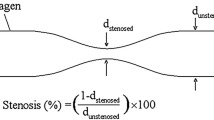
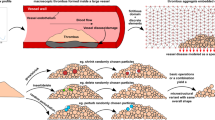
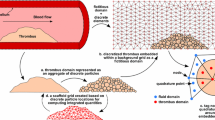
Abstract
The generation of occlusive thrombi in stenotic arteries involves the rapid deposition of millions of circulating platelets under high shear flow. The process is mediated by the formation of molecular bonds of several distinct types between platelets; the bonds capture the moving platelets and stabilize the growing thrombi under flow. We investigated the mechanisms behind occlusive thrombosis in arteries with a two-phase continuum model. The model explicitly tracks the formation and rupture of the two types of interplatelet bonds, the rates of which are coupled with the local flow conditions. The motion of platelets in the thrombi results from competition between the viscoelastic forces generated by the interplatelet bonds and the fluid drag. Our simulation results indicate that stable occlusive thrombi form only under specific combinations for the ranges of model parameters such as rates of bond formation and rupture, platelet activation time, and number of bonds required for platelet attachment.















Similar content being viewed by others
References
Anand M, Rajagopal K, Rajagopal K (2005) A model for the formation and lysis of blood clots. Pathophysiol Haemost Thromb 34:109–120
Arya M, López J, Romo G, Dong J, McIntire L, Moake J, Anvari B (2002) Measurement of the binding forces between von Willebrand factor and variants of platelet glycoprotein Ibalpha using optical tweezers. Lasers Surg Med 30:306–12
Bell G (1978) Models for the specific adhesion of cells to cells. Science 200:618–627
Bennett JS (2005) Structure and function of the platelet integrin \(\alpha _{\rm IIb} \beta _3\). J Clin Invest 115:3363–3369
Chtcheglova LA, Shubeita GT, Sekatskii SK, Dietler G (2004) Force spectroscopy with a small dithering of AFM tip: a method of direct and continuous measurement of the spring constant of single molecules and molecular complexes. Biophys J 86:1177–1184
Colace T, Diamond S (2013) Direct observation of von Willebrand factor elongation and fiber formation on collagen during acute whole blood exposure to pathological flow. Arterioscler Thromb Vasc Biol 33:105–13
Doggett T, Girdhar G, Lawshé A, Schmidtke D, Laurenzi I, Diamond S, Diacovo T (2002) Selectin-like kinetics and biomechanics promote rapid platelet adhesion in flow: the GPIb(alpha)-vWF tether bond. Biophys J 83:194–205
Dopheide S, Maxwell M, Jackson S (2002) Shear-dependent tether formation during platelet translocation on von Willebrand factor. Blood 99:159–67
Du J, Fogelson AL (2018) A two-phase mixture model of platelet aggregation. Math Med Biol 35:225–256
Du J, Kim D, Alhawael G, Ku DN, Fogelson AL (2020) Clot permeability, agonist transport, and platelet binding kinetics in arterial thrombosis. Biophys J 119(10):2102–2115
Du J, Aspray E, Fogelson A (2021) Computational investigation of platelet thrombus mechanics and stability in stenotic channels. J Biomech 122:110398
Fogelson AL (1992) Continuum models of platelet aggregation: formulation and mechanical properties. SIAM JAM 52:1089–1110
Fogelson AL, Guy RD (2004) Platelet-wall interactions in continuum models of platelet aggregation: formulation and numerical solution. Math Med Biol 21:293–334
Fogelson AL, Guy RD (2008) Immersed-boundary-type models of intravascular platelet aggregation. Comput Meth Appl Mech Eng 197:2087–2104
Fogelson A, Neeves K (2015) Fluid mechanics of blood clotting. Ann Rev Fluid Mech 47:377–403
Fu H, Jiang Y, Yang D, Scheiflinger F, Wong WP, Springer TA (2017) Flow-induced elongation of von Willebrand factor precedes tension-dependent activation. Nat Commun 8:1–12
Gardiner EE, Arthur JF, Shen Y, Karunakaran D, Moore LA, Am Esch JS, Andrew RK, Berndt MC (2010) GPIb\(\alpha\)-selective activation of platelets induces platelet signaling events comparable to GPVI activation events. Platelets 21:244–52
Goldsmith HL, Karino T (1987) Interactions of human blood cells with the vascular endothelium. Ann NY Acad Sci 516:468–483
Heemskerk JW, George M, Rook MB, Sage SO (2001) Ragged spiking of free calcium in ADP-stimulated human platelets: regulation of puff-like calcium signals in vitro and ex vivo. J Physiol 535:625–635
Jackson SP, Nesbitt WS, Kulkarni S (2003) Signaling events underlying thrombus formation. J Thromb Haemost 1:1602–1612
Jackson S, Nesbitt W, Westein E (2009) Dynamics of platelet thrombus formation. J Thromb Haemost 7:17–20
Kasirer-Friede A, Cozzi M, Mazzucato M, De Marco L, Ruggeri Z, Shattil S (2004) Signaling through GP Ib-IX-V activates alpha IIb beta 3 independently of other receptors. Blood 103:3403–11
