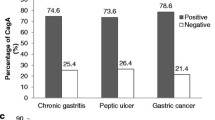
Abstract
The severity of Helicobacter pylori-related diseases varies greatly among infected individuals and seems to be influenced by both host and bacterial factors. Infection with a cytotoxin-associated gene pathogenicity island (Cag PAI)-positive H. Pylori strain causes a higher grade of gastric mucosal inflammation than an infection caused by a negative strain. Furthermore, such an infection is associated with severe atrophic gastritis and gastric adenocarcinoma. NOD1 protein is a cytosolic pattern recognition receptor that responds to peptidoglycan delivered by H. Pylori cag pathogenicity island. The aim of this study is to investigate whether the presence of the NOD1 G796A polymorphism has any influence on the clinical outcomes of Cag PAI-positive H. Pylori. Both Helicobacter pylori and CagA-positive 150 patients were considered eligible for the study. In this selected group, NOD1 G796A was detected by using polymerase chain reaction/restriction fragment length polymorphism. Activity and severity of gastritis, atrophy, intestinal metaplasia and Helicobacter pylori density were assessed in body and antral biopsies. Also post-therapy controls for predicting Helicobacter pylori persistence were done. The correlations of these parameters were determined by SPSS 15 packet program for statistical analysis. Of the 150 CagA-positive patients, 57 had (38%) heterozygote (GA), and 29 had (19.3%) homozygote (AA) mutant variants of NOD1. The other 64 patients had (42.7%) wild-type DNA(GG). NOD1 796A allele carriers had higher risk for antral atrophy (OR = 13.35, 95% CI = 5.12–34.82) and antral intestinal metaplasia (OR = 2.71, 95% CI = 1.26–5.80). Carriage of the single nucleotide polymorphism of NOD1 G796A proved to be a significant risk factor for the Helicobacter pylori therapy failure (OR = 4.62, 95% CI = 1.67–12.79). Our results suggest that carriage of the NOD1 G796A mutation increases the susceptibility of gastric epithelial cells for intestinal metaplasia and atrophy when infected by CagA-positive Helicobacter pylori strains. Additionally, it increases the ratio of eradication failure.

Similar content being viewed by others
References
Polenghi A, Bossi F, Fischetti F, Durigutto P, Cabrelle A, Tamassia T, Cassatella MA, Montecucco C, Tedesco F, Bernard M (2007) The neutrophil activating protein of Helicobacter pylori crosses endothelia to promote neutrophil adhesion in vitro. J Immunol 178:1312–1320
Backert S, Ziska E, Brinkman V, Zimmy-Arndt U, Fauconnier A, Jungblut PR, Naumann M, Meyer FT (2000) Translocation of the Helicobacter pylori CagA protein in gastric epithelial cells by a type 4 secretion apparatus. Cell Microbiol 2:155–164
Amieva MR, El-Omar EM (2008) Host bacterial interactions in Helicobacter pylori infection. Gastroenterology 134:306–323
George JT, Boughan PK, Karageorgiou H, Bajaj-Eliot M (2003) Host anti-microbial response to Helicobacter pylori infection. Mol Immunol 40:451–456
D’Ellos MM, Andersen LP (2007) Helicobacter pylori inflammation, immunity and vaccines. Helicobacter 12:15–19
Minohara Y, Boyd DK, Hawkins HK, Ernst PB, Patel J, Crowe SE (2007) The effect of the cag pathogenicity Island on binding of Helicobacter pylori to gastric epithelial cells and the subsequent induction of apoptosis. Helicobacter 12:583–590
Ikenoue T, Maeda S, Ogura K, Akanuma M, Mitsuno Y, Imai Y, Yoshida H, Shiratori Y, Omato M (2001) Determination of Helicobacter pylori virulence by simple gene analysis of the cag pathogenicity Island. Clin Diagn Lab Immunol 8:181–186
Salehi Z, Jelodar MH, Rassa M, Aheki M (2009) Helicobacter pylori CagA status and peptic ulcer disease in Iran. Dig Dis Sci 54(3):608–613
