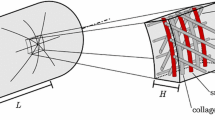
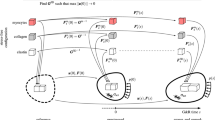
Abstract
A stabilizing criterion is derived for equations governing vascular growth and remodeling. We start from the integral state equations of the continuum-based constrained mixture theory of vascular growth and remodeling and obtain a system of time-delayed differential equations describing vascular growth. By employing an exponential form of the constituent survival function, the delayed differential equations can be reduced to a nonlinear ODE system. We demonstrate the degeneracy of the linearized system about the homeostatic state, which is a fundamental cause of the neutral stability observations reported in prior studies. Due to this degeneracy, stability conclusions for the original nonlinear system cannot be directly inferred. To resolve this problem, a sub-system is constructed by recognizing a linear relation between two states. Subsequently, Lyapunov’s indirect method is used to connect stability properties between the linearized system and the original nonlinear system, to rigorously establish the neutral stability properties of the original system. In particular, this analysis leads to a stability criterion for vascular expansion in terms of growth and remodeling kinetic parameters, geometric quantities and material properties. Numerical simulations were conducted to evaluate the theoretical stability criterion under broader conditions, as well as study the influence of key parameters and physical factors on growth properties. The theoretical results are also compared with prior numerical and experimental findings in the literature.




Similar content being viewed by others
References
Abdul-Hussien H, Soekhoe R, Weber E, Jan H, Kleemann R, Mulder A, Van Bockel J, Hanemaaijer R, Lindeman J (2007) Collagen degradation in the abdominal aneurysm: a conspiracy of matrix metalloproteinase and cysteine collagenases. Am J Pathol 170(3):809–817
Aparício P, Mandaltsi A, Boamah J, Chen H, Selimovic A, Bratby M, Uberoi R, Ventikos Y, Watton P (2014) Modelling the influence of endothelial heterogeneity on the progression of arterial disease: Application to abdominal aortic aneurysm evolution. Int J Numer Methods Biomed Eng 30(5):563–586
Austin G, Schievink W, Williams R (1989) Controlled pressure–volume factors in the enlargement of intracranial aneurysms. Neurosurgery 24(5):722–730
Baek S, Rajagopal K, Humphrey J (2005) Competition between radial expansion and thickening in the enlargement of an intracranial saccular aneurysm. J Elast 80(1–3):13–31
Baek S, Rajagopal K, Humphrey J (2006) A theoretical model of enlarging intracranial fusiform aneurysms. J Biomech Eng 128(1):142–149
Bogen D, McMahon T (1979) Do cardiac aneurysms blow out? Biophys J 27(2):301
Bots M, Hofman A, de Bruyn A, De Jong P, Grobbee D (1993) Isolated systolic hypertension and vessel wall thickness of the carotid artery. The Rotterdam Elderly Study. Arterioscler Thromb Vasc Biol 13(1):64–69
Choke E, Cockerill G, Wilson W, Sayed S, Dawson J, Loftus I, Thompson M (2005) A review of biological factors implicated in abdominal aortic aneurysm rupture. Eur J Vasc Endovasc Surg 30(3):227–244
Cyron C, Humphrey J (2014) Vascular homeostasis and the concept of mechanobiological stability. Int J Eng Sci 85:203–223
Cyron C, Wilson J, Humphrey J (2014) Mechanobiological stability: a new paradigm to understand the enlargement of aneurysms? J R Soc Interface 11(100):20140680
Dijk J, Van Der Graaf Y, Grobbee D, Banga J, Bots M et al (2004) Increased arterial stiffness is independently related to cerebrovascular disease and aneurysms of the abdominal aorta the second manifestations of arterial disease (smart) study. Stroke 35(7):1642–1646
Etminan N, Dreier R, Buchholz B, Beseoglu K, Bruckner P, Matzenauer C, Torner J, Brown R, Steiger H, Hänggi D et al (2014) Age of collagen in intracranial saccular aneurysms. Stroke 45(6):1757–1763
Figueroa A, Baek S, Taylor C, Humphrey J (2009) A computational framework for fluid–solid-growth modeling in cardiovascular simulations. Comput Methods Appl Mech Eng 198(45):3583–3602
Holzapfel G, Gasser T, Ogden R (2000) A new constitutive framework for arterial wall mechanics and a comparative study of material models. J Elast Phys Sci Solids 61(1–3):1–48
Huang Y, Gloviczki P, Duncan A, Kalra M, Hoskin T, Oderich G, McKusick M, Bower T (2008) Common iliac artery aneurysm: expansion rate and results of open surgical and endovascular repair. J Vasc Surg 47(6):1203–1211
Humphrey J (2006) Towards a theory of vascular growth and remodeling. In: Holzapfel G, Ogden R (eds) Mechanics of biological tissue. Springer, Berlin, pp 3–15. doi:10.1007/3-540-31184-X_1
Humphrey J (2013) Cardiovascular solid mechanics: cells, tissues, and organs. Springer, New York
Humphrey J, Rajagopal K (2002) A constrained mixture model for growth and remodeling of soft tissues. Mathematical Models and Methods in Applied Sciences 12(03):407–430
Nissen R, Cardinale G, Udenfriend S (1978) Increased turnover of arterial collagen in hypertensive rats. Proc Natl Acad Sci 75(1):451–453
Safar M, O’Rourke M, Frohlich E (2014) Blood pressure and arterial wall mechanics in cardiovascular diseases. Springer, Berlin
Satha G, Lindström S, Klarbring A (2014) A goal function approach to remodeling of arteries uncovers mechanisms for growth instability. Biomech Model Mechanobiol 13(6):1243–1259
Satta J, Juvonen T, Haukipuro K, Juvonen M, Kairaluoma M (1995) Increased turnover of collagen in abdominal aortic aneurysms, demonstrated by measuring the concentration of the aminoterminal propeptide of type iii procollagen in peripheral and aortal blood samples. J Vasc Surg 22(2):155–160
Sheidaei A, Hunley S, Zeinali-Davarani S, Raguin L, Baek S (2011) Simulation of abdominal aortic aneurysm growth with updating hemodynamic loads using a realistic geometry. Med Eng Phys 33(1):80–88
Valentin A, Humphrey J (2009) Evaluation of fundamental hypotheses underlying constrained mixture models of arterial growth and remodelling. Philos Trans R Soc Lond A Math Phys Eng Sci 367(1902):3585–3606
Verhulst F (1996) Nonlinear differential equations and dynamical systems. Springer, Berlin
Watton P, Raberger N, Holzapfel G, Ventikos Y (2009) Coupling the hemodynamic environment to the evolution of cerebral aneurysms: computational framework and numerical examples. J Biomech Eng 131(10):101003
Wolinsky H (1972) Long-term effects of hypertension on the rat aortic wall and their relation to concurrent aging changes: morphological and chemical studies. Circ Res 30(3):301–309
Wu J, Shadden S (2015) Coupled simulation of hemodynamics and vascular growth and remodeling in a subject-specific geometry. Ann Biomed Eng 43(7):1543–1554
Zeinali-Davarani S, Sheidaei A, Baek S (2011) A finite element model of stress-mediated vascular adaptation: application to abdominal aortic aneurysms. Comput Methods Biomech Biomed Eng 14(9):803–817
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, J., Shadden, S.C. Stability analysis of a continuum-based constrained mixture model for vascular growth and remodeling. Biomech Model Mechanobiol 15, 1669–1684 (2016). https://doi.org/10.1007/s10237-016-0790-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-016-0790-5