Abstract
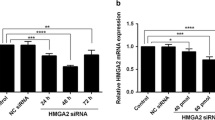
B7-H4 is over-expressed in various tumors and may affect many aspects of cancer biology. Our previous studies have reported that the over-expressed B7-H4 in serum or tumor tissue of colorectal carcinoma (CRC) patients was closely related to CRC progression. However, B7-H4 in cell biological characteristics of CRC is not well studied. Here, we investigate the effect of the B7-H4 on cell proliferation, migration and its expression regulated by PI3K/Akt/mTOR signaling pathway in CRC. Firstly, pSilencer 4.1-B7-H4-shRNA vector was constructed and stable transfection was performed on HT-29 cells. Secondly, cell proliferation, cell cycle, cell apoptosis and cell migration were evaluated after B7-H4 silencing, and the expression of Bcl-2, caspase-3, MMP-2 and MMP-9 was also measured. Finally, the regulation of B7-H4 by PI3K/Akt/mTOR signaling pathway was measured followed by treatment with or without PI3K/Akt and mTOR inhibitor. The results showed that the viability of HT-29 cells was significantly decreased after B7-H4 silencing (P < 0.05). B7-H4 silencing significantly increased the apoptosis rate and caspase-3 protein expression while decreased Bcl-2 protein expression (P all < 0.05). B7-H4 silencing also significantly reduced the migration of HT-29 cells (P < 0.01) and the secretion of MMP-2 or MMP-9 (P all < 0.05). Following treatment with PI3K/Akt and mTOR inhibitor in HT-29 cells, the expression of B7-H4 was significantly downregulated compared with untreated group (P all < 0.05). Our results strongly suggest that B7-H4 may be involved in cell proliferation and migration by PI3K/Akt/mTOR signaling pathway. Therefore, blocking B7-H4 signaling might be a novel treatment strategy for CRC.




Similar content being viewed by others
References
Vanova B, Kalman M, Jasek K, et al. Droplet digital PCR revealed high concordance between primary tumors and lymph node metastases in multiplex screening of KRAS mutations in colorectal cancer. Clin Exp Med. 2019;19(2):219–24.
Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58(2):71–96.
Zhu J, Tan Z, Hollis-Hansen K, et al. Epidemiological trends in colorectal cancer in China: an ecological study. Dig Dis Sci. 2017;62(1):235–43.
Ismaili N. Treatment of colorectal liver metastases. World J Surg Oncol. 2011;9:154.
Cho YB, Lee WY, Choi SJ, et al. CC chemokine ligand 7 expression in liver metastasis of colorectal cancer. Oncol Rep. 2012;28(2):689–94.
Zhang L, Wu H, Lu D, et al. The costimulatory molecule B7-H4 promote tumor progression and cell proliferation through translocating into nucleus. Oncogene. 2013;32(46):5347–58.
Choi IH, Zhu G, Sica GL, et al. Genomic organization and expression analysis of B7-H4, an immune inhibitory molecule of the B7 family. J Immunol. 2003;171(9):4650–4.
Liang L, Jiang Y, Chen JS, et al. B7-H4 expression in ovarian serous carcinoma: a study of 306 cases. Hum Pathol. 2016;57:1–6.
Liu CL, Zang XX, Huang H, et al. The expression of B7-H3 and B7-H4 in human gallbladder carcinoma and their clinical implications. Eur Rev Med Pharmacol Sci. 2016;20(21):4466–73.
Krambeck AE, Thompson RH, Dong H, et al. B7-H4 expression in renal cell carcinoma and tumor vasculature: associations with cancer progression and survival. Proc Natl Acad Sci U S A. 2006;103(27):10391–6.
Hong B, Qian Y, Zhang H, et al. Expression of B7-H4 and hepatitis B virus X in hepatitis B virus-related hepatocellular carcinoma. World J Gastroenterol. 2016;22(18):4538–46.
Jiang J, Zhu Y, Wu C, et al. Tumor expression of B7-H4 predicts poor survival of patients suffering from gastric cancer. Cancer Immunol Immunother. 2010;59(11):1707–14.
Liu WH, Chen YY, Zhu SX, et al. B7-H4 expression in bladder urothelial carcinoma and immune escape mechanisms. Oncol Lett. 2014;8(6):2527–34.
Zhu J, Chu BF, Yang YP, et al. B7-H4 expression is associated with cancer progression and predicts patient survival in human thyroid cancer. Asian Pac J Cancer Prev. 2013;14(5):3011–5.
Zhao LW, Li C, Zhang RL, et al. B7-H1 and B7-H4 expression in colorectal carcinoma: correlation with tumor FOXP3(+) regulatory T-cell infiltration. Acta Histochem. 2014;116(7):1163–8.
Wang P, Li C, Zhang F, et al. Clinical value of combined determination of serum B7-H4 with carcinoembryonic antigen, osteopontin, or tissue polypeptide-specific antigen for the diagnosis of colorectal cancer. Dis Markers. 2018: 4310790.
