up study. J Back Musculoskelet Rehabil. 2018;31:1005–12. https://doi.org/10.3233/bmr-170901.
Skuhala T, Atelj A, Prepolec J, Al-Mufleh M, Stanimirović A, Vukelić D. A case report of severe recurrent varicella in an ankylosing spondylitis patient treated with adalimumab - a new side effect after 15 years of usage. BMC Infect Dis. 2019;19:127. https://doi.org/10.1186/s12879-019-3768-y.
Khan MA, Mathieu A, Sorrentino R, Akkoc N. The pathogenetic role of HLA-B27 and its subtypes. Autoimmun Rev. 2007;6:183–9. https://doi.org/10.1016/j.autrev.2006.11.003.
Brewerton DA, Hart FD, Nicholls A, Caffrey M, James DC, Sturrock RD. Ankylosing spondylitis and HL-A 27. Lancet. 1973;1:904–7. https://doi.org/10.1016/s0140-6736(73)91360-3.
Khan MA. HLA-B27 and its subtypes in world populations. Curr Opin Rheumatol. 1995;7:263–9. https://doi.org/10.1097/00002281-199507000-00001.
Hanson A, Brown MA. Genetics and the causes of ankylosing spondylitis. Rheum Dis Clin North Am. 2017;43:401–14. https://doi.org/10.1016/j.rdc.2017.04.006.
Uygunoğlu U, Uluduz D, Taşçılar K, Saip S. Multiple sclerosis during adalimumab treatment in a case with ankylosing spondylitis. Rheumatol Int. 2014;34:141–3. https://doi.org/10.1007/s00296-012-2625-8.
Wang NG, Wang DC, Tan BY, Wang F, Yuan ZN. TNF-α and IL10 polymorphisms interaction increases the risk of ankylosing spondylitis in Chinese Han population. Int J Clin Exp Pathol. 2015;8:15204–9.
Hayden MS, Ghosh S. Shared principles in NF-kappaB signaling. Cell. 2008;132:344–62. https://doi.org/10.1016/j.cell.2008.01.020.
Grech AP, Amesbury M, Chan T, Gardam S, Basten A, Brink R. TRAF2 differentially regulates the canonical and noncanonical pathways of NF-kappaB activation in mature B cells. Immunity. 2004;21:629–42. https://doi.org/10.1016/j.immuni.2004.09.011.
Ishida TK, Tojo T, Aoki T, et al. TRAF5, a novel tumor necrosis factor receptor-associated factor family protein, mediates CD40 signaling. Proc Natl Acad Sci U S A. 1996;93:9437–42. https://doi.org/10.1073/pnas.93.18.9437.
Swaidani S, Liu C, Zhao J, Bulek K, Li X. TRAF regulation of IL-17 cytokine signaling. Front Immunol. 2019;10:1293. https://doi.org/10.3389/fimmu.2019.01293.
Zhong B, Liu X, Wang X, et al. Negative regulation of IL-17-mediated signaling and inflammation by the ubiquitin-specific protease USP25. Nat Immunol. 2012;13:1110–7. https://doi.org/10.1038/ni.2427.
Amatya N, Childs EE, Cruz JA, et al. IL-17 integrates multiple self-reinforcing, feed-forward mechanisms through the RNA binding protein Arid5a. Sci Signal. 2018. https://doi.org/10.1126/scisignal.aat4617.
Xiang Q, Chen L, Fang J, et al. TNF receptor-associated factor 5 gene confers genetic predisposition to acute anterior uveitis and pediatric uveitis. Arthritis Res Ther. 2013;15:R113. https://doi.org/10.1186/ar4293.
Potter C, Eyre S, Cope A, Worthington J, Barton A. Investigation of association between the TRAF family genes and RA susceptibility. Ann Rheum Dis. 2007;66:1322–6. https://doi.org/10.1136/ard.2006.065706.
Wang X, Yang J, Han L, et al. TRAF5-mediated Lys-63-linked polyubiquitination plays an essential role in positive regulation of RORgammat in promoting IL-17A expression. J Biol Chem. 2015;290:29086–94. https://doi.org/10.1074/jbc.M115.664573.
Liu J, Qi Y, Zheng L, et al. Xinfeng capsule improves pulmonary function in ankylosing spondylitis patients via NF-KappaB-iNOS-NO signaling pathway. J Tradit Chin Med. 2014;34:657–65.
Hong X, Hao K, Ladd-Acosta C, et al. Genome-wide association study identifies peanut allergy-specific loci and evidence of epigenetic mediation in US children. Nat Commun. 2015;6:6304. https://doi.org/10.1038/ncomms7304.
Edmunds L, Elswood J, Calin A. New light on uveitis in ankylosing spondylitis. J Rheumatol. 1991;18:50–2.
Linssen A, Rothova A, Valkenburg HA, et al. The lifetime cumulative incidence of acute anterior uveitis in a normal population and its relation to ankylosing spondylitis and histocompatibility antigen HLA-B27. Invest Ophthalmol Vis Sci. 1991;32:2568–78.