Andrzejewska J, Lee SK, Olbermann P, Lotzing N, Katzowitsch E, Linz B, Achtman M, Kado CI, Suerbaum S, Josenhans C (2006) Characterization of the pili ortholog of the Helicobacter pylori type 4 cag pathogenicity apparatus, a surface-associated protein expressed during infection. J Bacteriol 188:5865–5877
Israel DA, Peek RM Jr (2006) The role of persistence in Helicobacter pylori pathogenesis. Curr Opin Gastroenterol 22:3–7
Lochhead P, El-Omar EM (2007) Helicobacter pylori infection and gastric cancer. Best Pract Res Clin Gastroenterol 21:281–297
Viala J, Chaput C, Boneca IG, Cardona A, Girardin SE, Moran AP, Athman R, Mémet S, Huerre MR, Coyle AJ, DiStefano PS, Sansonetti PJ, Labigne A, Bertin J, Philpott DJ, Ferrero RL (2004) Nod1 responds to peptidoglycan delivered by the Helicobacter pylori cag pathogenicity island. Nat Immunol 5:1166–1174
Correia J, da S, Miranda Y, Leonard Y, Hsu J, Ulevitch RJ (2007) Regulation of NOD1 mediated signalling pathways. Cell Death Differ 14:830–839
Fritz JH, Bourhis LL, Sellge G, Magalhaes JG, Fsihi H, Kufer TA, Collins C, Viala J, Ferrero RL, Girardin SE, Philpott DJ (2007) Nod1 mediated innate immune recognition of peptidoglycan contributes to the onset of adaptive immunity. Immunity 26:445–459
Strober W, Murray PJ, Kitani A, Watanbe T (2006) Signalling pathways and molecular interactions of Nod1 and Nod2. Nat Rev 6:9–20
Chamaillard M, Hashimoto M, Horie Y, Masumoto J, Su Q, Saab L, Ogura Y, Kawasaki A, Fukase K, Kusumoto S, Valvano MA, Foster SJ, Mak TW, Nuñez G, Inohara N (2003) An essential role for Nod1 in host recognition of bacterial peptidoglycan containing diaminopimelic acid. Nat Immunol 4:702–707
Hasegewa M, Kawasaki A, Yang K, Fujimoto Y, Masumoto J, Breukink E, Nuñez G, Fukase K, Inohara N (2007) A role of lipophilic peptidoglycan related molecules in induction of Nod1-mediated immune responses. J Biol Chem 282:11757–11764
Hasegawa M, Yang K, Hashimoto M, Park J-H, Kim Y-G, Fujimoto Y, Nuñez G, Fukase K, Inohara N (2006) Differential release and distribution of Nod 1 and Nod 2 immunostimutory molecules among bacterial species and environments. J Biol Chem 281:29054–29063
Mcgovern DPB, Hysi P, Ahmad T, Van Heel DA, Moffatt MF, Carey A, Cookson WOC, Jewll DP (2005) Association between a complex insertion/deletion polymorphism in NOD1 and susceptibility to inflammatory bowel disease. Hum Mol Genet 14:1245–1250
Van Limbergen J, Nimmo ER, Russell RK, Drummond HE, Smith L, Anderson NH, Davies G, Arnott ID, Wilson DC, Satsangi J (2007) Investigation of NOD1/CARD4 variation in inflammatory bowel disease using a haplotype-tagging strategy. Hum Mol Genet 16:2175–2186
Hysi P, Kabesch M, Moffatt MF, Schedel M, Carr D, Zhang Y, Boardman B, Mutius E, Weiland SK, Leupold W, Fritzsch C, Klopp N, Musk AW, James A, Nunez G, Inohara N, Cookson WOC (2005) Nod1 variation, immunoglobulin E and asthma. Hum Mol Genet 14:935–941
Weidinger S, Klopp N, Rummler L, Wagenpfeil S, Novak N, Baurecht H-J, Groer W, Darsow U, Heinrich J, Gauger A, Schafer T, Jakob T, Behrendt H, Wichmann H-E, Ring J, Illig T (2005) Association of NOD1 polymorphisms with atopic eczema and related phenotypes. J Allergy Clin Immunol 116:177–184
Zouali H, Lesage S, Merlin F, Cėzard JP, Colombel JF, Belaiche J, Almer S, Tysk C, O’Morain C, Gassul M, Christensen S, Finkel Y, Modigliani R, Gower-Rousseau C, Macry J, Chamaillard M, Thomas G, Hugot JP (2003) CARD4/NOD1 is not involved in inflammatory bowel disease. Gut 52:71–74
Hofner P, Gyulai Z, Kiss ZF, Tiszai A, Tiszlavicz L, Gábor T, Szõke D, Molnăr B, Lonovics J, Tulassay Z, Măndi Y (2007) Genetic polymorphisms of Nod1 and IL-8 but not polymorphisms of TLR4 genes, are associated with Helicobacter pylori induced duedonal ulcer and gastritis. Helicobacter 12:124–131
