Abstract
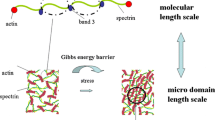
Studies of thermal fluctuations in discocytes, echinocytes, and spherocytes suggest that the coupling between lipid bilayer and cytoskeleton can affect viscoelastic behavior of single erythrocyte membranes. To test this hypothesis, we developed a 3D constitutive model describing viscoelastic behavior of erythrocyte membranes, at long relaxation times \(t \in [0.20\,\mathrm {s}, 1.05\,\mathrm {s}]\) and short relaxation times \(t \in [0.03\,\mathrm {s}, 0.20\,\mathrm {s}]\). The model was constructed using combination of spring and spring pot rheological elements arranged in parallel. The rearrangement of cytoskeleton induced by changing the bending state of lipid bilayer was described by a modified Eyring model. The model predictions point to an anomalous nature of energy dissipation and an ordered harmonic nature of the coupling mechanism described by a series of fractional derivatives of the order n \(\alpha \) (where \( n \in [- 1, 2]\)). As a result, the stress generated within the lipid bilayer is related to the rate of change of the irreversible stress within the cytoskeleton.




Similar content being viewed by others
References
Amin MS, Park YK, Lue N, Dasari RR, Badizadegan K, Feld MS, Popescu G (2007) Microrheology of red blood cell membrane using dynamics scattering microscopy. Opt Express 15(25):17001– 17009
Boss D, Hoffmann A, Rappaz B, Depeursinge C, Magistretti PJ, Van de Ville D, Marquet P (2012) Spatially-resolved eigenmode decomposition of red blood cells membrane fluctuations questions the role of ATP in flickering. PLoS ONE 7(8):e406671 1–e406671 10
Braunmuuller S, Schmid L, Sackmann E, Franke T (2012) Hydrodynamics deformation reveals two coupled modes/time scales of red blood cells relaxation. Soft Matter 8:11240–11248
Djordjevic VD, Jaric J, Fabry B, Fredberg JJ, Stamenovic D (2003) Fractional derivatives embody essential features of cell rheological behaviour. Ann Biomed Eng 31:692–699
Gov NS, Safran SA (2005) Red blood cell membrane fluctuations and shape controlled by ATP-Induced cytoskeletal defects. Biophys J 88:1859–1874
Gov NS (2007) Less is more: removing membrane attachments stiffness the RBC cytoskeleton. New J Phys 9(429):1–8
Gnanakaran S, Nymeyer H, Portman J, Kevin Y, Sanbonmatsu KY, Garcıa AE (2003) Peptide folding simulations. Curr Op Struct Biol 13:168–174
Gurtovenko AA, YuYa Gotlib (2001) Dynamics of inhomogeneous cross-linked polymers consisting of domains of different sizes. J Chem Phys 115(14):6785–6793
Harland CW, Bradley MJ, Parthasarathy R (2011) Retraction for “Phospholipid bilayers are viscoelastic”, by Harland CW, Bradley MJ, Parthasarathy R, which appeared in PNAS 107(45):19146–19150, 2010. PNAS 108(35):14705
Marmottant P, Mgharbel A, Kafer J, Audren B, Rieu JP, Vial JC, van der Sanden B, Maree AFM, Graner F, Delanoe-Ayari H (2009) The role of fluctuations and stress on the effective viscosity of cell aggregates. PNAS 106(41):17271–17275
Mikhalyov I, Samsonov A (2011) Lipid raft detecting in membranes of live erythrocytes. Biochem Biophys Acta 1808:1930–1939
Mofrad MRK, Karcher H, Kamm RD (2006) Continuum elastic or viscoelastic models for the cell. In: Mofrad MRK, Kamm RD (eds) Cytoskeletal mechanics, models and measurement. Cambridge University Press, New York, pp 71–84
Murell MP, Gardel ML (2012) F-actin buckling coordinates contractility and severing in a biomimeticactomyosin cortex. PNAS 109:20820–20825
Pajic-Lijakovic I, Ilic V, Bugarski B, Plavsic M (2010) The rearrangement of erythrocyte band 3 molecules and reversible osmotic holes formation under hypotonic conditions. Eur Biophys J 39(5):789–797
Park YK (2007) Optical rheology for live cell membranes. Master thesis Massachusetts Institute of Technology University Press, Boston
Peng Z, Asaro RJ, Zhu Q (2011) Multiscale modeling of erythrocytes in Stokes flow. J Fluid Mech 686:299–337
Peng Z, Li X, Pivkin IV, Dao M, Karniadakis GE, Suresh S (2013) Lipid bilayer and cytoskeletal interactions in a red blood cell. PNAS 110(33):13356–13361
Peng Z, Zhu Q (2013) Deformation of the erythrocyte cytoskeleton in tank treading motions. Soft Matter 9:7617–7627
Picas L, Rico F, Deforet M, Scheuring S (2013) Structural and mechanical heterogeneity of the erythrocyte membrane reveals hallmarks of membrane stability. ACS Nano 7(2):1054–1063
Podlubny I (1999) Fractional differential equations, mathematics in science and engineering, vol 198. London, Academic Press, p 78
Popescu G, Park YK, Dasari RR, Badizadegan K, Feld MS (2007) Coherence properties of red blood cell membrane motion. Phys Rev E 76(031902):1–5
Pradhan D, Tseng K, Cianci CD, Morrow JS (2004) Antibodies to hIA2 spectrin identify in-homogeneities in the erythrocyte membrane skeleton. Blood Cells Mol Dis 32:408–410
Puig-de-Morales-Marinkovic M, Turner KT, Butler JP, Fredberg JJ, Suresh S (2007) Viscoelasticity of the human red blood cell. Am J Physiol Cell Physiol 293:C597–C605
Rief M, Fernandez JM, Gaub HE (1998) Elastically coupled two-level systems as a model for biopolymer extensibility. Phys Rev Lett 81(21):4764–4767
Shkulipa SA, den Otter WK, Briels WJ (2005) Surface viscosity, diffusion and intermonolayer friction: simulating sheared amphipilic bilayers. Biophys J 89:823–829
Acknowledgments
The authors gratefully acknowledge funding support of the Ministry of Education, Science and Technological Development of the Republic Serbia (Grants III 46001, and III 46010). The authors thank Professor Gordana Vunjak-Novakovic (Columbia University) for useful suggestions which improved this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pajic-Lijakovic, I., Milivojevic, M. Modeling analysis of the lipid bilayer–cytoskeleton coupling in erythrocyte membrane. Biomech Model Mechanobiol 13, 1097–1104 (2014). https://doi.org/10.1007/s10237-014-0559-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-014-0559-7