Zeboulon N, Dougados M, Gossec L. Prevalence and characteristics of uveitis in the spondyloarthropathies: a systematic literature review. Ann Rheum Dis. 2008;67:955–9. https://doi.org/10.1136/ard.2007.075754.
Ritchie MD, Hahn LW, Roodi N, et al. Multifactor-dimensionality reduction reveals high-order interactions among estrogen-metabolism genes in sporadic breast cancer. Am J Hum Genet. 2001;69:138–47. https://doi.org/10.1086/321276.
Dervieux T, Wessels JA, Kremer JM, et al. Patterns of interaction between genetic and nongenetic attributes and methotrexate efficacy in rheumatoid arthritis. Pharmacogenet Genomics. 2012;22:1–9. https://doi.org/10.1097/FPC.0b013e32834d3e0b.
van den Berg R, de Hooge M, van Gaalen F, Reijnierse M, Huizinga T, van der Heijde D. Percentage of patients with spondyloarthritis in patients referred because of chronic back pain and performance of classification criteria: experience from the Spondyloarthritis Caught Early (SPACE) cohort. Rheumatology (Oxford). 2013;52:1492–9. https://doi.org/10.1093/rheumatology/ket164.
Calonga-Solís V, Amorim LM, Farias TDJ, Petzl-Erler ML, Malheiros D, Augusto DG. Variation in genes implicated in B-cell development and antibody production affects susceptibility to pemphigus. Immunology. 2021;162:58–67. https://doi.org/10.1111/imm.13259.
Bumiller-Bini V, Cipolla GA, Spadoni MB, et al. Condemned or not to die? gene polymorphisms associated with cell death in pemphigus foliaceus. Front Immunol. 2019;10:2416. https://doi.org/10.3389/fimmu.2019.02416.
Westra HJ, Peters MJ, Esko T, et al. Systematic identification of trans eQTLs as putative drivers of known disease associations. Nat Genet. 2013;45:1238–43. https://doi.org/10.1038/ng.2756.
Habelhah H, Takahashi S, Cho SG, Kadoya T, Watanabe T, Ronai Z. Ubiquitination and translocation of TRAF2 is required for activation of JNK but not of p38 or NF-kappaB. EMBO J. 2004;23:322–32. https://doi.org/10.1038/sj.emboj.7600044.
Zhang L, Blackwell K, Thomas GS, Sun S, Yeh WC, Habelhah H. TRAF2 suppresses basal IKK activity in resting cells and TNFalpha can activate IKK in TRAF2 and TRAF5 double knockout cells. J Mol Biol. 2009;389:495–510. https://doi.org/10.1016/j.jmb.2009.04.054.
Thomas GS, Zhang L, Blackwell K, Habelhah H. Phosphorylation of TRAF2 within its RING domain inhibits stress-induced cell death by promoting IKK and suppressing JNK activation. Cancer Res. 2009;69:3665–72. https://doi.org/10.1158/0008-5472.can-08-4867.
Lin Z, Su Y, Zhang C, et al. The interaction of BDNF and NTRK2 gene increases the susceptibility of paranoid schizophrenia. PLoS ONE. 2013. https://doi.org/10.1371/journal.pone.0074264.
Yeh WC, Shahinian A, Speiser D, et al. Early lethality, functional NF-kappaB activation, and increased sensitivity to TNF-induced cell death in TRAF2-deficient mice. Immunity. 1997;7:715–25. https://doi.org/10.1016/s1074-7613(00)80391-x.
Nakano H, Sakon S, Koseki H, et al. Targeted disruption of Traf5 gene causes defects in CD40- and CD27-mediated lymphocyte activation. Proc Natl Acad Sci U S A. 1999;96:9803–8. https://doi.org/10.1073/pnas.96.17.9803.
Tada K, Okazaki T, Sakon S, et al. Critical roles of TRAF2 and TRAF5 in tumor necrosis factor-induced NF-kappa B activation and protection from cell death. J Biol Chem. 2001;276:36530–4. https://doi.org/10.1074/jbc.M104837200.
Nagashima H, Okuyama Y, Hayashi T, Ishii N, So T. TNFR-associated factors 2 and 5 differentially regulate the instructive IL-6 receptor signaling required for Th17 development. J Immunol. 2016;196:4082–9. https://doi.org/10.4049/jimmunol.1501610.
Acknowledgements
We are deeply grateful to all the patients and healthy controls who participated in our study. And we would like to acknowledgement the national natural science foundation of China for its financial support.
Funding
The study was sponsored by the National Natural Science Foundation of China (82073655 and 81773514) and the Scientific Research Level upgrading Project of Anhui Medical University (2020xkjT006).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, S., Kong, J., Huang, L. et al. Single nucleotide polymorphisms of TRAF2 and TRAF5 gene in ankylosing spondylitis: a case–control study. Clin Exp Med 21, 645–653 (2021). https://doi.org/10.1007/s10238-021-00719-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-021-00719-7