Ichikawa M, Chen L. Role of B7-H1 and B7-H4 molecules in down-regulating effector phase of T-cell immunity: novel cancer escaping mechanisms. Front Biosci. 2005;10:2856–60.
Qian Y, Shen L, Cheng L, et al. B7-H4 expression in various tumors determined using a novel developed monoclonal antibody. Clin Exp Med. 2011;11(3):163–70.
Wang W, Xu C, Wang Y, et al. Prognostic values of B7-H4 in non-small cell lung cancer. Biomarkers. 2016; 1–16
Simon I, Liu Y, Krall KL, et al. Evaluation of the novel serum markers B7-H4, Spondin 2, and DcR3 for diagnosis and early detection of ovarian cancer. Gynecol Oncol. 2007;106(1):112–8.
Zhang C, Li Y, Wang Y. Diagnostic value of serum B7-H4 for hepatocellular carcinoma. J Surg Res. 2015;197(2):301–6.
Czauderna F, Fechtner M, Aygün H, et al. Functional studies of the PI(3)-kinase signalling pathway employing synthetic and expressed siRNA. Nucleic Acids Res. 2003;31(2):670–82.
Rebelo TM, Vania L, Ferreira E, et al. siRNA-mediated LRP/LR knock-down reduces cellular viability of malignant melanoma cells through the activation of apoptotic caspases. Exp Cell Res. 2018;368(1):1–12.
Danial NN, Korsmeyer SJ. Cell death: critical control points. Cell. 2004;116(2):205–19.
Song Z, Fan TJ. Tetracaine induces apoptosis through a mitochondrion-dependent pathway in human corneal stromal cells in vitro. Cutan Ocul Toxicol. 2018;37(4):350–8.
Clark AG, Vignjevic DM. Modes of cancer cell invasion and the role of the microenvironment. Curr Opin Cell Biol. 2015;36:13–22.
Massagué J, Obenauf AC. Metastatic colonization by circulating tumour cells. Nature. 2016;529(7586):298–306.
Gialeli C, Theocharis AD, Karamanos NK. Roles of matrix metalloproteinases in cancer progression and their pharmacological targeting. FEBS J. 2011;278(1):16–27.
Araújo RF Jr, Lira GA, Vilaça JA, et al. Prognostic and diagnostic implications of MMP-2, MMP-9, and VEGF-α expressions in colorectal cancer. Pathol Res Pract. 2015;211(1):71–7.
Ranogajec I, Jakić-Razumović J, Puzović V, et al. Prognostic value of matrix metalloproteinase-2 (MMP-2), matrix metalloproteinase-9 (MMP-9) and aminopeptidase N/CD13 in breast cancer patients. Med Oncol. 2012;29(2):561–9.
Waldmann TA, Chen J. Disorders of the JAK/STAT pathway in T cell lymphoma pathogenesis: implications for immunotherapy. Annu Rev Immunol. 2017;35:533–50.
Sokolowski KM, Koprowski S, Kunnimalaiyaan S, et al. Potential molecular targeted therapeutics: role of PI3-K/Akt/mTOR inhibition in cancer. Anticancer Agents Med Chem. 2016;16(1):29–37.
Du L, Li X, Zhen L, et al. Everolimus inhibits breast cancer cell growth through PI3K/AKT/mTOR signaling pathway. Mol Med Rep. 2018;17(5):7163–9.
Yan G, Ru Y, Wu K, et al. GOLM1 promotes prostate cancer progression through activating PI3K-AKT-mTOR signaling. Prostate. 2018;78(3):166–77.
Yang J, Pi C, Wang G. Inhibition of PI3K/Akt/mTOR pathway by apigenin induces apoptosis and autophagy in hepatocellular carcinoma cells. Biomed Pharmacother. 2018;103:699–707.
Pitts TM, Newton TP, Bradshaw-Pierce EL, et al. Dual pharmacological targeting of the MAP kinase and PI3K/mTOR pathway in preclinical models of colorectal cancer. PLoS One. 2014;9(11):e113037.
Francipane MG, Lagasse E. mTOR pathway in colorectal cancer: an update. Oncotarget. 2014;5(1):49–66.
Johnson SM, Gulhati P, Rampy BA, et al. Novel expression patterns of PI3K/Akt/mTOR signaling pathway components in colorectal cancer. J Am Coll Surg. 2010;210(5):767–78.
Acknowledgments
This research was supported by the Science and Technology Department of Jilin province (20170414030GH; 20190201220JC) and the Education Department of Jilin Province (JJKH20180353 KJ).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was conducted in accordance with the approved institutional guidelines of Beihua University in China. The Ethics Committee of Beihua University (2018015) approved this study and all experimental protocols.
Informed consent
All enrolled participants of this study provided written informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, C., Zhan, Y., Ma, X. et al. B7-H4 facilitates proliferation and metastasis of colorectal carcinoma cell through PI3K/Akt/mTOR signaling pathway. Clin Exp Med 20, 79–86 (2020). https://doi.org/10.1007/s10238-019-00590-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-019-00590-7