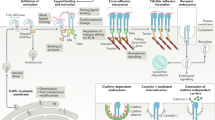
Abstract
Many types of cancer cells overexpress bulky glycoproteins to form a thick glycocalyx layer. The glycocalyx physically separates the cell from its surroundings, but recent work has shown that the glycocalyx can paradoxically increase adhesion to soft tissues and therefore promote the metastasis of cancer cells. This surprising phenomenon occurs because the glycocalyx forces adhesion molecules (called integrins) on the cell’s surface into clusters. These integrin clusters have cooperative effects that allow them to form stronger adhesions to surrounding tissues than would be possible with equivalent numbers of un-clustered integrins. These cooperative mechanisms have been intensely scrutinized in recent years. A more nuanced understanding of the biophysical underpinnings of glycocalyx-mediated adhesion could uncover therapeutic targets, deepen our general understanding of cancer metastasis, and elucidate general biophysical processes that extend far beyond the realm of cancer research. This work examines the hypothesis that the glycocalyx has the additional effect of increasing mechanical tension experienced by clustered integrins. Integrins function as mechanosensors that undergo catch bonding—meaning the application of moderate tension increases integrin bond lifetime relative to the lifetime of integrins experiencing low tension. In this work, a three-state chemomechanical catch bond model of integrin tension is used to investigate catch bonding in the presence of a bulky glycocalyx. A pseudo-steady-state approximation is applied, which relies on the assumption that integrin bond dynamics occur on a much faster timescale than the evolution of the full adhesion between the plasma membrane and the substrate. Force-dependent kinetic rate constants are used to calculate a steady-state distribution of integrin-ligand bonds for Gaussian-shaped adhesion geometries. The relationship between the energy of the system and adhesion geometry is then analyzed in the presence and absence of catch bonding in order to evaluate the extent to which catch bonding alters the energetics of adhesion formation. This modeling suggests that a bulky glycocalyx can lightly trigger catch bonding, increasing the bond lifetime of integrins at adhesion edges by up to 100%. The total number of integrin-ligand bonds within an adhesion is predicted to increase by up to ~ 60% for certain adhesion geometries. Catch bonding is predicted to decrease the activation energy of adhesion formation by ~ 1–4 kBT, which translates to a ~ 3–50 × increase in the kinetic rate of adhesion nucleation. This work reveals that integrin mechanics and clustering likely both contribute to glycocalyx-mediated metastasis.
Graphical abstract









Similar content being viewed by others
Data availability
All code and simulation data presented in this work is available upon reasonable request.
References
Asaro RJ, Lin K, Zhu Q (2019) Mechanosensitivity occurs along the adhesome’s force train and affects traction stress. Biophys J 117:1599–1614. https://doi.org/10.1016/j.bpj.2019.08.039
Atilgan E, Ovryn B (2009) Nucleation and growth of integrin adhesions. Biophys J 96:3555–3572. https://doi.org/10.1016/j.bpj.2009.02.023
Barnes JM et al (2018) A tension-mediated glycocalyx–integrin feedback loop promotes mesenchymal-like glioblastoma. Nat Cell Biol 20:1203–1214. https://doi.org/10.1038/s41556-018-0183-3
Bast RC Jr, Hennessy B, Mills GB (2009) The biology of ovarian cancer: new opportunities for translation. Nat Rev Cancer 9:415–428. https://doi.org/10.1038/nrc2644
Bell GI (1978) Models for the specific adhesion of cells to cells. Science 200:618–627
