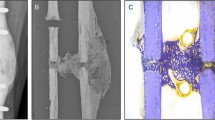
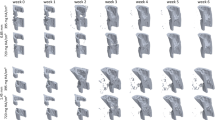
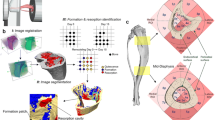
Abstract
Bone healing has been traditionally described as a four-phase process: inflammatory response, soft callus formation, hard callus development, and remodeling. The remodeling phase has been largely neglected in most numerical mechanoregulation models of fracture repair in favor of capturing early healing using a pre-defined callus domain. However, in vivo evidence suggests that remodeling occurs concurrently with repair and causes changes in cortical bone adjacent to callus that are typically neglected in numerical models of bone healing. The objective of this study was to use image processing techniques to quantify this early-stage remodeling in ovine osteotomies. To accomplish this, we developed a numerical method for radiodensity profilometry with optimization-based curve fitting to mathematically model the bone density gradients in the radial direction across the cortical wall and callus. After assessing data from 26 sheep, we defined a dimensionless density fitting function that revealed significant remodeling occurring in the cortical wall adjacent to callus during early healing, a 23% average reduction in density compared to intact. This fitting function is robust for modeling radial density gradients in both intact bone and fracture repair scenarios and can capture a wide variety of the healing responses. The fitting function can also be scaled easily for comparison to numerical model predictions and may be useful for validating future mechanoregulatory models of coupled fracture repair and remodeling.






Similar content being viewed by others
References
Ament C, Hofer EP (2000) A fuzzy logic model of fracture healing. J Biomech 33:961–968. https://doi.org/10.1016/S0021-9290(00)00049-X
Anani T, Castillo AB (2021) Mechanically-regulated bone repair. Bone. https://doi.org/10.1016/j.bone.2021.116223
Bahney CS, Zondervan RL, Allison P et al (2019) Cellular biology of fracture healing. J Orthop Res 37:35–50. https://doi.org/10.1002/jor.24170
Bissinger O, Götz C, Wolff KD et al (2017) Fully automated segmentation of callus by micro-CT compared to biomechanics. J Orthop Surg Res 12:1–9. https://doi.org/10.1186/s13018-017-0609-9
Buie HR, Campbell GM, Klinck RJ et al (2007) Automatic segmentation of cortical and trabecular compartments based on a dual threshold technique for in vivo micro-CT bone analysis. Bone 41:505–515. https://doi.org/10.1016/j.bone.2007.07.007
Campbell GM, Sophocleous A (2014) Quantitative analysis of bone and soft tissue by micro-computed tomography: applications to ex vivo and in vivo studies. Bonekey Rep 3:1–12. https://doi.org/10.1038/bonekey.2014.59
Claes L, Eckert-Hübner K, Augat P (2002) The effect of mechanical stability on local vascularization and tissue differentiation in callus healing. J Orthop Res 20:1099–1105. https://doi.org/10.1016/S0736-0266(02)00044-X
Claes L, Recknagel S, Ignatius A (2012) Fracture healing under healthy and inflammatory conditions. Nat Rev Rheumatol 8:133–143. https://doi.org/10.1038/nrrheum.2012.1
Einhorn TA, Gerstenfeld LC (2015) Fracture healing: mechanisms and interventions. Nat Rev Rheumatol 11:45–54. https://doi.org/10.1038/nrrheum.2014.164
Engelhardt L, Niemeyer F, Christen P et al (2021) Simulating metaphyseal fracture healing in the distal radius. Biomechanics 1:29–42. https://doi.org/10.3390/biomechanics1010003
Freedman BR, Rodriguez AB, Leiphart RJ et al (2018) Dynamic loading and tendon healing affect multiscale tendon properties and ECM stress transmission. Sci Rep 8:1–13. https://doi.org/10.1038/s41598-018-29060-y
Ghiasi MS, Chen J, Vaziri A et al (2017) Bone fracture healing in mechanobiological modeling: a review of principles and methods. Bone Rep 6:87–100. https://doi.org/10.1016/j.bonr.2017.03.002
Ghiasi MS, Chen JE, Rodriguez EK et al (2019) Computational modeling of human bone fracture healing affected by different conditions of initial healing stage. BMC Musculoskelet Disord 20:1–14. https://doi.org/10.1186/s12891-019-2854-z
Isaksson H, Wilson W, van Donkelaar CC et al (2006) Comparison of biophysical stimuli for mechano-regulation of tissue differentiation during fracture healing. J Biomech 39:1507–1516. https://doi.org/10.1016/j.jbiomech.2005.01.037
Lacroix D, Prendergast PJ (2002) A mechano-regulation model for tissue differentiation during fracture healing: analysis of gap size and loading. J Biomech 35:1163–1171. https://doi.org/10.1016/S0021-9290(02)00086-6
Loeffler J, Duda GN, Sass FA, Dienelt A (2018) The metabolic microenvironment steers bone tissue regeneration. Trends Endocrinol Metab 29:99–110. https://doi.org/10.1016/j.tem.2017.11.008
Manjubala I, Liu Y, Epari DR et al (2009) Spatial and temporal variations of mechanical properties and mineral content of the external callus during bone healing. Bone 45:185–192. https://doi.org/10.1016/j.bone.2009.04.249
Marsell R, Einhorn TA (2011) The biology of fracture healing. Injury 42:551–555. https://doi.org/10.1016/j.injury.2011.03.031
