Abstract
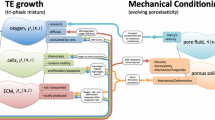
In this work a coupled model of solute transport and uptake, cell proliferation, extracellular matrix synthesis and remodeling of mechanical properties accounting for the impact of mechanical loading is presented as an advancement of a previously validated coupled model for free-swelling tissue-engineered cartilage cultures. Tissue-engineering constructs were modeled as biphasic with a linear elastic solid, and relevant intrinsic mechanical stimuli in the constructs were determined by numerical simulation for use as inputs of the coupled model. The mechanical dependent formulations were derived from a calibration and parametrization dataset and validated by comparison of normalized ratios of cell counts, total glycosaminoglycans and collagen after 24-h continuous cyclic unconfined compression from another dataset. The model successfully fit the calibration dataset and predicted the results from the validation dataset with good agreement, with average relative errors up to 3.1 and 4.3 %, respectively. Temporal and spatial patterns determined for other model outputs were consistent with reported studies. The results suggest that the model describes the interaction between the simultaneous factors involved in in vitro tissue-engineered cartilage culture under dynamic loading. This approach could also be attractive for optimization of culture protocols, namely through the application to longer culture times and other types of mechanical stimuli.









Similar content being viewed by others
References
Bandeiras C, Completo A (2013) Comparison between constitutive models for the solid phase of biphasic agarose/chondrocytes constructs for knee cartilage engineering. Comput Methods Biomech Biomed Eng 16(sup1):262–263
Bandeiras C, Completo A (2015) Comparison of mechanical parameters between tissue-engineered and native cartilage: a numerical study. Comput Methods Biomech Biomed Eng 18(sup1):1876–1877
Bandeiras C, Completo A, Ramos A (2014a) Compression, shear and bending on tissue-engineered cartilage: a numerical study. Comput Methods Biomech Biomed Eng 17(sup1):2–3
Bandeiras C, Completo A, Ramos A (2014b) Simulation of remodeling of tissue engineered condylar cartilage under static hydrostatic pressure. Biodental Eng III:83
Bandeiras C, Completo A, Ramos A (2015) Influence of the scaffold geometry on the spatial and temporal evolution of the mechanical properties of tissue-engineered cartilage: insights from a mathematical model. Biomech Model Mechanobiol 14(5):1057–1070
Benya PD, Shaffer JD (1982) Dedifferentiated chondrocytes reexpress the differentiated collagen phenotype when cultured in agarose gels. Cell 30(1):215–224
Buckley CT, Vinardell T, Kelly DJ (2010) Oxygen tension differentially regulates the functional properties of cartilaginous tissues engineered from infrapatellar fat pad derived mscs and articular chondrocytes. Osteoarthr Cartil 18(10):1345–1354
Buschmann MD, Kim YJ, Wong M, Frank E, Hunziker EB, Grodzinsky AJ (1999) Stimulation of aggrecan synthesis in cartilage explants by cyclic loading is localized to regions of high interstitial fluid flow. Arch biochem Biophys 366(1):1–7
Catt C, Schuurman W, Sengers B, Van Weeren P, Dhert W, Please C, Malda J (2011) Mathematical modelling of tissue formation in chondrocyte filter cultures. Eur Cell Mater 22:377–392
Chung C, Yang C, Chen C (2006) Analysis of cell growth and diffusion in a scaffold for cartilage tissue engineering. Biotechnol Bioeng 94(6):1138–1146
Chung C, Chen C, Chen C, Tseng C (2007) Enhancement of cell growth in tissue-engineering constructs under direct perfusion: modeling and simulation. Biotechnol Bioeng 97(6):1603–1616
Costa KD, Ho MM, Hung CT, (2003) Multi-scale measurement of mechanical properties of soft samples with atomic force microscopy. In: 2003 summer bioengineering conference. Florida, Key Biscaine, p 2
Davisson T, Kunig S, Chen A, Sah R, Ratcliffe A (2002) Static and dynamic compression modulate matrix metabolism in tissue engineered cartilage. J Orthop Res 20(4):842–848
