Abstract
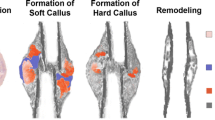
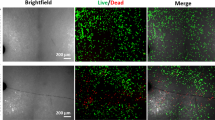
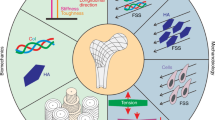
Uncovering the mechanisms of the sensitivity of bone healing to mechanical factors is critical for understanding the basic biology and mechanobiology of the skeleton, as well as for enhancing clinical treatment of bone injuries. This study refined an experimental method of measuring the strain microenvironment at the site of a bone injury during bone healing. This method used a rat model in which a well-controlled bending motion was applied to an osteotomy to induce the formation of pseudarthrosis that is composed of a range of skeletal tissues, including woven bone, cartilage, fibrocartilage, fibrous tissue, and clot tissue. The goal of this study was to identify both the features of the strain microenvironment associated with formation of these different tissues and the expression of proteins frequently implicated in sensing and transducing mechanical cues. By pairing the strain measurements with histological analyses that identified the regions in which each tissue type formed, we found that formation of the different tissue types occurs in distinct strain microenvironments and that the type of tissue formed is correlated most strongly to the local magnitudes of extensional and shear strains. Weaker correlations were found for dilatation. Immunohistochemical analyses of focal adhesion kinase and rho family proteins RhoA and CDC42 revealed differences within the cartilaginous tissues in the calluses from the pseudarthrosis model as compared to fracture calluses undergoing normal endochondral bone repair. These findings suggest the involvement of these proteins in the way by which mechanical stimuli modulate the process of cartilage formation during bone healing.










Similar content being viewed by others
References
Aizawa R et al (2012) Cdc42 is required for chondrogenesis and interdigital programmed cell death during limb development. Mech Dev 129:38–50. doi:10.1016/j.mod.2012.02.002
Alenghat FJ, Ingber DE (2002) Mechanotransduction: all signals point to cytoskeleton, matrix, and integrins. Sci STKE 2002:pe6. doi:10.1126/stke.2002.119.pe6
Arnsdorf EJ, Tummala P, Kwon RY, Jacobs CR (2009) Mechanically induced osteogenic differentiation-the role of RhoA. ROCKII and cytoskeletal dynamics. J Cell Sci 122:546–553. doi:10.1242/jcs.036293
Bonnarens F, Einhorn TA (1984) Production of a standard closed fracture in laboratory animal bone. J Orthop Res 2:97–101
Burke D, Dishowitz M, Sweetwyne M, Miedel E, Hankenson KD, Kelly DJ (2013) The role of oxygen as a regulator of stem cell fate during fracture repair in TSP2-null mice. J Orthop Res 31:1585–1596. doi:10.1002/jor.22396
Carter DR, Beaupre GS, Giori NJ, Helms JA (1998) Mechanobiology of skeletal regeneration. Clin Orthop 355:S41–S55
Claes L, Augat P, Suger G, Wilke HJ (1997) Influence of size and stability of the osteotomy gap on the success of fracture healing. J Orthop Res 15:577–584
Claes LE, Heigele CA (1999) Magnitudes of local stress and strain along bony surfaces predict the course and type of fracture healing. J Biomech 32:255–266
Cohen DM, Chen CS (2008) Mechanical control of stem cell differentiation. In StemBook, The Stem Cell Research Community StemBook (ed). http://www.stembook.org
Cullinane DM et al (2002) Induction of a neoarthrosis by precisely controlled motion in an experimental mid-femoral defect. J Orthop Res 20:579–586
Einhorn TA (1998) The cell and molecular biology of fracture healing. Clin Orthop 355:S7–S21
Epari DR, Schell H, Bail HJ, Duda GN (2006) Instability prolongs the chondral phase during bone healing in sheep. Bone 38:864–870
Etienne-Manneville S, Hall A (2002) Rho GTPases in cell biology. Nature 420:629–635
Gao L, McBeath R, Chen CS (2010) Stem cell shape regulates a chondrogenic versus myogenic fate through Rac1 and N-cadherin. Stem cells (Dayton, Ohio) 28:564–572