Molthen RC, Jameson J, Albert CI, Smith PA, Harris GF (2016) Role of micro-CT in the visualization, measurement, and quantification of bone structure in osteogenesis imperfecta. Shriners Hospitals for Children -Chicago, pp 195–216
Mora-macías J, Pajares A, Miranda P et al (2017) Mechanical characterization via nanoindentation of the woven bone developed during bone transport. J Mech Behav Biomed Mater 74:236–244. https://doi.org/10.1016/j.jmbbm.2017.05.031
Morgan E, Mason Z, Chien K, Pfeiffer A, Barnes G, Einhorn TGL, Morgan EF, Mason ZD et al (2009) Micro-computed tomography assessment of fracture healing: relationships among callus structure, composition, and mechanical function. Bone 44:335–344. https://doi.org/10.1016/j.bone.2008.10.039
Pietsch M, Niemeyer F, Simon U et al (2018) Modelling the fracture-healing process as a moving-interface problem using an interface-capturing approach. Comput Methods Biomech Biomed Eng 21:512–520. https://doi.org/10.1080/10255842.2018.1487554
Pivonka P, Dunstan CR (2012) Role of mathematical modeling in bone fracture healing. Bonekey Rep 1:1–10. https://doi.org/10.1038/bonekey.2012.221
Preininger B, Checa S, Molnar FL et al (2011) Spatial-temporal mapping of bone structural and elastic properties in a sheep model following osteotomy. Ultrasound Med Biol 37:474–483. https://doi.org/10.1016/j.ultrasmedbio.2010.12.007
Ren T, Dailey HL (2020) Mechanoregulation modeling of bone healing in realistic fracture geometries. Biomech Model Mechanobiol. https://doi.org/10.1007/s10237-020-01340-5
Scheuren AC, Vallaster P, Kuhn GA et al (2020) Mechano-regulation of trabecular bone adaptation is controlled by the local in vivo environment and logarithmically dependent on loading frequency. Front Bioeng Biotechnol 8:1–13. https://doi.org/10.3389/fbioe.2020.566346
Schwarzenberg P, Klein K, Ferguson SJ et al (2021a) Virtual mechanical tests out-perform morphometric measures for assessment of mechanical stability of fracture healing in vivo. J Orthop Res 39:727–738. https://doi.org/10.1002/jor.24866
Schwarzenberg P, Ren T, Klein K et al (2021b) Domain-independent simulation of physiologically relevant callus shape in mechanoregulated models of fracture healing. J Biomech. https://doi.org/10.1016/j.jbiomech.2021.110300
Seeman E (2015) Growth and age-related abnormalities in cortical structure and fracture risk. Endocrinol Metab 30:419–428. https://doi.org/10.3803/EnM.2015.30.4.419
Shefelbine SJ, Augat P, Claes L, Simon U (2005) Trabecular bone fracture healing simulation with finite element analysis and fuzzy logic. J Biomech 38:2440–2450. https://doi.org/10.1016/j.jbiomech.2004.10.019
Simon U, Augat P, Utz M, Claes L (2011) A numerical model of the fracture healing process that describes tissue development and revascularisation. Comput Methods Biomech Biomed Eng 14:79–93. https://doi.org/10.1080/10255842.2010.499865
Steiner M, Claes L, Ignatius A et al (2013) Prediction of fracture healing under axial loading, shear loading and bending is possible using distortional and dilatational strains as determining mechanical stimuli. J R Soc Interface. https://doi.org/10.1098/rsif.2013.0389
Steiner M, Claes L, Ignatius A et al (2014) Disadvantages of interfragmentary shear on fracture healing—mechanical insights through numerical simulation. J Orthop Res 32:865–872. https://doi.org/10.1002/jor.22617
Wang M, Yang N (2018) Three-dimensional computational model simulating the fracture healing process with both biphasic poroelastic finite element analysis and fuzzy logic control. Sci Rep 8:1–14. https://doi.org/10.1038/s41598-018-25229-7
Wehner T, Steiner M, Ignatius A et al (2014) Prediction of the time course of callus stiffness as a function of mechanical parameters in experimental rat fracture healing studies—a numerical study. PLoS ONE 9:1–16. https://doi.org/10.1371/journal.pone.0115695
Wilson CJ, Schütz MA, Epari DR (2017) Computational simulation of bone fracture healing under inverse dynamisation. Biomech Model Mechanobiol 16:5–14. https://doi.org/10.1007/s10237-016-0798-x
Acknowledgements
The preclinical studies from which this data was obtained were funded by the Johnson & Johnson Family of Companies. The authors wish to thank Beat Lechmann (Johnson & Johnson Family of Companies) for agreeing to grant access to the ovine study data for our analyses. This material is based upon work supported by the National Science Foundation under Grant No. 1943287. Any opinions, findings, and conclusions or recommendations expressed in this material are those of the authors and do not necessarily reflect the views of the Johnson & Johnson Family of Companies or the National Science Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
BvR is a paid consultant and receives research support as a principal investigator from Johnson & Johnson Family of Companies. The other authors report no conflicts relevant to this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ren, T., Klein, K., von Rechenberg, B. et al. Image-based radiodensity profilometry measures early remodeling at the bone-callus interface in sheep. Biomech Model Mechanobiol 21, 615–626 (2022). https://doi.org/10.1007/s10237-021-01553-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-021-01553-2