Bidone TC, Skeeters AV, Oakes PW, Voth GA (2019) Multiscale model of integrin adhesion assembly. PLoS Comput. Biol. 15:e1007077. https://doi.org/10.1371/journal.pcbi.1007077
Blanchard A (2021) Burnt bridge ratchet motor force scales linearly with polyvalency: a computational study. Soft Matter. https://doi.org/10.1039/D1SM00676B
Blanchard AT, Salaita K (2021) Multivalent molecular tension probes as anisotropic mechanosensors: concept and simulation. Phys. Biol. 18:034001
Brockman JM et al (2017) Mapping the 3D orientation of piconewton integrin traction forces. Nat Methods 15:115. https://doi.org/10.1038/nmeth.4536
Cabodi S et al (2004) Integrin regulation of epidermal growth factor (EGF) receptor and of EGF-dependent responses. Biochem Soc Trans 32:438–442. https://doi.org/10.1042/bst0320438
Desgrosellier JS, Cheresh DA (2010) Integrins in cancer: biological implications and therapeutic opportunities. Nat Rev Cancer 10:9–22. https://doi.org/10.1038/nrc2748
Elosegui-Artola A et al (2014) Rigidity sensing and adaptation through regulation of integrin types. Nat Mater 13:631–637. https://doi.org/10.1038/nmat3960
Freeman SA et al (2016) Integrins form an expanding diffusional barrier that coordinates phagocytosis. Cell 164:128–140. https://doi.org/10.1016/j.cell.2015.11.048
Gallego O (2015) Nonsurgical treatment of recurrent glioblastoma. Current oncol (Toronto, Ont.) 22:e273-281. https://doi.org/10.3747/co.22.2436
Geiger B, Yamada KM (2011) Molecular architecture and function of matrix adhesions. Cold Spring Harbor Perspect Biol. https://doi.org/10.1101/cshperspect.a005033
Helfrich W (1973) Elastic properties of lipid bilayers: theory and possible experiments. Zeitschrift Für Naturforschung C 28:693–703
Hingorani SR et al (2016) Phase Ib study of PEGylated recombinant human hyaluronidase and gemcitabine in patients with advanced pancreatic cancer. Clin Cancer Res 22:2848–2854. https://doi.org/10.1158/1078-0432.Ccr-15-2010
Hynes RO (2002) Integrins: bidirectional, allosteric signaling machines. Cell 110:673–687. https://doi.org/10.1016/S0092-8674(02)00971-6
Kechagia JZ, Ivaska J, Roca-Cusachs P (2019) Integrins as biomechanical sensors of the microenvironment. Nat Rev Mol Cell Biol 20:457–473. https://doi.org/10.1038/s41580-019-0134-2
Kong F, García AJ, Mould AP, Humphries MJ, Zhu C (2009) Demonstration of catch bonds between an integrin and its ligand. J Cell Biol 185:1275. https://doi.org/10.1083/jcb.200810002
Li J, Springer TA (2017) Integrin extension enables ultrasensitive regulation by cytoskeletal force. Proc Natl Acad Sci 114:4685. https://doi.org/10.1073/pnas.1704171114
Louis DN et al (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820. https://doi.org/10.1007/s00401-016-1545-1
MacKay L, Khadra A (2020) The bioenergetics of integrin-based adhesion, from single molecule dynamics to stability of macromolecular complexes. Comput Struct Biotechnol J 18:393–416. https://doi.org/10.1016/j.csbj.2020.02.003
Moro L et al (2002) Integrin-induced epidermal growth factor (EGF) receptor activation requires c-Src and p130Cas and leads to phosphorylation of specific EGF receptor tyrosines. J Biol Chem 277:9405–9414. https://doi.org/10.1074/jbc.M109101200
Nordenfelt P, Elliott HL, Springer TA (2016) Coordinated integrin activation by actin-dependent force during T-cell migration. Nat Commun 7:13119. https://doi.org/10.1038/ncomms13119
Nordenfelt P et al (2017) Direction of actin flow dictates integrin LFA-1 orientation during leukocyte migration. Nat Commun 8:2047. https://doi.org/10.1038/s41467-017-01848-y
Oria R et al (2017) Force loading explains spatial sensing of ligands by cells. Nature 552:219. https://doi.org/10.1038/nature24662
Paszek MJ, Boettiger D, Weaver VM, Hammer DA (2009) Integrin clustering is driven by mechanical resistance from the glycocalyx and the substrate. PLoS Comput. Biol. 5:e1000604. https://doi.org/10.1371/journal.pcbi.1000604
