Abstract
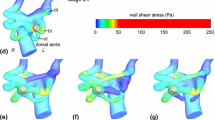
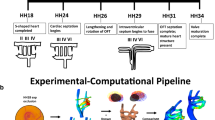
The microstructure for mature vessels has been investigated in detail, while there is limited information about the embryonic stages, in spite of their importance in the prognosis of congenital heart defects. It is hypothesized that the embryonic vasculature represents a disorganized but dynamic soft tissue, which rapidly evolves toward a specialized multi-cellular vascular structure under mechanical loading. Here the microstructural evolution process of the embryonic pharyngeal aortic arch structure was simulated using an in ovo validated long-term growth and remodeling computational model, implemented as an in-house FEBio plug-in. Optical coherence tomography-guided servo-null pressure measurements are assigned as boundary conditions through the critical embryonic stages. The accumulation of key microstructural constituents was recorded through zoom confocal microscopy for all six embryonic arch arteries simultaneously. The total amount and the radial variation slope of the collagen along the arch wall thickness in different arch types and for different embryonic times, with different dimension scales, were normalized and compared statistically. The arch growth model shows that the stress levels around the lumen boundary increase from \(\approx 270 \;{\text{Pa}}\) (Stage 18) to a level higher than \(\approx 600 \;{\text{Pa}}\) (Stage 24), depending on matrix constituent production rates, while the homeostatic strain level is kept constant. The statistical tests show that although the total collagen levels differentiate among bilateral positions of the same arch, the shape coefficient of the matrix microstructural gradient changes with embryonic time, proving radial localization, in accordance with numerical model results. In vivo cell number (DAPI) and vascular endothelial growth factor distributions followed similar trends.








Similar content being viewed by others
References
Alford PW, Humphrey JD, Taber LA (2008) Growth and remodeling in a thick-walled artery model: effects of spatial variations in wall constituents. Biomech Model Mechanobiol 7:245–262. https://doi.org/10.1007/s10237-007-0101-2
Ateshian GA, Morrison B, Holmes JW, Hung CT (2012) Mechanics of cell growth. Mech Res Commun 42:118–125. https://doi.org/10.1016/j.mechrescom.2012.01.010
Baaijens F, Bouten C, Driessen N (2010) Modeling collagen remodeling. J Biomech 43:166–175. https://doi.org/10.1016/j.jbiomech.2009.09.022
Bergwerff M, Verberne ME, DeRuiter MC, Poelmann RE, Gittenberger-de-Groot AC (1998) Neural crest cell contribution to the developing circulatory system. Circ Res 82:221
Celik MGS, Karakaya C, Cakiroglu AI, Karahuseyinoglu S, Lashkarinia S, Pekkan K (2019) How great arteries grow? Early embryonic aortic arch micro-structure and its reversibility following altered mechanical load release in review not published
Chuong CJ, Fung YC (1983) Three-dimensional stress distribution in arteries. J Biomech Eng 105:268–274. https://doi.org/10.1115/1.3138417
Chuong CJ, Fung YC (1986) On residual stresses in arteries. J Biomech Eng 108:189–192. https://doi.org/10.1115/1.3138600
Clark EB, Hu N (1982) Developmental hemodynamic changes in the chick embryo from stage 18 to 27. Circ Res 51:810–815
DeRuiter MC, Gittenberger-de Groot AC, Poelmann RE, VanIperen L, Mentink MM (1993) Development of the pharyngeal arch system related to the pulmonary and bronchial vessels in the avian embryo. With a concept on systemic-pulmonary collateral artery formation. Circulation 87:1306
Donmazov S, Piskin S, Pekkan K (2015) Noninvasive in vivo determination of residual strains and stresses. J Biomech Eng 137:061011. https://doi.org/10.1115/1.4030071
Driessen NJB, Peters GWM, Huyghe JM, Bouten CVC, Baaijens FPT (2003) Remodelling of continuously distributed collagen fibres in soft connective tissues. J Biomech 36:1151–1158. https://doi.org/10.1016/S0021-9290(03)00082-4
Goktas S, Uslu FE, Kowalski WJ, Ermek E, Keller BB, Pekkan K (2016) Time-series interactions of gene expression. Vascular growth and hemodynamics during early embryonic arterial development. PLoS One 11:e0161611. https://doi.org/10.1371/journal.pone.0161611
Göktepe S, Abilez OJ, Kuhl E (2010) A generic approach towards finite growth with examples of athlete’s heart, cardiac dilation, and cardiac wall thickening. J Mech Phys Solids 58:1661–1680. https://doi.org/10.1016/j.jmps.2010.07.003
Gürsan Çoban MÇ, Ermek E, Pekkan K (2018) Isotropic material properties of embryonic aortic arches from OCT guided micro-vascular pressure measurements (O0019). In: 8th World congress of biomechanics, Dublin, Ireland, 8th July, 2018
Hamburger V, Hamilton HL (1992) A series of normal stages in the development of the chick embryo. Dev Dyn 195:231–272. https://doi.org/10.1002/aja.1001950404
Hu N, Clark EB (1989) Hemodynamics of the stage 12 to stage 29 chick embryo. Circ Res 65:1665
Humphrey JD (2008) Mechanisms of arterial remodeling in hypertension. Hypertension 52:195