Gerstenfeld LC et al (2006) Three-dimensional reconstruction of fracture callus morphogenesis. J Histochem Cytochem 54:1215–1228
Gokhale N, Richards M, Oberai A, Barbone P (2005) Simultaneous elastic image registration and elastic modulus reconstruction. In Paper presented at the IEEE international symposium on biomedical imaging
Gomez-Benito MJ, Garcia-Aznar JM, Kuiper JH, Doblare M (2005) Influence of fracture gap size on the pattern of long bone healing: a computational study. J Theor Biol 235:105–119. doi:10.1016/j.jtbi.2004.12.023
Goodship AE, Kenwright J (1985) The influence of induced micromovement upon the healing of experimental tibial fractures. J Bone Joint Surg Br 67:650–655
Guilak F, Cohen DM, Estes BT, Gimble JM, Liedtke W, Chen CS (2009) Control of stem cell fate by physical interactions with the extracellular matrix. Cell stem cell 5:17–26
Haudenschild DR, Chen J, Pang N, Lotz MK, D’Lima DD (2010) Rho kinase-dependent activation of SOX9 in chondrocytes. Arthritis Rheum 62:191–200. doi:10.1002/art.25051
Hayward LN, Morgan EF (2009) Assessment of a mechano-regulation theory of skeletal tissue differentiation in an in vivo model of mechanically induced cartilage formation. Biomech Model Mechanobiol 8:447–455
Hosmer DW, Lemeshow S, Sturdivant RX (2013) Applied logistic regression, 3rd edn. Wiley, Hoboken
Ingber DE (2006) Cellular mechanotransduction: putting all the pieces together again. Faseb J 20:811–827
Isaksson H, van Donkelaar CC, Huiskes R, Ito K (2006) Corroboration of mechanoregulatory algorithms for tissue differentiation during fracture healing: comparison with in vivo results. J Orthop Res 24:898–907
Isaksson H, van Donkelaar CC, Ito K (2009) Sensitivity of tissue differentiation and bone healing predictions to tissue properties. J Biomech 42:555–564
Jin G, Sah RL, Li YS, Lotz M, Shyy JY, Chien S (2000) Biomechanical regulation of matrix metalloproteinase-9 in cultured chondrocytes. J Orthop Res 18:899–908. doi:10.1002/jor.1100180608
Kaibuchi K, Kuroda S, Amano M (1999) Regulation of the cytoskeleton and cell adhesion by the Rho family GTPases in mammalian cells. Annu Rev Biochem 68:459–486
Kim JB, Leucht P, Luppen CA, Park YJ, Beggs HE, Damsky CH, Helms JA (2007) Reconciling the roles of FAK in osteoblast differentiation, osteoclast remodeling, and bone regeneration. Bone 41:39–51. doi:10.1016/j.bone.2007.01.024
Kim YH, Lee JW (2009) Targeting of focal adhesion kinase by small interfering RNAs reduces chondrocyte redifferentiation capacity in alginate beads culture with type II collagen. J Cell Physiol 218:623–630. doi:10.1002/jcp.21637
Lacroix D, Prendergast PJ (2002) A mechano-regulation model for tissue differentiation during fracture healing: analysis of gap size and loading. J Biomech 35:1163–1171
Leucht P, Kim JB, Currey JA, Brunski J, Helms JA (2007) FAK-mediated mechanotransduction in skeletal regeneration. PLoS One 2:e390
McBeath R, Pirone DM, Nelson CM, Bhadriraju K, Chen CS (2004) Cell shape, cytoskeletal tension, and RhoA regulate stem cell lineage commitment. Dev Cell 6:483–495
McNary SM, Athanasiou KA, Reddi AH (2014) Transforming growth factor beta-induced superficial zone protein accumulation in the surface zone of articular cartilage is dependent on the cytoskeleton. Tissue Eng Part A 20:921–929. doi:10.1089/ten.TEA.2013.0043
Morgan EF, Salisbury Palomares KT, Gleason RE, Bellin DL, Chien KB, Unnikrishnan GU, Leong PL (2010) Correlations between local strains and tissue phenotypes in an experimental model of skeletal healing. J Biomech 43:2418–2424
Nagelkerke NJD (1991) A note on a general definition of the coefficient of determination. Biometrika 78:691–692
Oberai AA, Gokhale NH, Feijoo GR (2003) Solution of inverse problems in elasticity imaging using the adjoint method. Inverse Probl 19:297–313
Park MS, Kim YH, Lee JW (2010) FAK mediates signal crosstalk between type II collagen and TGF-beta 1 cascades in chondrocytic cells. Matrix Biol 29:135–142. doi:10.1016/j.matbio.2009.10.001