Dijkman P, Bader D, Lee D, Chowdhury T, Heywood H (2005) Glucose utilisation of chondrocytes, with or without loading. In: External placement report, Technical University of Eindhoven and Queen Mary University, London
Evans RC, Quinn TM (2006) Dynamic compression augments interstitial transport of a glucose-like solute in articular cartilage. Biophys J 91(4):1541–1547
Foldager CB, Nielsen AB, Munir S, Ulrich-Vinther M, Søballe K, Bünger C, Lind M (2011) Combined 3D and hypoxic culture improves cartilage-specific gene expression in human chondrocytes. Acta Orthop 82(2):234–240
Galban CJ, Locke BR (1999) Analysis of cell growth kinetics and substrate diffusion in a polymer scaffold. Biotechnol Bioeng 65(2):121–132
Gao X, Zhu Q, Gu W (2015) Analyzing the effects of mechanical and osmotic loading on glycosaminoglycan synthesis rate in cartilaginous tissues. J Biomech 48(4):573–577
Gardiner B, Smith D, Pivonka P, Grodzinsky A, Frank E, Zhang L (2007) Solute transport in cartilage undergoing cyclic deformation. Comput Methods Biomech Biomed Eng 10(4):265–278
Haider MA, Olander JE, Arnold RF, Marous DR, McLamb AJ, Thompson KC, Woodruff WR, Haugh JM (2011) A phenomenological mixture model for biosynthesis and linking of cartilage extracellular matrix in scaffolds seeded with chondrocytes. Biomech Model Mechanobiol 10(6):915–924
Hossain MS, Bergstrom D, Chen X (2015) Modelling and simulation of the chondrocyte cell growth, glucose consumption and lactate production within a porous tissue scaffold inside a perfusion bioreactor. Biotechnol Rep 5:55–62
Hung CT, Mauck RL, Wang CCB, Lima EG, Ateshian GA (2004) A paradigm for functional tissue engineering of articular cartilage via applied physiologic deformational loading. Ann Biomed Eng 32(1):35–49
Khoshgoftar M, Wilson W, Ito K, van Donkelaar CC (2013) The effect of tissue-engineered cartilage biomechanical and biochemical properties on its post-implantation mechanical behavior. Biomech Model Mechanobiol 12(1):43–54
Kisiday JD, Jin M, DiMicco MA, Kurz B, Grodzinsky AJ (2004) Effects of dynamic compressive loading on chondrocyte biosynthesis in self-assembling peptide scaffolds. J Biomech 37(5):595–604
Knight M, Lee D, Bader D (1998) The influence of elaborated pericellular matrix on the deformation of isolated articular chondrocytes cultured in agarose. Biochimica et Biophysica Acta (BBA)-Molecular. Cell Res 1405(1):67–77
Lee DA, Bader DL (1997) Compressive strains at physiological frequencies influence the metabolism of chondrocytes seeded in agarose. J Orthop Res 15(2):181–188
Lee RB, Urban JP (1997) Evidence for a negative pasteur effect in articular cartilage. Biochem J 321(1):95–102
Lelli C, Sacco R, Causin P, Raimondi MT (2015) A poroelastic mixture model of mechanobiological processes in tissue engineering. part I: mathematical formulation. arXiv p 1512.02182
Li S, Oreffo RO, Sengers BG, Tare RS (2014) The effect of oxygen tension on human articular chondrocyte matrix synthesis: integration of experimental and computational approaches. Biotechnol Bioeng 111(9):1876–1885
Lin WY, Chang YH, Wang HY, Yang TC, Chiu TK, Huang SB, Wu MH (2014) The study of the frequency effect of dynamic compressive loading on primary articular chondrocyte functions using a microcell culture system. BioMed Res Int 2014:762570
Mauck RL, Hung CT, Ateshian GA (2003) Modeling of neutral solute transport in a dynamically loaded porous permeable gel: implications for articular cartilage biosynthesis and tissue engineering. J Biomech Eng 125(5):602–614
Mesallati T, Buckley CT, Nagel T, Kelly DJ (2013) Scaffold architecture determines chondrocyte response to externally applied dynamic compression. Biomech Model Mechanobiol 12(5):889–899
Mow VC, Kuei S, Lai WM, Armstrong CG (1980) Biphasic creep and stress relaxation of articular cartilage in compression: theory and experiments. J Biomech Eng 102(1):73–84
Myers K, Ateshian GA (2014) Interstitial growth and remodeling of biological tissues: tissue composition as state variables. J Mech Behav Biomed Mater 29:544–556