Karakaya C, Goktas S, Celik M, Kowalski WJ, Keller BB, Pekkan K (2018) Asymmetry in mechanosensitive gene expression during aortic arch morphogenesis. Sci Rep 8:1–14
Kolesová H, Roelink H, Grim M (2008) Sonic hedgehog is required for the assembly and remodeling of branchial arch blood vessels. Dev Dyn 237:1923–1934. https://doi.org/10.1002/dvdy.21608
Kowalski WJ, Dur O, Wang Y, Patrick MJ, Tinney JP, Keller BB, Pekkan K (2013) Critical transitions in early embryonic aortic arch patterning and hemodynamics. PLoS One 8:e60271. https://doi.org/10.1371/journal.pone.0060271
Kowalski WJ, Teslovich NC, Menon PG, Tinney JP, Keller BB, Pekkan K (2014) Left atrial ligation alters intracardiac flow patterns and the biomechanical landscape in the chick embryo. Dev Dyn 243:652–662
Lindsey SE et al (2015) Growth and hemodynamics after early embryonic aortic arch occlusion. Biomech Model Mechanobiol 14:735–751. https://doi.org/10.1007/s10237-014-0633-1
Lindsey SE, Butcher JT, Vignon-Clementel IE (2018) Cohort-based multiscale analysis of hemodynamic-driven growth and remodeling of the embryonic pharyngeal arch arteries. Development 145:dev162578. https://doi.org/10.1242/dev.162578
Maas SA, Ellis BJ, Ateshian GA, Weiss JA (2012) FEBio: finite elements for biomechanics. J Biomech Eng 134:011005. https://doi.org/10.1115/1.4005694
Machyshyn IM, Bovendeerd PHM, van de Ven AAF, Rongen PMJ, van de Vosse FN (2010) A model for arterial adaptation combining microstructural collagen remodeling and 3D tissue growth. Biomech Model Mechanobiol 9:671–687. https://doi.org/10.1007/s10237-010-0204-z
Pekkan K, Chang B, Uslu F, Mani K, Chen C-Y, Holzman R (2016) Characterization of zebrafish larvae suction feeding flow using μPIV and optical coherence tomography. Exp Fluids 57:112. https://doi.org/10.1007/s00348-016-2197-6
Plein A, Fantin A, Ruhrberg C (2015) Chapter six: neural crest cells in cardiovascular development. In: Trainor PA (ed) Current topics in developmental biology, vol 111. Academic Press, Cambridge, pp 183–200. https://doi.org/10.1016/bs.ctdb.2014.11.006
Ramos H (2007) A non-standard explicit integration scheme for initial-value problems. Appl Math Comput 189:710–718. https://doi.org/10.1016/j.amc.2006.11.134
Rausch MK, Dam A, Göktepe S, Abilez OJ, Kuhl E (2011) Computational modeling of growth: systemic and pulmonary hypertension in the heart. Biomech Model Mechanobiol 10:799–811. https://doi.org/10.1007/s10237-010-0275-x
Reinhardt JW, Gooch KJ (2018) An agent-based discrete collagen fiber network model of dynamic traction force-induced remodeling. J Biomech Eng 140:051003–051013. https://doi.org/10.1115/1.4037947
Rodriguez EK, Hoger A, McCulloch AD (1994) Stress-dependent finite growth in soft elastic tissues. J Biomech 27:455–467. https://doi.org/10.1016/0021-9290(94)90021-3
Roman BL, Pekkan K (2012) Mechanotransduction in embryonic vascular development. Biomech Model Mechanobiol 11:1149–1168. https://doi.org/10.1007/s10237-012-0412-9
Rueden CT, Schindelin J, Hiner MC, DeZonia BE, Walter AE, Arena ET, Eliceiri KW (2017) Image J2: ImageJ for the next generation of scientific image data. BMC Bioinform 18:529. https://doi.org/10.1186/s12859-017-1934-z
Ruhrberg C et al (2002) Spatially restricted patterning cues provided by heparin-binding VEGF-A control blood vessel branching morphogenesis. Genes Dev 16:2684–2698. https://doi.org/10.1101/gad.242002
Ryvlin J, Lindsey SE, Butcher JT (2019) Systematic analysis of the smooth muscle wall phenotype of the pharyngeal arch arteries during their reorganization into the great vessels and its association with hemodynamics. Anat Rec 302:153–162. https://doi.org/10.1002/ar.23942
Taber LA (1998a) A model for aortic growth based on fluid shear and fiber stresses. J Biomech Eng 120:348–354. https://doi.org/10.1115/1.2798001
Taber LA (1998b) An optimization principle for vascular radius including the effects of smooth muscle tone. Biophys J 74:109–114. https://doi.org/10.1016/S0006-3495(98)77772-0
Wang Y, Dur O, Patrick MJ, Tinney JP, Tobita K, Keller BB, Pekkan K (2009) Aortic arch morphogenesis and flow modeling in the chick embryo. Ann Biomed Eng 37:1069–1081. https://doi.org/10.1007/s10439-009-9682-5
Yablonka-Reuveni Z, Christ B, Benson JM (1998) Transitions in cell organization and in expression of contractile and extracellular matrix proteins during development of chicken aortic smooth muscle: evidence for a complex spatial and temporal differentiation program. Anat Embryol 197:421–437. https://doi.org/10.1007/s004290050154
Zahka KG, Hu N, Brin KP, Yin FC, Clark EB (1989) Aortic impedance and hydraulic power in the chick embryo from stages 18 to 29. Circ Res 64:1091
Acknowledgements
Funding was provided by the European Research Council (ERC) Grant 307460 (KP). We acknowledge Dr. Anthony Townley for his help in English proofreading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lashkarinia, S.S., Çoban, G., Ermek, E. et al. Spatiotemporal remodeling of embryonic aortic arch: stress distribution, microstructure, and vascular growth in silico. Biomech Model Mechanobiol 19, 1897–1915 (2020). https://doi.org/10.1007/s10237-020-01315-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-020-01315-6