Kim J, Zhang C, Zhang X, Springer T (2010) A mechanically stabilized receptor-ligand flex-bond important in the vasculature. Nature 466:992–995
Leiderman K, Fogelson AL (2011) Grow with the flow: a spatial-temporal model of platelet deposition and blood coagulation under flow. Math Med Biol 28:47–84
Litvinov RI, Barsegov V, Schissler AJ, Fisher AR, Bennet JS, Weisel JW, Shuman H (2011) Dissociation of bimolecular \(\alpha _{\rm IIb} \beta _3\)-fibrinogen complex under a constant tensile force. Biophys J 100:165–173
Matsui H, Sugimoto M, Mizuno T, Tsuji S, Miyata S, Matsuda MAY (2002) Distinct and concerted functions of von Willebrand factor and fibrinogen in mural thrombus growth under high shear flow. Blood 100:3604–3610
McCabe W, Smith J, Harriott P (2004) Unit operations of chemical engineering, 7th edn. McGraw Hill, Englewood Cliffs
Mitchell J, Dunster J, Kriek N, Unsworth A, Sage T, Mohammed Y, De Simone I, Taylor K, Bye A, Ólafsson G, Brunton M, Mark S, Dymott L, Whyte A, Ruparelia N, Gibbins J, Jones C (2023) The rate of platelet activation determines thrombus size and structure at arterial shear. J Thromb Haemost 23:00333
Ruggeri Z (2009) Platelet adhesion under flow. Microcirculation 16:58–83
Ruggeri ZM, Mendolicchio GL (2015) Interaction of von Willebrand fractor with platelets and the vessel wall. Hämostaseologie 35:211–224
Schneider SW, Nuschele S, Wixforth A, Gorzelanny C et al (2007) Shear-induced unfolding triggers adhesion of von Willebrand factor fibers. Proc Natl Acad Sci 104:7899–7903
Steppich D, Angerer J, Opfer J, Sritharan K, Schneider S, Thalhammer S, Wixforth A, Alexander-Katz A, Schneider M (2008) Relaxation of ultralarge vWF bundles in a microfluidic-AFM hybrid reactor. Biochem Biophys Res Commun 369:507–12
Storti F, van de Vosse F (2014) A continuum model for platelet plug formation, growth, and deformation. Int J Numer Methods Biomed Eng 30:1541–1557
Storti F, van Kempen T, van de Vosse F (2014) A continuum model for platelet plug formation and growth. Int J Numer Methods Biomed Eng 30:634–658
Tokarev A, Sirakov I, Panasenko G, Volpert V, Shnol E, Butylin A, Ataullakhanov F (2012) A continuous mathematical model of platelet thrombus formation in blood flow. Russ J Numer Anal Math Model 27:191–212
Weisel JW, Litvinov RI (2017) Fibrin formation, structure and properties. Subcell Biochem 82:405–456
Weller F (2010) A free boundary problem modeling thrombus growth. J Math Biol 61:805–818
Welling PJ, Ku DN (2012) Mechanisms of platelet capture under very high shear. Cardiovasc Eng Technol 3:161–170
Xu S, Xu Z, Kim OV, Litvinov RI, Weisel JW, Alber M (2017) Model predictions of deformation, embolization and permeability of partially obstructive blood clots under variable shear flow. J R Soc Interface 14:20170441
Yago T, Lou J, Wu T, Yang J, Miner J, Coburn L, Lopez JA, Cruz MA, Dong J, McIntire LV, McEver RP, Zhu C (2008) Platelet glycoprotein Ib\(\alpha\) forms catch bonds with human wt vWF but not with type 2b von Willebrand disease vWF. J Clin Investig 118:3195–207
Yan B, Hu DD, Knowles SK, Smith JW (2000) Probing chemical and conformational differences in the resting and active conformers of platelet integrin \(\alpha _{\rm IIb} \beta _3\). J Biol Chem 275:7249–7260
Zheng Y, Chen J, López J (2015) Flow-driven assembly of vWF fibres and webs in in vitro microvessels. Nat Commun 6:7858
Zheng X, Yasdani A, Li H, Humphrey JD, Karniadakis GE (2020) A three-dimensional phase-field model for multiscale modeling of thrombus biomechanics in blood vessels. PLoS Comput Biol 16:1007709
Zhussupbekov M, Rojano RM, Wu W-T, Antaki JF (2022) von Willebrand factor unfolding mediates platelet deposition in a model of high-shear thrombosis. Biophys J 121:4033–4047
Acknowledgements
The authors thank David N. Ku for helpful discussions about arterial thrombosis.
Funding
Funding for the project was provided in part by NSF Grant DMS-1716898 and NIH Grant 1R01HL151984–01.
Author information
Authors and Affiliations
Contributions
JD and AF contributed to the development of the model and computational methods, and the design of the computational simulations. JD carried out the simulations. JD and AF analyzed the results and wrote and edited the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Du, J., Fogelson, A.L. A computational investigation of occlusive arterial thrombosis. Biomech Model Mechanobiol 23, 157–178 (2024). https://doi.org/10.1007/s10237-023-01765-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-023-01765-8