Nava MM, Raimondi MT, Pietrabissa R (2013) A multiphysics 3D model of tissue growth under interstitial perfusion in a tissue-engineering bioreactor. Biomech Model Mechanobiol 12(6):1169–1179
Ng KW, Wang CCB, Mauck RL, Kelly TAN, Chahine NO, Costa KD, Ateshian GA, Hung CT (2005) A layered agarose approach to fabricate depth-dependent inhomogeneity in chondrocyte-seeded constructs. J Orthop Res 23(1):134–141
Nikolaev N, Obradovic B, Versteeg HK, Lemon G, Williams DJ (2010) A validated model of gag deposition, cell distribution, and growth of tissue engineered cartilage cultured in a rotating bioreactor. Biotechnol Bioeng 105(4):842–853
Obradovic B, Meldon JH, Freed LE, Vunjak-Novakovic G (2000) Glycosaminoglycan deposition in engineered cartilage: experiments and mathematical model. AIChE J 46(9):1860–1871
Pierce DM, Ricken T, Holzapfel GA (2013) A hyperelastic biphasic fibre-reinforced model of articular cartilage considering distributed collagen fibre orientations: continuum basis, computational aspects and applications. Comput Methods Biomech Biomed Eng 16(12):1344–1361
Sacco R, Causin P, Zunino P, Raimondi MT (2011) A multiphysics/multiscale 2D numerical simulation of scaffold-based cartilage regeneration under interstitial perfusion in a bioreactor. Biomech Model Mechanobiol 10(4):577–589
Sengers B, Van Donkelaar C, Oomens C, Baaijens F (2004a) The local matrix distribution and the functional development of tissue engineered cartilage, a finite element study. Ann Biomed Eng 32(12):1718–1727
Sengers BG, Oomens CW, Baaijens FP (2004b) An integrated finite-element approach to mechanics, transport and biosynthesis in tissue engineering. J Biomech Eng 126(1):82–91
Sengers BG, Heywood HK, Lee DA, Oomens CW, Bader DL (2005) Nutrient utilization by bovine articular chondrocytes: a combined experimental and theoretical approach. J Biomech Eng 127(5):758–766
Takahashi T, Ogasawara T, Asawa Y, Mori Y, Uchinuma E, Takato T, Hoshi K (2007) Three-dimensional microenvironments retain chondrocyte phenotypes during proliferation culture. Tissue Eng 13(7):1583–1592
Tasci A, Ferguson SJ, Büchler P (2011) Numerical assessment on the effective mechanical stimuli for matrix-associated metabolism in chondrocyte-seeded constructs. J Tissue Eng Regen Med 5(3):210–219
Tsuang YH, Lin Y, Chen L, Cheng C, Sun JS (2008) Effect of dynamic compression on in vitro chondrocyte metabolism. Int J Artif Organs 31(5):439–449
Williamson AK, Chen AC, Sah RL (2001) Compressive properties and function composition relationships of developing bovine articular cartilage. J Orthop Res 19(6):1113–1121
Wilson CG, Bonassar LJ, Kohles SS (2002) Modeling the dynamic composition of engineered cartilage. Arch Biochem Biophys 408(2):246–254
Wilson W, Huyghe J, Van Donkelaar C (2007) Depth-dependent compressive equilibrium properties of articular cartilage explained by its composition. Biomech Model Mechanobiol 6(1–2):43–53
Wimmer M, Guenther H, Grad S, Lee C, Haenni M, Gogolewski S, Alini M (2003) Numerical simulation as a tool to evaluate loading regimes for tissue engineering. In: 49th annual meeting of the orthopaedic research society, New Orleans, LA
Zhang L, Szeri A (2005) Transport of neutral solute in articular cartilage: effects of loading and particle size. Proc R Soc Lond A Math Phys Eng Sci R Soc 461:2021–2042
Zhou S, Cui Z, Urban JP (2008) Nutrient gradients in engineered cartilage: metabolic kinetics measurement and mass transfer modeling. Biotechnol Bioeng 101(2):408–421
Acknowledgments
This work is supported by FCT Fundação para a Ciência e Tecnologia with the reference Project PTDC/EMS-TEC/3263/2014 and by Programa Operacional Competitividade e Internacionalização through the Project POCI-01-0145-FEDER-016574.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bandeiras, C., Completo, A. A mathematical model of tissue-engineered cartilage development under cyclic compressive loading. Biomech Model Mechanobiol 16, 651–666 (2017). https://doi.org/10.1007/s10237-016-0843-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-016-0843-9