Prendergast PJ, Huiskes R, Soballe K (1997) ESB Research Award 1996. Biophysical stimuli on cells during tissue differentiation at implant interfaces. J Biomech 30:539–548
Sakai D et al (2011) Remodeling of actin cytoskeleton in mouse periosteal cells under mechanical loading induces periosteal cell proliferation during bone formation. PLoS One 6:e24847. doi:10.1371/journal.pone.0024847
Salisbury Palomares KT, Gleason RE, Mason ZD, Cullinane DM, Einhorn TA, Gerstenfeld LC, Morgan EF (2009) Mechanical stimulation alters tissue differentiation and molecular expression during bone healing. J Orthop Res 27:1123–1132
Sanz-Ramos P, Mora G, Ripalda P, Vicente-Pascual M, Izal-Azcarate I (2012) Identification of signalling pathways triggered by changes in the mechanical environment in rat chondrocytes. Osteoarthritis Cartilage 20:931–939. doi:10.1016/j.joca.2012.04.022
Shefelbine SJ, Augat P, Claes L, Simon U (2005) Trabecular bone fracture healing simulation with finite element analysis and fuzzy logic. J Biomech 38:2440–2450. doi:10.1016/j.jbiomech.2004.10.019
Simon U, Augat P, Utz M, Claes L (2011) A numerical model of the fracture healing process that describes tissue development and revascularisation. Comput Methods Biomech Biomed Engin 14:79–93. doi:10.1080/10255842.2010.499865
Sordella R, Jiang W, Chen GC, Curto M, Settleman J (2003) Modulation of Rho GTPase signaling regulates a switch between adipogenesis and myogenesis. Cell 113:147–158
Steiner M, Claes L, Ignatius A, Niemeyer F, Simon U, Wehner T (2013) Prediction of fracture healing under axial loading, shear loading and bending is possible using distortional and dilatational strains as determining mechanical stimuli. J R Soc Interface 10:20130389. doi:10.1098/rsif.2013.0389
Tong L, Buchman SR, Ignelzi MA Jr, Rhee S, Goldstein SA (2003) Focal adhesion kinase expression during mandibular distraction osteogenesis: evidence for mechanotransduction. Plast Reconstr Surg 111:211–222 discussion 223–214
Wang G, Beier F (2005) Rac1/Cdc42 and RhoA GTPases antagonistically regulate chondrocyte proliferation, hypertrophy, and apoptosis. J Bone Miner Res 20:1022–1031. doi:10.1359/JBMR.050113
Wang G, Woods A, Sabari S, Pagnotta L, Stanton LA, Beier F (2004) RhoA/ROCK signaling suppresses hypertrophic chondrocyte differentiation. J Biol Chem 279:13205–13214
Woods A, Beier F (2006) RhoA/ROCK signaling regulates chondrogenesis in a context-dependent manner. J Biol Chem 281:13134–13140. doi:10.1074/jbc.M509433200
Woods A, Wang G, Beier F (2007) Regulation of chondrocyte differentiation by the actin cytoskeleton and adhesive interactions. J Cell Physiol 213:1–8
Acknowledgments
The authors would like to acknowledge Paul Barbone and the Boston University Immunohistochemistry Core Facility for their technical support. Funding was provided by the National Institutes of Health (NIH) AR53353 (EFM), the National Science Foundation Civil, Mechanical and Manufacturing Innovation Division (NSF CMMI) 1266243 (EFM), and the Boston University Clinical and Translational Science Institute (BU CTSI) Grant #UL1RR025771.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10237_2015_670_MOESM1_ESM.pdf
Supplementary Material provides additional details and figures regarding the surgical and mechanical stimulation protocols, specimen preparation and image capture for strain measurements, the accuracy of the measurements of the displacement and strain, the sampling and variability of the strain fields and corresponding histology slides, as well as additional relative frequency and logistic regression plots for Edil, max and Edil, min (Pdf 1463 KB)
Rights and permissions
About this article
Cite this article
Miller, G.J., Gerstenfeld, L.C. & Morgan, E.F. Mechanical microenvironments and protein expression associated with formation of different skeletal tissues during bone healing. Biomech Model Mechanobiol 14, 1239–1253 (2015). https://doi.org/10.1007/s10237-015-0670-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-015-0670-4