Abstract
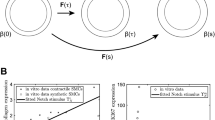
Mechanical stresses influence the structure and function of adult and developing blood vessels. When these stresses are perturbed, the vessel wall remodels to return the stresses to homeostatic levels. Constrained mixture models have been used to predict remodeling of adult vessels in response to step changes in blood pressure, axial length and blood flow, but have not yet been applied to developing vessels. Models of developing blood vessels are complicated by continuous and simultaneous changes in the mechanical forces. Understanding developmental growth and remodeling is important for treating human diseases and designing tissue-engineered blood vessels. This study presents a constrained mixture model for postnatal development of mouse aorta with multiple step increases in pressure, length and flow. The baseline model assumes that smooth muscle cells (SMCs) in the vessel wall immediately constrict or dilate the inner radius after a perturbation to maintain the shear stress and then remodel the wall thickness to maintain the circumferential stress. The elastin, collagen and SMCs have homeostatic stretch ratios and passive material constants that do not change with developmental age. The baseline model does not predict previously published experimental data. To approximate the experimental data, it must be assumed that the SMCs dilate a constant amount, regardless of the step change in mechanical forces. It must also be assumed that the homeostatic stretch ratios and passive material constants change with age. With these alterations, the model approximates experimental data on the mechanical properties and dimensions of aorta from 3- to 30-day-old mice.
Similar content being viewed by others
References
Alford PW, Humphrey JD, Taber LA (2008) Growth and remodeling in a thick-walled artery model: effects of spatial variations in wall constituents. Biomech Model Mechanobiol 7(4): 245–262. doi:10.1007/s10237-007-0101-2
Baek S, Rajagopal KR, Humphrey JD (2006) A theoretical model of enlarging intracranial fusiform aneurysms. J Biomech Eng 128(1): 142–149
Cardamone L, Valentin A, Eberth JF, Humphrey JD (2010) Modelling carotid artery adaptations to dynamic alterations in pressure and flow over the cardiac cycle. Math Med Biol. doi:dqq001
Clark ER (1918) Studies on the growth of blood-vessels in the tail of the frog larva—by observation and experiment on the living animal. Am J Anat 23: 37–88
Davis EC (1995) Elastic lamina growth in the developing mouse aorta. J Histochem Cytochem 43(11): 1115–1123
Di Stefano I, Koopmans DR, Langille BL (1998) Modulation of arterial growth of the rabbit carotid artery associated with experimental elevation of blood flow. J Vasc Res 35(1): 1–7. doi:jvr35001
Dobrin PB, Canfield TR (1984) Elastase, collagenase, and the biaxial elastic properties of dog carotid artery. Am J Physiol 247(1 Pt 2): H124–H131
Dye WW, Gleason RL, Wilson E, Humphrey JD (2007) Altered biomechanical properties of carotid arteries in two mouse models of muscular dystrophy. J Appl Physiol 103(2): 664–672
Faury G, Maher GM, Li DY, Keating MT, Mecham RP, Boyle WA (1999) Relation between outer and luminal diameter in cannulated arteries. Am J Physiol 277(5 Pt 2): H1745–H1753
Fonck E, Prod’hom G, Roy S, Augsburger L, Rufenacht DA, Stergiopulos N (2004) Effect of elastin degradation on carotid wall mechanics as assessed by a constituent-based biomechanical model. Am J Physiol Heart Circ Physiol 292(6): H2754–H2763. doi:01108.2006
Gleason RL, Humphrey JD (2004) A mixture model of arterial growth and remodeling in hypertension: altered muscle tone and tissue turnover. J Vasc Res 41(4): 352–363
Gleason RL, Taber LA, Humphrey JD (2004) A 2-d model of flow-induced alterations in the geometry, structure and properties of carotid arteries. J Biomech Eng 126: 371–381
Guo X, Kassab GS (2003) Variation of mechanical properties along the length of the aorta in c57bl/6 mice. Am J Physiol Heart Circ Physiol 285(6): H2614–H2622
Holzapfel GA, Gasser TC, Ogden RW (2000) A new constitutive framework for arterial wall mechanics and a comparative study of material models. J Elast 61(1–3): 1–48
Hornstra IK, Birge S, Starcher B, Bailey AJ, Mecham RP, Shapiro SD (2003) Lysyl oxidase is required for vascular and diaphragmatic development in mice. J Biol Chem 278(16): 14387–14393
Huang Y, Guo X, Kassab GS (2006) Axial nonuniformity of geometric and mechanical properties of mouse aorta is increased during postnatal growth. Am J Physiol Heart Circ Physiol 290(2): H657–H664