Salih BA, Abasiyanik MF, Ahmed N (2007) A preliminary study on the genetic profile of cag pathogenicity-island and other virulent gene loci of Helicobacter pylori strains from Turkey. Infect Genet Evol 7:509–512
Sezikli M, Guliter S, Apan TZ, Aksoy A, Keles H, Ozkurt ZN (2006) Frequencies of serum antibodies to Helicobacter pylori CagA and VacA in a Turkish population with various gastroduedonal diseases. J Clin Pract 60:1239–1243
Kantarceken B, Aladag M, Atik E, Koksal F, Harputluoglu MM, Harputluoglu H, Karincaoglu M, Ates M, Yildirim B, Hilmioglu F (2003) Association of CagA and VacA presence with ulcer and non-ulcer dyspepsia in a Turkish population. World J Gastroenterol 9:1580–1583
Erzin Y, Altun S, Dobrucali A, Aslan M, Erdamar S, Dirican A, Tuncer M, Kocazeybek B (2006) Analysis of serum antibody profile against Helicobacter pylori VacA and CagA antigens in Turkish patients with duedonal ulcer. World J Gastroenterol 12:6869–6873
Gurbuz AK, Ozel AM, Yazgan Y, Gunay A, Ozdemir S, Demirturk L, Gurol E (2001) Seropositivity against Helicobacter pylori AagA in Turkish gastric cancer patients. J Clin Gastroenterol 33:389–392
Serin E, Yilmaz U, Kunefeci G, Kunefeci G, Ozer B, Gumurdulu Y, Guçlu M, Kayaselcuk F, Boyacioğlu S (2003) Serum positive CagA in patients with non-ulcer dyspepsia and peptic ulcer disease from two centers in different regions of Turkey. World J Gastroenterol 9:833–835
Saribasak H, Salih BA, Yamaoka Y, Sander E (2004) Analysis of Helicobacter pylori genotypes and correlation with clinical outcome in Turkey. J Clin Microbiol 42:1648–1651
Yilmaz M, Aydin A, Ungan M, Gunsar F, Ersoz G, Ozutemiz O, Akarca U, Cavusoglu H (1999) The relationship between CagA positivity and serum gastrin and TNF-alpha levels in patients with chronic active gastritis and duedonal ulcer associated with Helicobacter pylori. Turk J Gastroenterol 10:385–390
Fenoglio-Preiser CM, Noffsinger AE, Stemmermann GN, Lantz PE, Isaacson PG (2008) The nonneoplastic stomach. In: Lewin KJ, Appelman HD (eds) Gastrointestinal pathology, an atlas and text, 3rd edn. Lippincott Williams and Wilkins, Philadelphia, pp 135–233
Molnar T, Hofner P, Nagy F, Lakatos PL, Fischer S, Lakatos L, Kovacs A, Altorjay I, Papp M, Palatka K, Demeter P, Tulassay Z, Nyari T, Miheller P, Papp J, Mandi Y, Lonovics J (2007) NOD1 gene E266K polymorphism is associated with disease susceptibility but not with disease phenotype or NOD2/CARD15 in Hungarian patients with Crohn’s disease. Dig Liver Dis 39:1064–1070
Ohkuma K, Okada M, Murayama H, Seo M, Maeda K, Kanda M, Okaba N (2000) Association of Helicobacter pylori infection with atrophic gastritis and intestinal metaplasia. J Gastroenterol Hepatol 15:1105–1112
Satoh K, Osawa H, Yoshizawa M, Nakano H, Hirasawa T, Kihira K, Sunago K (2008) Assessment of atrophic gastritis using the OLGA system. Helicobacter 13:225–229
Matsuhisa TM, Yamada NY, Kato SK, Matsukura NM (2003) Helicobacter pylori infection, mucosal atrophy and intestinal metaplasia in Asian populations: a comparative study in age, gender and endoscopic diagnosis matched subjects. Helicobacter 8:29–35
Ding S-Z, Torok AM, Smith MF, Goldberg JB (2005) Toll like receptor 2 mediated gene expression in epithelial cells during Helicobacter pylori infection. Helicobacter 10:193–204
Ding S-Z, Minohara Y, Fan XJ, Wang J, Reyes VE, Patel J, Dirden-Kramer B, Boldogh I, Ernst PB, Crowe SE (2007) Helicobacter pylori infection induces oxidative stress and programmed cell death in human gastric epithelial cells. Infect Immun 75:4030–4039