Paszek MJ et al (2014) The cancer glycocalyx mechanically primes integrin-mediated growth and survival. Nature 511:319. https://doi.org/10.1038/nature13535
Pinho SS, Reis CA (2015) Glycosylation in cancer: mechanisms and clinical implications. Nat Rev Cancer 15:540. https://doi.org/10.1038/nrc3982
Rahn JJ, Dabbagh L, Pasdar M, Hugh JC (2001) The importance of MUC1 cellular localization in patients with breast carcinoma. Cancer 91:1973–1982
Samadi-Dooki A, Shodja HM, Malekmotiei L (2015) The effect of the physical properties of the substrate on the kinetics of cell adhesion and crawling studied by an axisymmetric diffusion-energy balance coupled model. Soft Matter 11:3693–3705. https://doi.org/10.1039/C5SM00394F
Sarvestani AS (2013) The effect of substrate rigidity on the assembly of specific bonds at biological interfaces. Soft Matter 9:5927–5932. https://doi.org/10.1039/C3SM00036B
Shibue T, Weinberg RA (2009) Integrin beta1-focal adhesion kinase signaling directs the proliferation of metastatic cancer cells disseminated in the lungs. Proc Natl Acad Sci USA 106:10290–10295. https://doi.org/10.1073/pnas.0904227106
Sun Z, Costell M, Fässler R (2019) Integrin activation by talin, kindlin and mechanical forces. Nat Cell Biol 21:25–31. https://doi.org/10.1038/s41556-018-0234-9
Thomas W et al (2006) Catch-bond model derived from allostery explains force-activated bacterial adhesion. Biophys J 90:753–764. https://doi.org/10.1529/biophysj.105.066548
Thompson CB et al (2010) Enzymatic depletion of tumor hyaluronan induces antitumor responses in preclinical animal models. Mol Cancer Ther 9:3052–3064. https://doi.org/10.1158/1535-7163.Mct-10-0470
Tinder TL et al (2008) MUC1 enhances tumor progression and contributes toward immunosuppression in a mouse model of spontaneous pancreatic adenocarcinoma. J Immunol 181:3116–3125. https://doi.org/10.4049/jimmunol.181.5.3116
Wade A et al (2013) Proteoglycans and their roles in brain cancer. FEBS J 280:2399–2417
Wang X, Ha T (2013) Defining single molecular forces required to activate integrin and notch signaling. Science 340:991. https://doi.org/10.1126/science.1231041
Wang X, Lan H, Li J, Su Y, Xu L (2015) Muc1 promotes migration and lung metastasis of melanoma cells. Am J Cancer Res 5:2590–2604
Woods EC, Yee NA, Shen J, Bertozzi CR (2015) Glycocalyx engineering with a recycling glycopolymer that increases cell survival in vivo. Angewandte Chemie Int Ed Engl 54:15782–15788. https://doi.org/10.1002/anie.201508783
Woods EC et al (2017) A bulky glycocalyx fosters metastasis formation by promoting G1 cell cycle progression. eLlife 6:e25752. https://doi.org/10.7554/eLife.25752
Xiao H, Woods EC, Vukojicic P, Bertozzi CR (2016) Precision glycocalyx editing as a strategy for cancer immunotherapy. Proc Natl Acad Sci 113:10304–10309. https://doi.org/10.1073/pnas.1608069113
Zhang Y et al (2018) Platelet integrins exhibit anisotropic mechanosensing and harness piconewton forces to mediate platelet aggregation. Proc Natl Acad Sci U S A 115:325. https://doi.org/10.1073/pnas.1710828115
Acknowledgements
This work was supported by the United States National Cancer Institute F99/K00 fellowship, grant number F99CA245789 / K00CA245789.
Author information
Authors and Affiliations
Contributions
A.T.B. Performed all work
Corresponding author
Ethics declarations
Competing interests
This author has no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Blanchard, A.T. Can a bulky glycocalyx promote catch bonding in early integrin adhesion? Perhaps a bit. Biomech Model Mechanobiol 23, 117–128 (2024). https://doi.org/10.1007/s10237-023-01762-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-023-01762-x