Humphrey JD, Eberth JF, Dye WW, Gleason RL (2009) Fundamental role of axial stress in compensatory adaptations by arteries. J Biomech 42(1): 1–8. doi:S0021-9290(08)00595-2
Humphrey JD, Rajagopal KR (2002) A constrained mixture model for growth and remodeling of soft tissues. Math Models Methods Appl Sci 12(3): 407–430
Ishii T, Kuwaki T, Masuda Y, Fukuda Y (2001) Postnatal development of blood pressure and baroreflex in mice. Auton Neurosci 94(1–2): 34–41
Jackson ZS, Dajnowiec D, Gotlieb AI, Langille BL (2005) Partial off-loading of longitudinal tension induces arterial tortuosity. Arter Thromb Vasc Biol 25(5): 957–962
Jackson ZS, Gotlieb AI, Langille BL (2002) Wall tissue remodeling regulates longitudinal tension in arteries. Circ Res 90(8): 918–925
Kelleher CM, McLean SE, Mecham RP (2004) Vascular extracellular matrix and aortic development. Curr Top Dev Biol 62: 153–188
Langille BL (1993) Remodeling of developing and mature arteries: endothelium, smooth muscle, and matrix. J Cardiovasc Pharmacol 21(1): S11–S17
Langille BL, Bendeck MP, Keeley FW (1989) Adaptations of carotid arteries of young and mature rabbits to reduced carotid blood flow. Am J Physiol 256(4 Pt 2): H931–H939
Langille BL, Brownlee RD, Adamson SL (1990) Perinatal aortic growth in lambs: relation to blood flow changes at birth. Am J Physiol 259(4 Pt 2): H1247–H1253
Li DY, Faury G, Taylor DG, Davis EC, Boyle WA, Mecham RP, Stenzel P, Boak B, Keating MT (1998) Novel arterial pathology in mice and humans hemizygous for elastin. J Clin Invest 102(10): 1783–1787
Li DY, Toland AE, Boak BB, Atkinson DL, Ensing GJ, Morris CA, Keating MT (1997) Elastin point mutations cause an obstructive vascular disease, supravalvular aortic stenosis. Hum Mol Genet 6(7): 1021–1028
Nakamura T, Lozano PR, Ikeda Y, Iwanaga Y, Hinek A, Minamisawa S, Cheng CF, Kobuke K, Dalton N, Takada Y, Tashiro K, Ross J Jr, Honjo T, Chien KR (2002) Fibulin-5/dance is essential for elastogenesis in vivo. Nature 415(6868): 171–175
Olivetti G, Anversa P, Melissari M, Loud AV (1980) Morphometric study of early postnatal development of the thoracic aorta in the rat. Circ Res 47(3): 417–424
Valentin A, Cardamone L, Baek S, Humphrey JD (2009) Complementary vasoactivity and matrix remodelling in arterial adaptations to altered flow and pressure. J R Soc Interface 6(32): 293–306. doi:92812083762PK762
Valentin A, Humphrey JD (2009) Modeling effects of axial extension on arterial growth and remodeling. Med Biol Eng Comput 47(9): 979–987. doi:10.1007/s11517-009-0513-5
Wagenseil JE, Ciliberto CH, Knutsen RH, Levy MA, Kovacs A, Mecham RP (2009) Reduced vessel elasticity alters cardiovascular structure and function in newborn mice. Circ Res 104(10): 1217–1224. doi:CIRCRESAHA.108.192054
Wagenseil JE, Mecham RP (2009) Vascular extracellular matrix and arterial mechanics. Physiol Rev 89(3): 957–989. doi:89/3/957
Wagenseil JE, Nerurkar NL, Knutsen RH, Okamoto RJ, Li DY, Mecham RP (2005) Effects of elastin haploinsufficiency on the mechanical behavior of mouse arteries. Am J Physiol Heart Circ Physiol 289(3): H1209–H1217
Wan W, Hansen L, Gleason RL Jr (2009) A 3-d constrained mixture model for mechanically mediated vascular growth and remodeling. Biomech Model Mechanobiol. doi:10.1007/s10237-009-0184-z
Westerhof N, Stergiopulos N, Noble M (2004) Snapshots of hemodynamics: an aid for clinical research and graduate education. p 42
Wiesmann F, Ruff J, Hiller KH, Rommel E, Haase A, Neubauer S (2000) Developmental changes of cardiac function and mass assessed with MRI in neonatal, juvenile, and adult mice. Am J Physiol Heart Circ Physiol 278(2): H652–H657
Wong LC, Langille BL (1996) Developmental remodeling of the internal elastic lamina of rabbit arteries: effect of blood flow. Circ Res 78(5): 799–805
Yanagisawa H, Davis EC, Starcher BC, Ouchi T, Yanagisawa M, Richardson JA, Olson EN (2002) Fibulin-5 is an elastin-binding protein essential for elastic fibre development in vivo. Nature 415(6868): 168–171
Zeugolis DI, Paul RG, Attenburrow G (2008) Factors influencing the properties of reconstituted collagen fibers prior to self-assembly: animal species and collagen extraction method. J Biomed Mater Res A 86(4): 892–904. doi:10.1002/jbm.a.31694
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wagenseil, J.E. A constrained mixture model for developing mouse aorta. Biomech Model Mechanobiol 10, 671–687 (2011). https://doi.org/10.1007/s10237-010-0265-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10237-010-0265-z