Weck MN, Brenner H (2008) Association of Helicobacter pylori infection with chronic atrophic gastritis: meta analysis according to type of disease definition. Int J Cancer 123:874–881
Le’Negarate G, Ricci V, Hofman V, Mograbi B, Hofman P, Rossi B (2001) Epithelial intestinal cell apoptosis induced by Helicobacter pylori depends on expression of the cag pathogenicity Island phenotype. Infect Immun 69:5001–5009
Uemura N, Okamato S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, Taniyama K, Sasaki N, Schlemper RJ (2001) Helicobacter pylori infection and the development of gastric cancer. NEJM 345:784–789
Mimuro H, Suzuki T, Nagai S, Rieder G, Suzuki M, Nagai T, Fujita Y, Nagamatsu K, Ishijima N, Koyasu S, Haas R, Sasakawa C (2007) Helicobacter pylori dampens gut epithelial self renewal by inhibiting apoptosis, a bacterial strategy to enhance colonization of the stomach. Cell Host Microbe 2:250–263
Weck MN, Brenner H (2008) Association of Helicobacter pylori infection with chronic atrophic gastritis: meta analysis according to type of disease definition. Int J Cancer 123:874–881
Vorobjova T, Maaroos HI, Uibo R (2008) Immune response to Helicobacter pylori and its association with the dynamics of chronic gastritis in the antrum and corpus. APMIS 116:465–476
Hatekeyama M (2006) Helicobacter pylori CagA a bacterial intruder conspiring gastric carcinogenesis. Int J Cancer 119:1217–1223
Backert S, Selbach M (2008) Role of type 4 secretion in Helicobacter pylori pathogenesis. Cell Microbiol 10:1573–1581
Demirturk L, Melih AM, Yazgan Y, Solmazgul E, Yildirim S, Gultepe M, Gurbuz AK (2001) CagA status in dyspeptic patients with and without peptic ulcer disease in Turkey: association with histopathologic findings. Helicobacter 6:163–168
Wu AH, Crabtree JE, Leslie Berstein, Hawtin P, Cockburn M, Chiu-chen Tseng, Forman D (2003) Role of Helicobacter pylori CagA+ strains and risk of adenocarcinoma of the stomach and esophagus. Int J Cancer 103:815–821
Murata-Kamiya N, Kurashima Y, Teishikata Y, Yamahashi Y, Saito Y, Aburatani H, Akiyama T Jr, Azuma T, Hatakeyama M (2007) Helicobacter pylori CagA interacts with e-cadherin and deregulates the β-catenin signal that promotes intestinal transdifferentiation in gastric epithelial cells. Oncogene 26:4617–4626
Parsonnet J, Freidman GD, Orentreich N, Vogelman H (1997) Risk of gastric cancer in people with CagA positive or CagA negative Helicobacter pylori infection. Gut 40:297–301
Fischer W, Püls J, Buhrdorf R, Gebert B, Odenbreit S, Haas R (2001) Systematic mutagenesis of the Helicobacter pylori cag pathogenicity island: essential genes for CagA translocation in host cells and induction of interleukin-8. Mol Microbiol 42:1337–1348
Carneiro LAM, Travassos LH, Girardin SE (2007) Nod like receptors in innate immunity and inflammatory diseases. Ann Med 39:581–593
Ferrero RL (2005) Innate immune recognition of the extracellular mucosal pathogen, Helicobacter pylori. Mol Immunol 42:879–885
Werts C, Girardin SE, Philpott DJ (2006) TIR, CARD and PYRIN: three domains for an antimicrobial triad. Cell Death Differ 13:798–815
Yoo NJ, Park WS, Kim SY, Reed JC, Son SG, Lee JY, Lee SH (2002) Nod1, a CARD protein, enhances pro-interleukin-1β processing through the interaction wit pro-caspase 1. Biochem Biophys Res Commun 299:652–658
Conflict of interest statement
We have no conflict of interest related to the publication of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kara, B., Akkiz, H., Doran, F. et al. The significance of E266K polymorphism in the NOD1 gene on Helicobacter Pylori infection: an effective force on pathogenesis?. Clin Exp Med 10, 107–112 (2010). https://doi.org/10.1007/s10238-009-0077-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10238-009-0077-6